AI for Clinical Trials: Optimizing Recruitment, Smarter Trials

Cut Recruitment Time by 50% with AI Clinical Trial Recruitment
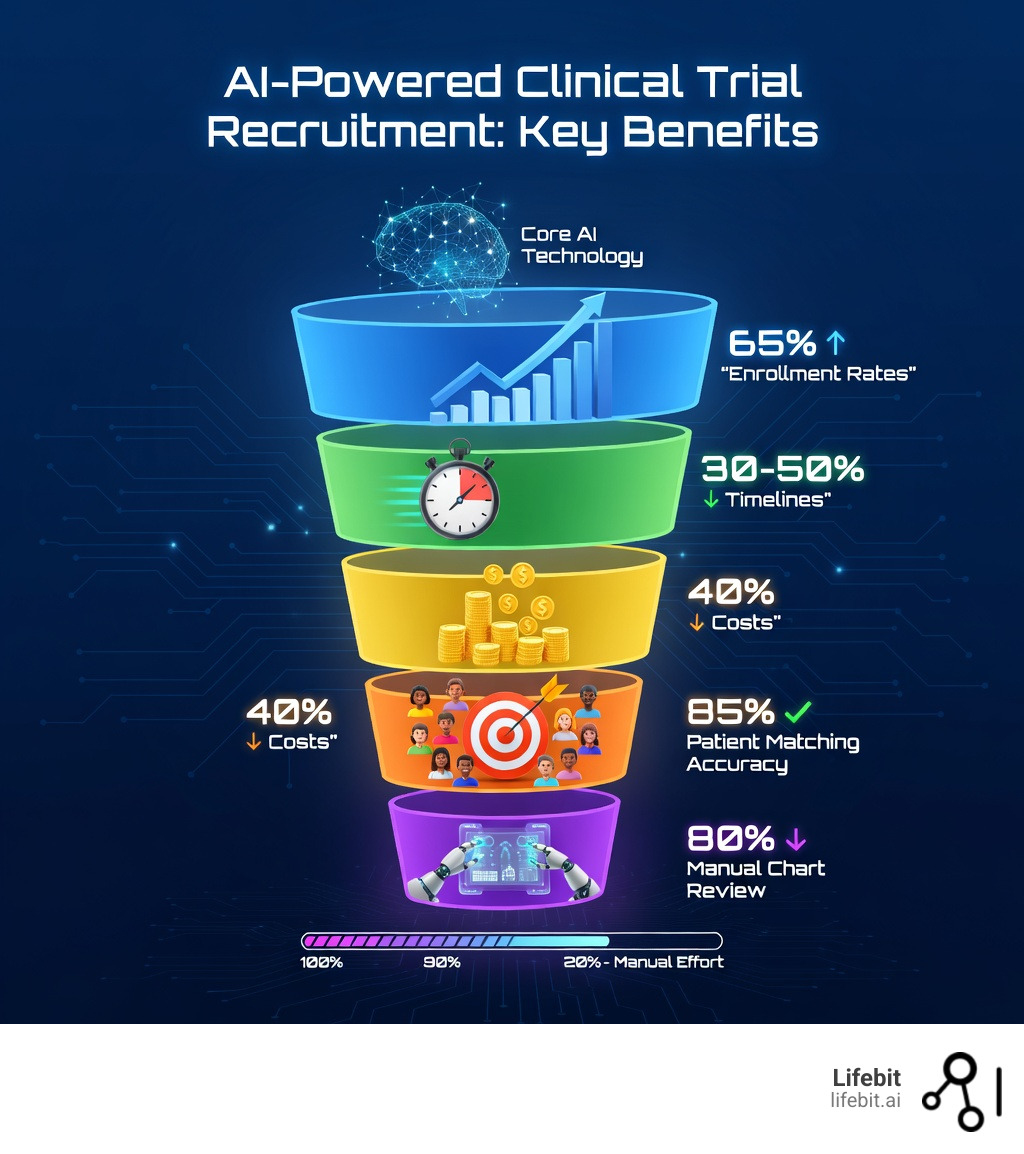
AI clinical trial recruitment is changing how pharmaceutical companies and research institutions find eligible patients, using machine learning and natural language processing to automatically screen electronic health records, predict enrollment likelihood, and match participants to trials—cutting recruitment time by 30-50% and improving enrollment rates by 65%.
Key benefits of AI clinical trial recruitment:
- Faster patient identification – AI screens thousands of medical records in minutes instead of weeks
- Higher accuracy – Predictive models achieve 85% accuracy in matching patients to eligibility criteria
- Cost reduction – AI integration reduces trial costs by up to 40% through automation
- Better diversity – Algorithms identify underrepresented populations missed by manual methods
- Reduced manual workload – AI cuts chart review requirements by 40-85% depending on the application
More than 80% of clinical trials struggle to meet recruitment goals on time. This bottleneck delays potentially life-saving treatments by months or years, adds hundreds of millions in costs, and forces many studies to be abandoned entirely. Manual screening of electronic health records is time-consuming, error-prone, and often excludes eligible patients due to overly restrictive criteria copied from previous studies.
AI addresses these challenges by analyzing vast datasets—from genomics to social media—to identify suitable participants faster and more inclusively than traditional methods. Natural language processing extracts eligibility information from unstructured medical notes, while machine learning predicts which patients are most likely to enroll and complete the trial. The result: recruitment timelines accelerate by 30-50%, costs drop by 40%, and more diverse patient populations gain access to experimental therapies.
I’m Dr. Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve built a federated genomics platform that powers AI clinical trial recruitment across secure, compliant environments for global pharmaceutical and public health organizations. With over 15 years in computational biology, AI, and health-tech, I’ve seen how the right technology can break down data silos and accelerate precision medicine at scale.
Ai clinical trial recruitment basics:
- clinical trial recruitment strategies
- digital clinical trial recruitment
- patient recruitment in clinical trials
The Enrollment Crisis: Why 80% of Clinical Trials Fail
The pharmaceutical industry is currently struggling with what researchers call “Eroom’s Law”—the frustrating observation that drug discovery is becoming slower and more expensive over time, despite massive technological gains. Today, bringing a single medication to market can cost over $1 billion and take a decade. Clinical trials are the biggest drain on these resources, accounting for half of the total time and money spent.
The most significant hurdle? Recruitment. Statistics show that more than 80% of clinical trials fail to meet their original enrollment timelines. This isn’t just a scheduling issue; it’s a systemic failure. When trials can’t find enough patients, the study’s statistical power is weakened, costs skyrocket, and life-changing medications remain stuck in the lab.
The Protocol Complexity Trap
One reason for this crisis is the increasing complexity of clinical protocols. Over the last decade, the average number of endpoints in a Phase III trial has increased by 86%. This means eligibility criteria have become so narrow that the “ideal” patient often doesn’t exist in the real world. Traditional recruitment relies on manual screening—a process where clinical research coordinators painstakingly comb through patient charts. This method is:
- Incredibly Slow: For example, standard prescreening for breast cancer patients takes an average of 19 days, while lung cancer can take a staggering 263 days.
- Inaccurate: Manual reviews are prone to human error and often miss eligible candidates buried in unstructured data. Research indicates that up to 50% of eligible patients are overlooked during manual chart reviews.
- Restrictive: Many trials use “copy-paste” eligibility criteria from previous studies. Research shows that 80% of advanced non-small cell lung cancer patients are excluded by criteria that may not even be necessary for safety, such as specific lab values that don’t impact the drug’s efficacy.
The Site Selection Paradox
Furthermore, the industry suffers from a “site selection paradox.” Roughly 70% of clinical trials are conducted at the same 5% of investigative sites. This leads to “patient fatigue” at top-tier academic centers while community hospitals, where the vast majority of patients receive care, remain untapped. Without AI to identify where the patients actually are, sponsors continue to pour money into sites that fail to recruit a single participant.
According to scientific research on drug development costs, the number of new drugs approved per billion dollars in R&D spending has halved every nine years for the last six decades. We are essentially running faster just to stay in the same place, and recruitment is the primary friction point holding the industry back.
How AI Clinical Trial Recruitment Matches Patients in Minutes, Not Weeks
This is where ai clinical trial recruitment steps in to flip the script. By leveraging machine learning (ML) and natural language processing (NLP), we can move from manual “hunting” to automated “matching.”
AI doesn’t just look at structured data like age or gender. It dives into the “messy” parts of medical records—physician progress notes, pathology reports, and radiology summaries. Advanced AI matching platforms have demonstrated a 24% to 50% increase in the number of patients accurately identified as eligible compared to standard practices. More impressively, what takes humans weeks can be done by AI in mere minutes.
The Power of Natural Language Processing (NLP)
NLP is the engine behind modern recruitment. It uses techniques like Named Entity Recognition (NER) to identify specific medical conditions, medications, and lab results within unstructured text. Crucially, advanced NLP can handle “negation” and “temporality.” For instance, it can distinguish between a patient who “has a history of heart failure” (exclusion) and a patient whose “mother had heart failure” (eligible). This level of nuance is impossible for basic keyword searches but is handled effortlessly by Large Language Models (LLMs) trained on medical corpora.
According to scientific research on AI trial acceleration, AI integration can accelerate trial timelines by 30–50%. When you consider that every day of delay can cost a sponsor millions in lost revenue and operational overhead, the value proposition is clear.
Real-World Evidence (RWE) Integration
AI also allows for the integration of Real-World Evidence (RWE) into the recruitment strategy. By analyzing data from insurance claims, pharmacy records, and even wearable devices, AI can build a 360-degree view of the patient. This helps in:
- Predictive Analytics: Modern models achieve 85% accuracy in predicting trial outcomes and enrollment likelihood. This helps sponsors choose the right sites and strategies before the first patient is even screened.
- Digital Biomarkers: AI can process data from wearables to detect adverse events with 90% sensitivity, making trials safer and more appealing to participants.
- Broadening Access: Algorithms like Stanford’s “Trial Pathfinder” analyze EHRs to suggest loosening restrictive criteria. In lung cancer trials, this approach doubled the pool of eligible patients without increasing the risk of adverse events.
Categorizing the current AI tool landscape
The world of ai clinical trial recruitment is diverse. We generally categorize these tools into four buckets:
- Medical Tools: Specialized software that integrates directly into clinical workflows to automate patient identification at the point of care.
- Traditional Model-Based Approaches: These make up about 43% of current studies and use established statistical ML (like Random Forests or SVMs) to find patterns in structured data.
- Deep Learning Models: A smaller but rapidly growing segment (about 10%) that uses neural networks to handle complex, multimodal data, including medical imaging and genomic sequences.
- Text-Mining & NLP: Automated systems that reduce the number of patient notes needing manual review by up to 85%, allowing coordinators to focus only on the most likely candidates.
Why Deep Learning Is Replacing Traditional AI in Clinical Trials
While the benefits are clear, we must be honest about the problems. The biggest technical challenge is the “black box” problem. Many deep learning models are incredibly accurate but difficult to explain. In a regulated industry like healthcare, “the computer said so” isn’t an acceptable answer for eligibility. However, the shift toward Deep Learning is inevitable because of the sheer volume of unstructured data.
Generative AI and Synthetic Control Arms
One of the most transformative applications of deep learning is the creation of Synthetic Control Arms (SCAs). By using AI to generate a “digital twin” of a control group based on historical trial data and RWE, researchers can reduce the number of patients who need to be recruited for the placebo arm. This not only speeds up recruitment but also makes the trial more ethical, as more participants receive the active treatment. Deep learning models like Generative Adversarial Networks (GANs) are now being used to ensure these synthetic patients are statistically indistinguishable from real ones.
Furthermore, data quality remains a bottleneck. AI is only as good as the data it’s fed, and currently, data quality issues affect roughly 50% of clinical trial datasets. We are seeing a major shift from traditional models toward deep learning and medical tools that can handle this complexity through “data cleaning” algorithms that automatically identify and correct inconsistencies in EHR records.
Automated EHR Text-Mining in Practice
One of the most exciting developments is the use of automated EHR text-mining. For cardiovascular trials, this has shown a 79.9% reduction in the number of patients that need to be screened manually. By incorporating data directly from the EHR, algorithms have reduced manual chart reviews by 40.5% while ensuring no eligible patients were excluded.
| Feature | Traditional ML Models | Deep Learning Models |
|---|---|---|
| Data Type | Structured (Tables) | Unstructured (Notes, Images, Genomics) |
| Explainability | High (Decision Trees) | Low (Neural Networks/XAI required) |
| Setup Time | Months | Weeks (with pre-trained Transformers) |
| Accuracy | 70-80% | 85-95%+ |
| Manual Review Reduction | Moderate | High (up to 85%) |
Recent scientific research on medical AI efficiency warns that we should not mistake raw accuracy for operational efficiency. The tool must fit into the human workflow of the clinical site to truly save time. This is why “Human-in-the-loop” AI, where the algorithm provides a recommendation that a clinician then verifies, is becoming the industry standard.
Protect Patient Privacy: Solving the Ethics Gap in AI Recruitment
As we lean more on ai clinical trial recruitment, we must address the ethical elephant in the room. AI platforms require access to sensitive patient data, and if not handled correctly, this creates massive risks for privacy and trust. The industry is moving toward a “privacy-by-design” architecture to mitigate these concerns.
The Federated Learning Revolution
Lifebit champions federated learning as the solution to the data privacy dilemma. In traditional AI, data is moved to a central server for training. In federated learning, the AI model travels to the data. The raw patient records never leave the hospital’s secure firewall; only the “learnings” (mathematical weights) are sent back to the central model. This approach satisfies strict regulatory requirements like GDPR in Europe and HIPAA in the US, while still allowing for large-scale multi-center studies.
Managing risks in AI clinical trial recruitment
- Data Privacy: The security of confidential data is the #1 priority. Beyond federated learning, techniques like Differential Privacy add mathematical “noise” to datasets to ensure that no individual patient can be re-identified from the AI’s output.
- Selection Bias: If an AI is trained on data from only one demographic, it will inadvertently perpetuate that bias. We’ve seen cases where models are less accurate for minority groups because they weren’t represented in the training set. To combat this, AI developers are now using “bias detection” algorithms to ensure training sets are representative of the global population.
- Algorithmic Transparency: To build trust, we need “Explainable AI” (XAI). This involves using techniques like SHAP (SHapley Additive exPlanations) to show exactly which features (e.g., a specific lab value or a mention of a symptom) led the AI to match a patient to a trial.
- Informed Consent: Digital change must include clear, simple explanations of how their data is used. AI is now being used to create “dynamic consent” platforms where patients can see in real-time which studies are accessing their de-identified data and opt-out at any time.
By using AI to critique restrictive rules, we can actually make trials more inclusive. AI has helped double the enrollment of women, minorities, and older patients in certain oncology trials by proving that strict lab-test exclusions (like minor kidney function variations) were often arbitrary and did not impact safety.
Stop Trial Dropouts: Using AI for Participant Retention
Recruiting a patient is only half the battle; keeping them is the other. Historically, there has been a lack of research into using AI for participant retention, but that is changing as the industry realizes that a 30% dropout rate (the current average) can invalidate years of work.
Predictive Modeling for Attrition
ML models can now identify “high-risk” participants who might quit the trial based on their engagement patterns. For example, if a patient stops logging into the trial app or misses a non-critical check-in, the AI can flag this to the site coordinator. By analyzing historical data, AI can predict dropout with up to 90% accuracy, allowing for proactive interventions like providing transportation vouchers or adjusting the visit schedule to better suit the patient’s life.
Decentralized Clinical Trials (DCTs) and AI
The COVID-19 pandemic accelerated the shift toward Decentralized Clinical Trials (DCTs), where patients participate from home. AI is the backbone of this shift, powering:
- AI-Powered Navigators: Conversational interfaces (chatbots) that provide 24/7 support, answering patient questions about side effects or medication timing in plain language.
- Remote Monitoring: AI analyzes data from wearable sensors to detect subtle changes in a patient’s health, often before the patient even notices a symptom. This reduces the need for frequent, burdensome hospital visits.
- Simplified Jargon: AI-driven summarization tools can take a 50-page consent form and turn it into a patient-friendly summary, improving satisfaction and understanding. Studies show that patients who fully understand the trial requirements are 40% less likely to drop out.
By focusing on the patient experience through AI, we move from a “site-centric” model to a “patient-centric” model, which is the key to long-term trial success.
Stop the Confusion: AI Clinical Trial Recruitment FAQ
How does AI reduce clinical trial costs?
AI cuts costs by up to 40% primarily through automation and efficiency. It slashes the time staff spend on manual chart reviews (by up to 85%), which is one of the highest labor costs in a trial. Furthermore, it reduces the need for expensive “rescue” recruitment campaigns—which can cost hundreds of thousands of dollars per month—by ensuring enrollment stays on track from day one. Every month saved in a Phase III trial can save a sponsor between $600,000 and $8 million in operational costs.
What are the primary challenges in AI implementation?
The biggest problems are data interoperability (getting different hospital systems to talk to each other), regulatory uncertainty, and the “black box” nature of complex algorithms. There is also a significant need for high-quality, diverse training data to prevent algorithmic bias. Many institutions also face “cultural resistance,” where staff are hesitant to trust an algorithm over their own manual processes. Overcoming this requires robust validation studies and user-friendly interfaces.
Why is AI used for EHR mining?
EHR mining allows researchers to find “needles in haystacks.” AI can search millions of data points across thousands of patients in seconds. This has been shown to reduce the number of patients needing manual screening by 79.9% in cardiovascular trials and identify 24-50% more eligible patients in oncology practices that would have otherwise been missed. It is particularly effective at identifying patients with rare diseases who might only appear once in a database of 100,000 people.
Can AI help with trial diversity?
Yes. By analyzing broader datasets and identifying patients in community health settings rather than just major academic centers, AI helps reach underrepresented populations. It can also identify and suggest the removal of “proxy” exclusion criteria that disproportionately affect minority groups, such as certain socioeconomic indicators or non-essential lab requirements.
The Future of Research: Faster Cures Through Federated AI
At Lifebit, we believe the future of ai clinical trial recruitment isn’t just about better algorithms; it’s about better access. Our next-generation federated AI platform allows researchers to run advanced analytics on global biomedical data without the data ever leaving its secure home.
By using our Trusted Research Environment (TRE) and Trusted Data Lakehouse (TDL), pharmaceutical companies can collaborate with governments and public health agencies across five continents. This “data-in-place” approach solves the privacy and security challenges that have long hindered AI adoption.
Smarter trials mean faster cures. By integrating AI-driven insights and real-time evidence, we can move away from the “Eroom’s Law” era of inefficiency and into a future where every eligible patient has the opportunity to participate in the research that could save their life.
Ready to accelerate your recruitment?
Learn how Lifebit’s federated platform can power your AI recruitment strategies.

