The Robot Will See Your Prescription Now

AI in Pharma: Cut Drug Discovery Timelines by 40% and Save $25B
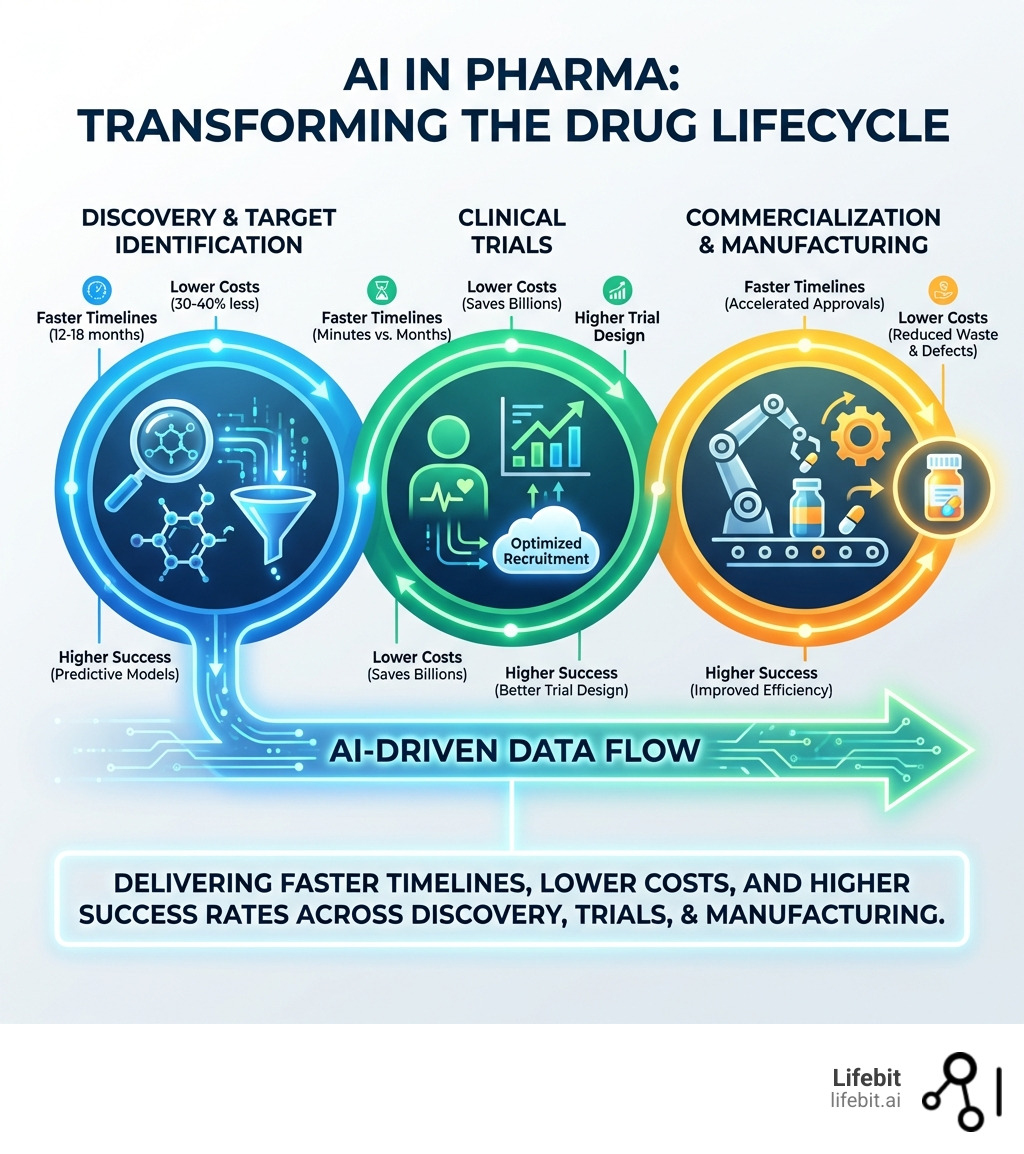
AI in pharma is revolutionizing drug discovery, clinical trials, and manufacturing—cutting preclinical timelines by up to 40%, reducing clinical trial costs by $25 billion, and accelerating new drug approvals from over a decade to as little as 12 months.
Quick Answer: What AI in Pharma Does
- Drug Discovery: Reduces time from 5-6 years to 12-18 months and cuts costs by 30-40%
- Clinical Trials: Saves $25 billion through optimized patient recruitment and trial design
- Manufacturing: Improves production efficiency by 15% and reduces waste by 25%
- Market Growth: Expected to reach $16.49 billion by 2034 (27% CAGR from 2025)
- Adoption Rate: 95% of pharmaceutical companies are now investing in AI capabilities
The pharmaceutical industry faces a brutal reality: developing a single drug costs over $2.6 billion and takes 14.6 years on average. Nine out of ten drug candidates fail in clinical trials. This isn’t sustainable.
AI is changing that equation. Fast.
By 2025, 30% of new drugs will be discovered using AI. Companies like Pfizer, AstraZeneca, and Roche aren’t just experimenting—they’re rebuilding their entire R&D pipelines around artificial intelligence. The global AI in pharmaceutical market, valued at $1.94 billion in 2025, is projected to explode to $16.49 billion by 2034.
But this isn’t just about speed. AI is making drug development smarter. It’s predicting which molecules will work before they enter expensive clinical trials. It’s identifying the right patients for studies in minutes instead of months. It’s catching manufacturing defects before they become batch failures.
The companies that master AI now will dominate the next decade of pharmaceutical innovation. The rest will struggle to compete.
Yet critical challenges remain: data silos trap valuable patient information, regulatory frameworks lag behind technology, and most pharmaceutical companies lack the infrastructure to deploy AI at scale across federated, compliant environments.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over a decade building federated AI platforms that unlock siloed genomic and clinical data for pharmaceutical research. Having contributed to breakthrough tools like Nextflow and worked with global health organizations, I’ve seen how AI in pharma transforms from theoretical promise to measurable impact when the right infrastructure exists.
Glossary for AI in pharma:
Stop Losing Ground: How AI in Pharma is Driving a $16B Market Explosion
The numbers don’t lie: the era of “tinkering” with algorithms is over. We are currently witnessing a massive structural shift in how medicine is conceived and created. The global AI in pharma market is estimated at $1.94 billion in 2025 and is forecasted to reach a staggering $16.49 billion by 2034. This represents a fundamental pivot from traditional R&D models to a “TechBio” approach where data is the primary asset.
This isn’t just organic growth; it’s an arms race. With a compound annual growth rate (CAGR) of 27%, the industry is pouring capital into AI-driven drug discovery and biopharma data analytics to solve the “Eroom’s Law” problem. Eroom’s Law—the inverse of Moore’s Law—observes that drug discovery is becoming exponentially slower and more expensive despite technological advances. AI is the only tool capable of inverting this trend by automating the analysis of biological complexity that has previously baffled human researchers.
Strategic readiness is now the primary differentiator. According to the Statista AI readiness index 2023, companies like Roche are leading the pack, not just by hiring data scientists, but by acquiring tech-first firms and integrating AI drug discovery platforms into their core identity. This shift is moving the industry away from the “blockbuster” drug model toward precision medicine, where treatments are tailored to specific genetic subpopulations.
How Leading Pharma Innovators Are Using AI
We see a clear divide emerging between “redesigners” and “tinkerers.” The redesigners—major players like Janssen, AstraZeneca, and Pfizer—are fundamentally reimagining their workflows. They are moving beyond pilot projects to enterprise-wide deployments that touch every stage of the value chain.
For instance:
- Janssen has launched over 100 AI projects, using platforms like Trials360.ai to streamline everything from patient recruitment to discovery. Their focus on “data-first” research has allowed them to identify novel targets for complex diseases like multiple myeloma.
- AstraZeneca is leveraging collaborations with firms like Qure.ai to develop treatments for chronic kidney disease and pulmonary fibrosis, effectively merging biological expertise with machine learning precision. They are also using AI to analyze vast biobanks, identifying genetic variants that correlate with disease progression.
- Pfizer utilized AI to optimize vaccine trials during the pandemic, proving that under pressure, AI can shrink timelines that used to be measured in years down to months. This success has become the blueprint for their future oncology and immunology pipelines.
- Sanofi recently announced a “large-scale” partnership with AI-native firms to build a pipeline of first-in-class and best-in-class small molecules, signaling that even the most established giants are now betting their future on algorithmic discovery.
This shift requires more than just buying a software license. It demands biopharma data integration across 5 continents, ensuring that researchers in London, New York, and Singapore can collaborate on the same population genomics datasets without compromising security. The goal is a unified data fabric that allows for real-time insights across the entire global organization.

AI in Pharma: Move from Biological Hypothesis to Lead Candidate 40% Faster
Traditional drug development is a game of expensive “guess and check.” It takes 14.6 years and around $2.6 billion to bring a molecule to market. AI is effectively a time machine for this process, replacing physical trial-and-error with high-fidelity digital simulations.
By using AI-powered target identification, researchers can now save up to 40% of time and 30% of costs when moving from a biological hypothesis to a preclinical candidate. In fact, experts predict that by 2025, 30% of new drugs will be discovered using AI. This isn’t just about speed; it’s about exploring a “chemical space” that is far too vast for humans to navigate alone. There are an estimated 10^60 potential drug-like molecules; AI allows us to search this space with surgical precision.
AI for Protein Structure Prediction: Faster, Smarter, Cheaper
One of the biggest hurdles in drug design has always been understanding the 3D shape of proteins. If you don’t know the shape of the “lock,” you can’t design the “key” (the drug). For decades, this required expensive X-ray crystallography or cryo-electron microscopy.
Tools like AlphaFold and Genie have turned this decades-long problem into a computational task.
- AlphaFold 3 uses Deep Neural Networks (DNN) to predict not just protein folding, but the interactions between proteins, DNA, RNA, and ligands with accuracy that rivals physical lab experiments. This allows researchers to see how a drug might bind to its target in a complex cellular environment.
- Genie and other diffusion models go a step further, using generative AI to design entirely new proteins that don’t even exist in nature. This “de novo” design capability is opening doors to treating “undruggable” targets that lack natural binding sites.
This allows for leveraging AI for target validation at an unprecedented scale. Instead of spending months in a wet lab, we can now run millions of simulations to see how a drug interacts with its target before a single pipette is touched. This “dry lab” first approach ensures that only the most promising candidates ever reach the expensive “wet lab” phase.
High-Speed Molecular Design: From Idea to Lead in Days
We are moving toward an era of “generative chemistry”—where AI generates novel molecular structures from scratch based on desired properties like solubility, toxicity, and metabolic stability. Platforms like the Centaur Chemist platform have already demonstrated that AI can design a drug candidate and get it ready for clinical trials in just 12 to 18 months, compared to the traditional 5-year timeline.
Traditional vs. AI-Driven Drug Development
| Metric | Traditional Method | AI-Enabled Method |
|---|---|---|
| Time to Market | 10–15 Years | 5–7 Years |
| Discovery Cost | ~$2.6 Billion | 30–40% Reduction |
| Preclinical Success | ~10% | Significantly Higher (Projected) |
| Target ID Time | 2–3 Years | Weeks to Months |
| Chemical Space Search | Limited/Manual | Exhaustive/Algorithmic |
This high-speed design is powered by end-to-end drug discovery workflows that integrate real-world data and biomarker discovery. By using Knowledge Graphs to connect disparate data points from scientific literature, clinical notes, and genomic databases, AI can identify hidden relationships between genes and diseases that were previously invisible to the human eye.
End Trial Failure: How AI in Pharma Saves $25B in Clinical Costs
The clinical trial phase is where most drugs go to die. It is also the most expensive part of the journey, accounting for nearly 60% of total R&D costs. AI involvement in clinical development is projected to lead to up to $25 billion savings globally by reducing the high rate of trial attrition.
By optimizing trial design and using real-time data analysis, AI can cut down clinical trial duration by up to 10% without compromising data integrity. This is the difference between a pharmaceutical company being first to market or losing billions to a competitor. Furthermore, the FDA Modernization Act 2.0 has paved the way for AI by allowing for alternatives to animal testing, such as computer modeling and organ-on-a-chip technology, which AI can analyze at scale.
AI-Powered Patient Recruitment: No More Empty Trials
Did you know that 25% of studies fail simply because they can’t find enough patients? It’s a heartbreaking reason for a potentially life-saving drug to stall. AI tools like TrialGPT are changing this by:
- Automated Screening: Scanning thousands of Electronic Health Records (EHRs) in seconds to match patients to specific trial criteria, including complex inclusion/exclusion parameters that humans might miss.
- Diversity Optimization: Ensuring cohorts are representative of the global population. AI can identify underserved communities and suggest recruitment sites that improve the ethnic and geographic diversity of the trial, a growing requirement for regulatory approval in the US and Europe.
- Predictive Enrollment: Modeling which sites are likely to recruit the fastest based on historical performance and local disease prevalence, allowing resources to be allocated where they will have the most impact.
Decentralized Trials and Digital Twins: Fewer Patients, Better Data
The future of research is moving away from centralized hospital hubs. Decentralized Clinical Trials (DCTs) allow patients to participate from home, monitored by wearable sensors and AI-driven apps. This reduces the burden on patients and leads to higher retention rates.
But perhaps the most “sci-fi” advancement is the use of digital twins. These are virtual models of patients used to simulate how a person might respond to a drug. In a landmark study on Alzheimer’s disease, digital twins were used to create Synthetic Control Arms (SCAs). This reduced the number of actual human participants required in control arms by 35%, as the AI could accurately predict the disease progression of a placebo group.
Think about that: we can achieve better results with fewer people, lower costs, and less risk to patients. This is the future of drug development—what we call Drug Discovery 2.0. By using AI to monitor trials in real-time, companies can also identify safety signals much earlier, potentially stopping a failing trial before costs spiral out of control.
AI in Pharma: Slash Production Waste by 25% and End Batch Failures
The transformation doesn’t stop once the drug is discovered. Pharmaceutical manufacturing is a high-stakes environment where a single “batch deviation” can cost millions and lead to life-threatening drug shortages. Currently, 79% of pharmaceutical executives believe that intelligent automation will have a significant impact on their industry by 2030.
We are moving toward “Pharma 4.0,” where factories are self-correcting and supply chains are transparent. This involves the integration of the Internet of Medical Things (IoMT), where every piece of equipment on the factory floor is connected and monitored by AI.
Key AI applications in manufacturing include:
- The “Golden Batch”: AI analyzes historical data from thousands of successful runs to identify the perfect conditions (temperature, pressure, humidity, flow rates) for a successful batch. It then uses digital twins of the manufacturing process to replicate those conditions every single time, adjusting in real-time if sensors detect a drift.
- Predictive Maintenance: Instead of waiting for a machine to break or performing maintenance on a fixed schedule, AI sensors predict failure weeks in advance by analyzing vibration and heat patterns. This prevents costly downtime and ensures continuous production.
- Waste Reduction: AI-based methods can reduce production waste by up to 25% by optimizing chemical synthesis and solvent recovery. This not only saves money but also helps pharmaceutical companies meet their increasingly stringent ESG (Environmental, Social, and Governance) goals.
- Continuous Manufacturing: AI is enabling the shift from batch manufacturing to continuous manufacturing, where drugs are produced in a single, uninterrupted flow. This is more efficient, requires a smaller physical footprint, and allows for much tighter quality control.
- Counterfeit Resistance: AI-powered computer vision can track packaging and labeling at speeds impossible for human inspectors. By analyzing microscopic patterns in packaging, AI can ensure that only genuine medicine reaches patients, protecting both revenue and public health.
By achieving just 1/6th of the deviations per 1,000 batches compared to average competitors, top-performing manufacturers are seeing 14 times lower quality costs. This is the power of AI-powered drug development.
AI in Pharma: Why You Need Trilingual Teams, Not Just Algorithms
A common fear is that “The Robot” will replace the scientist. But the reality of AI in pharma is much more collaborative. AI is an assistive technology, not a replacement. It excels at processing data at a scale humans cannot, but it lacks the intuition and ethical judgment required for medical breakthroughs.
We still need humans to:
- Interpret the “Why”: AI is great at finding patterns (the “what”), but it often lacks the biological context to explain the “why.” A scientist must determine if a correlation found by AI is biologically relevant or just a statistical fluke.
- Solve the Black Box: Many AI models, particularly deep learning models, are opaque. Regulatory bodies like the FDA and EMA require “explainable AI” (XAI). Scientists must be able to explain the logic behind an AI’s prediction to ensure that decisions are based on sound science, not just algorithmic correlations. This is critical for safety and regulatory compliance.
- Handle Ethics and Bias: If an AI is trained on data from only one ethnic group, its predictions will be biased. Human oversight is mandatory to ensure secure data solutions and equitable outcomes. We must ensure that AI-driven medicine works for everyone, not just the populations represented in existing datasets.
The workforce is evolving. We don’t need fewer scientists; we need “trilingual” professionals who understand biology, data science, and business strategy. This new breed of professional can bridge the gap between a complex biological problem and a technical AI solution.
Furthermore, the rise of Federated Learning is changing how these teams work. Federated learning allows AI models to be trained on sensitive patient data across different institutions without the data ever leaving its original location. This preserves patient privacy and meets strict GDPR/HIPAA standards while still allowing for the massive datasets required for high-accuracy AI. This is why scaling biomarker discovery workflows now requires a combination of human expertise and federated AI infrastructure.
AI in Pharma: 4 Critical Questions Every Leader Must Answer
What is the projected growth of AI in the pharmaceutical industry?
The market is exploding. From a base of roughly $1.8 billion in 2023, the global AI in pharma market is expected to reach $16.49 billion by 2034. This is driven by 95% of companies now investing in AI capabilities to offset the rising costs of traditional R&D and the looming “patent cliff” where many blockbuster drugs will lose exclusivity.
Can AI fully replace human roles in drug development?
No. AI is primarily used for decision support and accelerating manual tasks. Regulatory requirements and the complexity of biological systems mean that human intervention is—and will remain—mandatory for the foreseeable future. AI acts as a “co-pilot” for researchers, handling the data-heavy lifting so scientists can focus on high-level strategy and creative problem-solving.
What is the difference between Predictive AI and Generative AI in pharma?
Predictive AI is used to forecast outcomes based on existing data—for example, predicting which patients are most likely to respond to a specific treatment. Generative AI, on the other hand, creates new data or structures—such as designing a completely new molecular structure that has never been seen before. Both are essential: Predictive AI helps us choose the right path, while Generative AI helps us build the tools to walk it.
What are the biggest trends for AI in biotech for 2025?
We expect to see four major trends:
- Generative AI for Biologics: Moving beyond small molecules to designing entirely new classes of complex biologics, including antibodies and mRNA therapies.
- Federated Learning at Scale: Allowing global collaborations on rare disease data without moving sensitive information across borders, preserving data sovereignty.
- AI-Driven Precision Medicine: Using genomic data insights to tailor treatments to individual patients. We are moving toward a “N-of-1” trial model where the drug is optimized for a single person’s genetic makeup.
- Regulatory-Grade AI: The development of standardized frameworks for AI validation, making it easier for companies to get AI-discovered drugs through the FDA and EMA approval processes.
AI in Pharma: Unlock Siloed Data and Lead the $16B Transformation
The pharmaceutical industry has reached a tipping point. The old way of developing drugs—long, expensive, and siloed—is dead. What’s replacing it is a faster, smarter, and more collaborative model powered by AI in pharma.
At Lifebit, we believe the greatest bottleneck to this revolution isn’t the algorithms—it’s the data. Most of the world’s most valuable biomedical data is locked away in silos for security and regulatory reasons. Our Lifebit Federated AI Platform solves this by bringing the AI to the data, rather than the data to the AI.
By providing a Trusted Research Environment (TRE) and a Trusted Data Lakehouse, we enable biopharma companies to collaborate securely across borders, powering the AI revolution in drug development.
Whether you are identifying a new target for a rare disease or optimizing a global supply chain, the message is clear: the robot is ready to see your prescription. The only question is, are you ready to lead the transformation?
Learn more about our solutions for commercial pharma and join us in supercharging the journey from lab to life.

