The Patient Will See You Now: Patient Driven Registry Software

Patient Driven Registry Software: Build Research-Ready Datasets 10x Faster
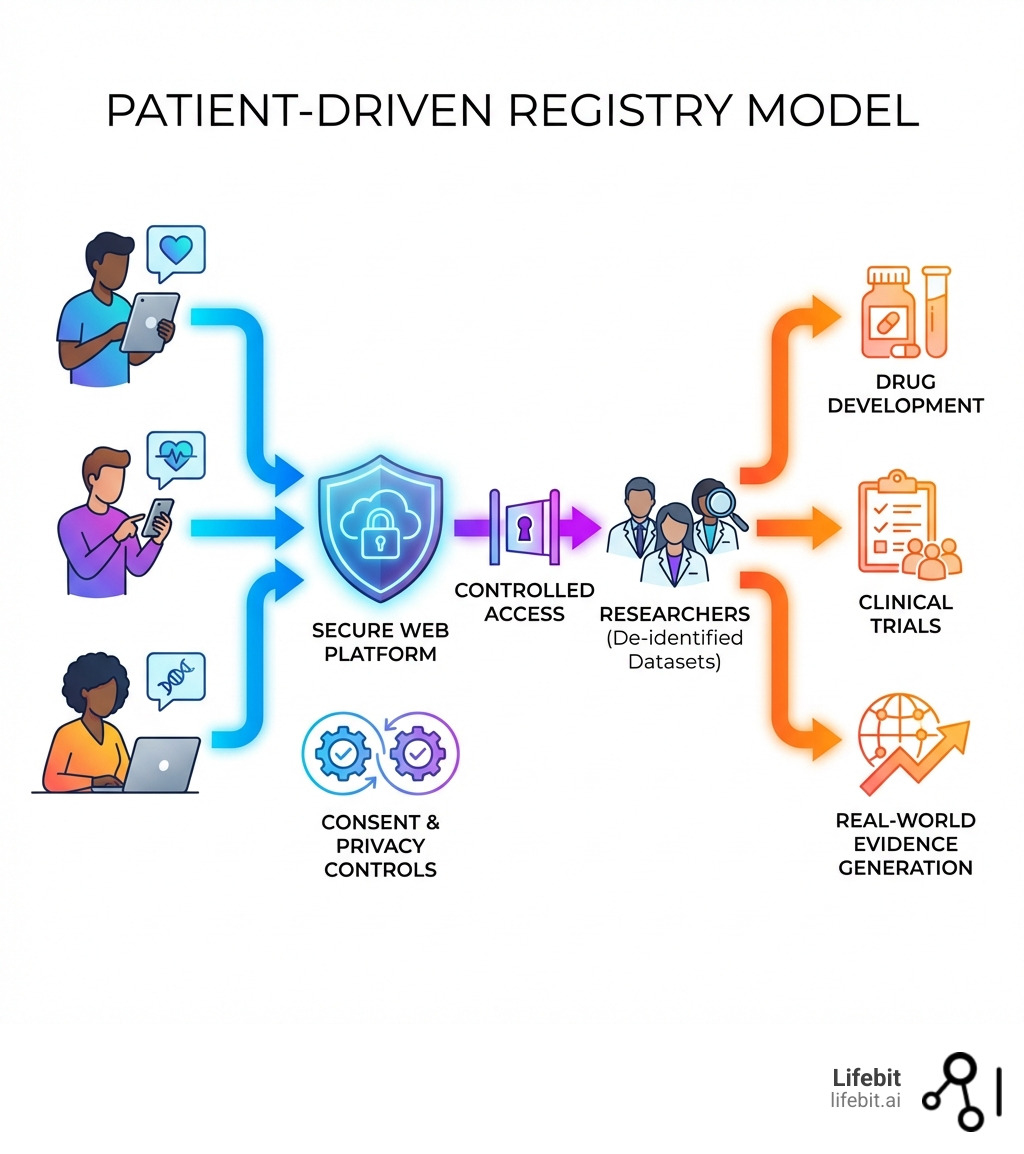
Patient driven registry software is a digital platform that enables patients, guardians, or caregivers to directly register, consent, and contribute their health data for research—without requiring clinic visits or researcher intermediaries. Key characteristics include:
- Direct patient enrollment: Individuals register themselves via web-based portals
- Self-reported data: Patients enter clinical, symptom, and treatment information
- Dynamic consent management: Participants control what data they share and with whom
- Global accessibility: Web-based platforms allow participation from any location
- Longitudinal tracking: Systems collect data over months or years to study disease progression
Traditional registries rely on clinicians to extract data from medical records and enter it into databases. Patient-driven registries flip this model—placing patients at the center of data collection. This shift is particularly powerful for rare diseases, where patients often know more about their condition than their local providers and are geographically dispersed across continents.
The Global Angelman Syndrome Registry demonstrated this potential by achieving 286 patient registrations from 32 countries in just eight months after deployment—a reach impossible with traditional clinic-based approaches. For drug developers, these registries create research-ready natural history datasets that previously took years and millions of dollars to assemble through in-person studies.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over a decade building federated data analysis platforms that power secure, compliant patient driven registry software for pharmaceutical organizations and public sector institutions worldwide. Through this work, we’ve seen how patient-led data collection accelerates rare disease research and transforms clinical trial recruitment.
Handy patient driven registry software terms:
Why Traditional Registries Are Dead—And How Patient Driven Registry Software Fixes It
To understand why patient driven registry software is making such waves, we first have to look at what it’s replacing. For decades, patient registries were essentially static filing cabinets. A doctor would see a patient, write down notes, and—if the patient was lucky enough to be part of a study—a research coordinator would manually transcribe those notes into a database.
This “traditional” model is researcher-centric. The institution owns the data, the clinician controls the entry, and the patient is often left in the dark about how their information is being used.
In contrast, a patient-driven model utilizes specialized software to cut out the middleman. Here is how they differ:
- Data Ownership and Control: In a patient-driven setup, the individual (or their caregiver) holds the keys. They decide which research projects can access their “digital twin” of data.
- Direct Entry: Instead of waiting for a hospital bill or an EHR update, patients enter real-world data directly. This includes Patient-Reported Outcomes (PROs) like daily pain levels, sleep quality, or medication side effects that a doctor might never see in a 15-minute clinic visit.
- Observational Reach: Traditional registries are often limited to a single hospital or network. Patient-driven software is web-based, meaning a family in rural Canada can contribute to a study based in New York just as easily as someone living next door to the lab.
As the NIH registry directory suggests, these databases are vital for understanding the “natural history” of a disease—basically, how it behaves in the real world over time. When we ask what are patient registries, we are really talking about the foundation of modern evidence-based medicine.
Empowering Individuals with Patient Driven Registry Software
The true magic of this software isn’t just the data—it’s the empowerment. When a patient uses a registry portal, they transition from a “subject” to a “partner.”
This software allows for:
- Self-Management: Many platforms provide patients with dashboards to track their own symptoms over time, helping them manage their condition more effectively.
- Autonomy: Through dynamic consent, patients can opt-in or opt-out of specific substudies. If they decide they no longer want to participate in a genetic study but want to remain in a lifestyle survey, they can change that with a click.
- Bridging the Gap: We often discuss what are patient registries why are they important in the context of clinical gaps. Patients live with their diseases 24/7; clinicians see them twice a year. Patient driven registry software captures the 99% of the patient experience that happens outside the hospital walls.
Stop Chasing Patients: Recruit Rare Disease Cohorts in Seconds
If you are researching a common condition like hypertension, you have millions of patients to study. If you are researching a rare disease like Angelman Syndrome or Gaucher Disease, your “n” (the number of patients) is tiny and scattered across the globe.
This is where patient driven registry software becomes a literal lifesaver. By removing geographical barriers, patient-led registries allow advocacy groups to build a “critical mass” of data.
- Clinical Recruitment: Finding 50 patients for a rare disease trial used to take years. With a patient-driven registry, researchers can send a notification to a pre-screened, “research-ready” cohort in seconds.
- Natural History Studies: The FDA patient-focused data guidance emphasizes that drug development must be informed by the patient’s perspective. Registries provide the longitudinal data needed to show the FDA that a drug actually improves a patient’s quality of life, not just their lab results.
There are four benefits of patient registries rare disease communities value most: faster diagnosis, better care coordination, accelerated drug development, and a stronger community voice.
Accelerating Breakthroughs with Real-World Evidence
Real-World Evidence (RWE) is the “gold standard” of the modern era. While clinical trials happen in a controlled “bubble,” RWE shows how a drug works in the messy, complicated real world.
Patient driven registry software is the primary engine for RWE. It allows researchers to:
- Use External Control Arms: Instead of giving half the participants a placebo in a trial, researchers can use registry data from untreated patients as a “virtual” control group. This is more ethical and much faster.
- Support Drug Approvals: The FDA and EMA are increasingly accepting registry data to support label expansions or new drug applications.
- Track Post-Market Safety: Once a drug is on the market, registries can monitor thousands of patients for rare side effects that didn’t show up in smaller trials.
For a deeper dive, our rare diseases registry complete guide explores how these data points move from a patient’s smartphone to a regulatory submission.
5 Security Features Your Patient Driven Registry Software Is Missing
Building a registry isn’t just about making a pretty website; it’s about building a fortress for sensitive data. When we evaluate patient driven registry software, we look for five non-negotiable features:
- Dynamic Consent: The ability for patients to update their permissions in real-time.
- Multilingual Support: To be truly global, the software must speak the patient’s language—literally.
- Strict Compliance: Adherence to HIPAA (USA), GDPR (Europe), and ISO 27001 standards is mandatory.
- Interoperability: The software must “talk” to other systems using standards like FHIR and OMOP.
- Data Quality Tools: Automated validation (e.g., preventing someone from entering a birth year of 2029) ensures the data is actually usable for science.
Registry Deployment Options: SaaS vs. On-Premise
| Feature | SaaS (Cloud-Hosted) | On-Premise / Private Cloud |
|---|---|---|
| Speed to Launch | Very Fast (Weeks) | Slower (Months) |
| Maintenance | Handled by Vendor | Handled by In-house IT |
| Data Control | Shared with Provider | Total Institutional Control |
| Scalability | High/Elastic | Limited by Hardware |
| Best For | Advocacy Groups, Startups | Large Governments, Hospitals |
Establishing high quality patient registries requires choosing the deployment model that matches your regulatory burden. At Lifebit, we often recommend a hybrid approach: the ease of a web interface with the security of a Trusted Research Environment (TRE).
Future Outlook for Patient Driven Registry Software
The future of these registries is “interoperable.” We are moving away from data silos and toward a world where your registry data can be securely linked with your electronic health records (EHR) and even your wearable device data (like Fitbit or Apple Watch).
Standards like FHIR (Fast Healthcare Interoperability Resources) and the OMOP Common Data Model are the “universal translators” making this possible. When you choose patient registry software, ensuring it supports these standards is the only way to “future-proof” your research.
Stop Losing Data: Fix the 3 Biggest Registry Hurdles
It’s not all sunshine and data points. Launching a patient-driven registry comes with significant hurdles.
- Data Silos: Many organizations still struggle to share data across borders due to strict sovereignty laws.
- Funding Sustainability: Registries are expensive to maintain. While a grant might cover the launch, long-term success requires a business model—often through partnerships with biopharma companies looking for RWE.
- Engagement Drop-off: Collecting data once is easy. Keeping patients engaged for five years is hard. Successful registries use “gamification,” educational content, and regular “data give-backs” to keep families involved.
We’ve analyzed the challenges facing patient registries in the us, and the consensus is clear: governance is the most important factor. You need a board that includes patients, clinicians, and ethicists to ensure the registry remains trustworthy.
Analyze Global Data Without Moving It: Federated AI for Registries
The most exciting frontier in patient driven registry software is Federated Analytics.
Historically, to analyze data from three different countries, you had to physically move that data to one central server. This is a nightmare for privacy and compliance (especially under GDPR).
Lifebit’s federated approach flips this: we bring the analysis to the data.
By using federated learning and AI, researchers can:
- Identify “hidden” patient cohorts across global datasets without ever seeing a patient’s name.
- Integrate multi-omic data (genomics, proteomics) with clinical registry data to find new biomarkers.
- Run predictive AI models to see which patients are at the highest risk of disease progression.
To see if your current system measures up, you can evaluate your registry analytics using this checklist. For a look at how this works in the real world, check out this use case on patient registries.
Patient Driven Registry Software: Privacy and Speed Questions Answered
How is patient data kept private in these registries?
Data privacy is handled through layers: Encryption (locking data during transit and at rest), De-identification (removing names and IDs), and Federated Access (allowing researchers to “ask questions” of the data without ever seeing or downloading the raw files).
How long does it take to launch a patient-driven registry?
A simple “Minimum Viable Product” (MVP) registry can be launched in 3–6 months. However, complex, multi-site registries with deep regulatory requirements typically take 12–18 months to fully mature.
What is the difference between a registry and a clinical trial?
A clinical trial is a controlled experiment where researchers give a specific treatment to see if it works. A registry is observational—it simply records what is happening to patients in their normal lives. Registries are often used to find patients for trials or to track a drug’s performance after a trial ends.
The End of Passive Research: Launch Your Registry Now
The era of patients being passive participants in research is over. Patient driven registry software has turned the healthcare hierarchy on its head, giving individuals the power to drive the cures they need.
At Lifebit, we believe that the most valuable data in the world shouldn’t be locked in a silo. Our next-generation federated AI platform is designed to make this global, patient-led data accessible, secure, and research-ready. By providing a Trusted Research Environment (TRE), we allow researchers to collaborate across five continents—from New York to Singapore—without ever compromising patient privacy.
The patient is ready to see you now. Are you ready to listen to their data?

