How AI Solutions are Transforming the Pharmaceutical Industry

How Pharmaceutical AI Solutions Cut R&D Timelines by 80%
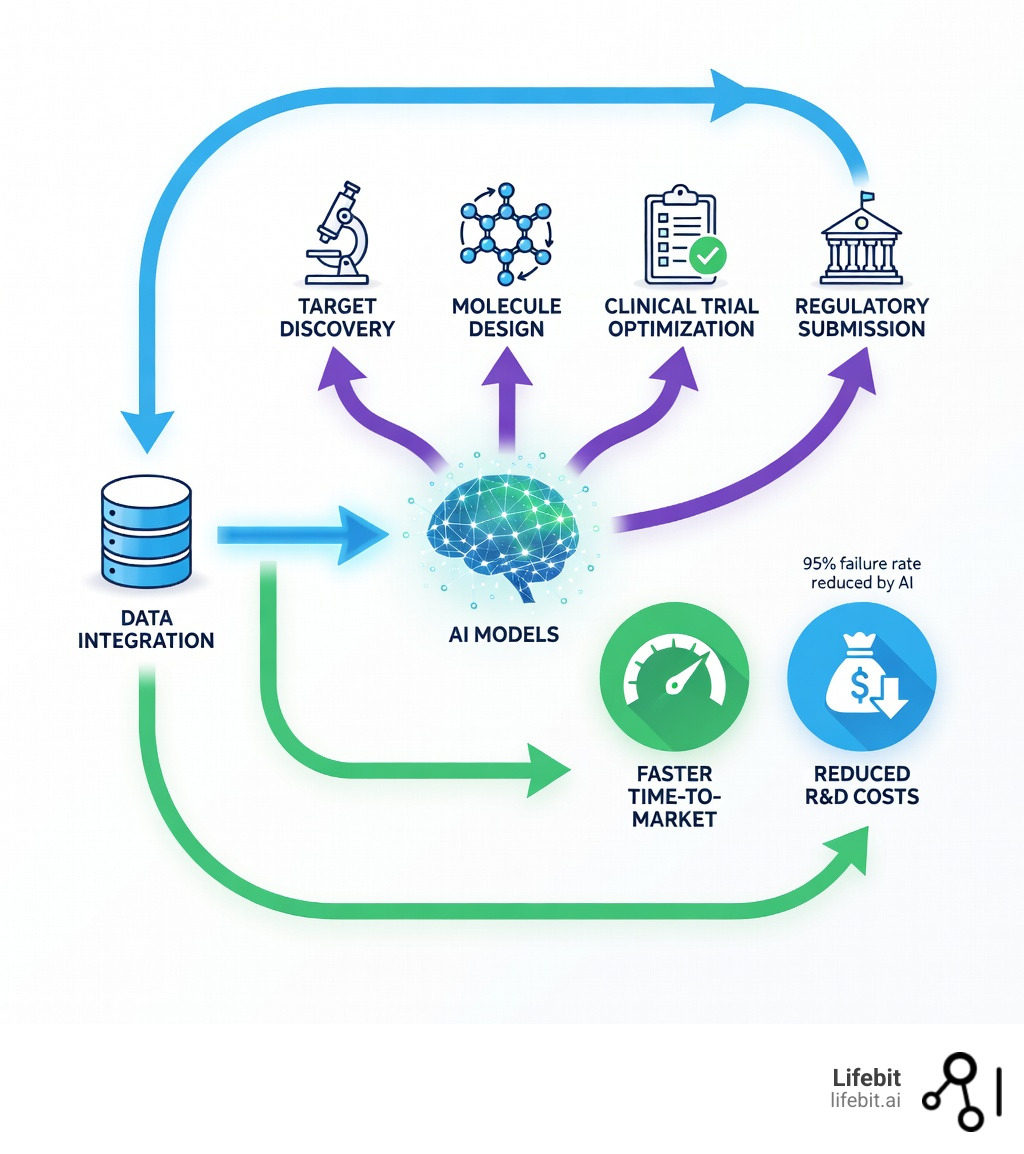
Pharmaceutical AI solutions are revolutionizing how the industry finds drugs, runs clinical trials, and delivers medicines to patients. Here’s what you need to know:
Key Applications:
- Drug Findy – AI reduces findy timelines from 5-6 years to just 1 year
- Clinical Trials – 70% cost savings and 80% timeline reductions per trial
- Supply Chain – Real-time forecasting, cold chain management, and counterfeit prevention
- Regulatory Compliance – FDA-aligned frameworks for AI-powered submissions
Market Impact:
- $350-410 billion in annual value projected by 2025
- 95% of pharmaceutical companies now investing in AI
- 42.68% CAGR growth between 2024-2029
- Only 5% of drugs reach market—AI aims to improve these odds
The pharmaceutical industry faces a brutal reality: developing a new drug takes 10-15 years, costs over $5 billion, and sees a 95% failure rate. Traditional methods can’t keep pace with the complexity of modern disease biology or the massive datasets now available. AI is changing that equation by analyzing billions of molecular compounds in weeks, predicting clinical outcomes before trials begin, and automating the manual work that slows everything down.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve built a federated biomedical data platform that enables secure, compliant analysis across siloed datasets—powering Pharmaceutical AI solutions for drug findy and clinical development. With over 15 years in computational biology, genomics, and AI, I’ve seen how these technologies can accelerate the path from target identification to patient impact.
Cut Drug Findy from 6 Years to 1 with AI
Traditional drug findy is like trying to find a specific grain of sand on a beach while wearing a blindfold. It typically takes five to six years just to identify a viable candidate. However, Pharmaceutical AI solutions are effectively taking that blindfold off.
By leveraging accelerated computing and deep learning, we are seeing the findy phase shrink to as little as one year. This isn’t just a marginal improvement; it’s a total reimagining of the R&D lifecycle. Today, 80% of pharmaceutical and life sciences professionals are already using AI for drug findy, driven by the need to find needles in the “multi-omic” haystack.
One of the most powerful shifts is the transition to a “lab-in-the-loop” approach. Instead of running thousands of physical experiments that might fail, companies like Genentech are using generative AI to propose designs for candidate molecules. These designs are then tested in the lab, and the results are fed back into the AI to refine the next round of predictions. It’s an iterative cycle that turns drug findy from a game of chance into a high-speed engineering discipline.
The Rise of Generative Chemistry and ADMET Profiling
Beyond simple identification, AI is now mastering ADMET profiling (Absorption, Distribution, Metabolism, Excretion, and Toxicity). Historically, many drugs failed late in the process because they were toxic to the liver or weren’t absorbed properly by the human gut. AI models, trained on decades of historical trial data and chemical properties, can now predict these failures before a single molecule is synthesized. By using Graph Neural Networks (GNNs), researchers can represent molecules as mathematical graphs, allowing the AI to understand the relationship between chemical structure and biological activity with unprecedented nuance.
We are seeing this play out across several critical areas:
- Protein Structure Prediction: Tools like OpenFold (available via NVIDIA BioNeMo) allow researchers to predict how proteins fold with incredible speed. Since misfolded proteins contribute to diseases like Alzheimer’s and Parkinson’s, understanding these structures is the first step toward a cure.
- Target Identification: AI models can sift through billions of data points—from genomics to transcriptomics—to identify the specific biological “targets” (like a protein or gene) that a drug should interact with.
- Small Molecule Generation: Using generative chemistry, AI can design entirely new molecules that have never existed in nature but are perfectly “shaped” to bind to a disease target.
For a deeper look at how this works, check out our ai-powered drug discovery ultimate guide.
Changing Biologics and Small Molecule Generation
The “old way” of finding molecules involved screening massive libraries of existing compounds. The “new way” is de novo design.
Using transformer models—the same architecture behind ChatGPT—scientists are now treating biology like a language. If DNA is the alphabet and proteins are the sentences, AI is the ultimate editor. This allows for Drug Discovery With Generative AI where models propose molecular designs that are optimized for both efficacy and “synthesizability” (the ability to actually make the drug in a lab). This is particularly critical for Antibody-Drug Conjugates (ADCs), a new class of highly potent cancer drugs that require precise engineering to ensure they only kill cancer cells while sparing healthy tissue.
| Phase | Traditional Timeline | AI-Driven Timeline | Efficiency Gain |
|---|---|---|---|
| Target Identification | 2-3 Years | Weeks/Months | ~90% faster |
| Lead Generation | 3 Years | 1 Year | ~65% faster |
| Candidate Nomination | 1-2 Years | 3-6 Months | ~70% faster |
Platforms like NVIDIA Clara and BioNeMo are providing the backbone for this, offering pre-trained models for ai-powered target identification and De Novo Molecule Generation Using AI Transformer Models. This is particularly exciting for biologics—complex drugs derived from living organisms—where AI can optimize antibodies to be more stable and less likely to be rejected by the human immune system.
Stop Wasting Billions: Cut Clinical Trial Timelines by 80%
If drug findy is the sprint, clinical trials are the marathon—and they are where 90% of candidates go to die. They are also astronomically expensive. However, Pharmaceutical AI solutions are delivering cost savings of 70% per trial and timeline reductions of 80%.
One of the biggest headaches in any trial is patient recruitment. Finding the right patients who meet strict criteria can take months or even years. AI-powered recruitment tools can now scan electronic health records (EHRs) and genomic data in seconds to identify ideal candidates. For a full breakdown, see our ai clinical trial recruitment complete guide.
Synthetic Control Arms and Virtual Trials
Perhaps the most disruptive application of AI in clinical development is the use of Synthetic Control Arms (SCAs). Traditionally, a trial requires a control group of patients who receive a placebo. In rare diseases or life-threatening conditions, recruiting this group is difficult and often ethically complex. AI can generate a synthetic control group using historical trial data and real-world evidence (RWE). This reduces the number of human participants needed, slashes costs, and accelerates the path to regulatory approval.
Furthermore, Natural Language Processing (NLP) is being used to optimize trial protocols. By analyzing thousands of previous trial failures, AI can flag “unrealistic” inclusion criteria or logistical bottlenecks in a draft protocol before the trial even begins. This proactive optimization prevents the mid-trial amendments that currently plague 60% of all clinical programs.
Consider the Clinical Study Report (CSR). Traditionally, medical writers spend an average of 180 hours (about 2-3 weeks) sifting through thousands of pages of data to create a first draft. Merck recently implemented a generative AI platform that slashed this time to just 80 hours—a 55% reduction. Even better, the AI reduced errors in data, citations, and terminology by 50%.
This is the power of ai for clinical trials: it doesn’t just make things faster; it makes them more accurate, reducing the risk of regulatory rejection.
Optimizing Pharmaceutical AI Solutions for Clinical Success
We are also entering the era of the “Digital Patient.” Instead of relying solely on physical trials, researchers are using generative ai and omop revolutionizing real world evidence to create virtual models of patients.
These digital twins allow for:
- Virtual Clinical Simulations: Testing a drug’s impact on a “virtual” population before the first human dose is ever given.
- Subgroup Analysis: Identifying which specific groups of patients (e.g., those with a specific genetic mutation) will respond best to a treatment.
- Dosing Optimization: Using AI to predict the perfect dose that maximizes healing while minimizing side effects. This is particularly vital in oncology, where the therapeutic window between “effective” and “toxic” is incredibly narrow.
By using Real-World Data (RWD) and multi-omics integration, we can de-risk trials before they even begin. This is a game-changer for precision medicine, ensuring that the right drug gets to the right patient at the right time.
How Asepha and AI Cut Pharmacy Labor Costs by 32% in 30 Days
While findy and trials get the headlines, Pharmaceutical AI solutions are also changing the “back office” of the industry—the pharmacies and supply chains that get medicine to the people who need it.
In the retail pharmacy space, manual labor is a massive drain on resources. Pharmacists spend hours on data entry, insurance triage, and patient follow-ups. Startups like Asepha are changing this with intelligent data entry and voice AI assistants. Within just one month of implementation, pharmacy operations have seen a 32% reduction in manual labor costs. Tasks that used to take all morning are now being completed 4x faster.
Smart Manufacturing and Industry 4.0
Beyond the pharmacy counter, AI is securing the global supply chain and revolutionizing manufacturing through Industry 4.0 principles. Pharmaceutical manufacturing has traditionally been a “batch” process, which is slow and prone to waste. AI-driven continuous manufacturing allows for real-time monitoring of chemical reactions. Sensors powered by AI can detect minute deviations in temperature or pressure, automatically adjusting the machinery to prevent a batch from being ruined. This “predictive maintenance” alone is estimated to save the industry billions in lost inventory.
Key supply chain impacts include:
- Inventory Forecasting: Predicting demand spikes (e.g., during flu season) to ensure life-saving drugs don’t go out of stock.
- Cold Chain Management: Using IoT sensors and AI to monitor temperature-sensitive biologics (like mRNA vaccines) in real-time, predicting potential spoilage before it happens based on transit delays.
- Counterfeit Prevention: Automated quality assurance systems can spot counterfeit packaging or pills that human inspectors might miss, using computer vision to verify holographic seals and microscopic print patterns.
- Personalized Medicine Logistics: For therapies like CAR-T (where a patient’s own cells are modified), AI manages the complex “vein-to-vein” logistics, ensuring the right cells return to the right patient across a global network.
These efficiencies are critical for ai for pharmacovigilance complete guide, helping companies monitor the safety of their drugs long after they hit the market.
Stop FDA Rejections: Why Transparent AI is Mandatory
You can’t just “move fast and break things” in the pharmaceutical industry. Patient safety is paramount. This is why regulatory bodies like the FDA are leaning heavily into AI.
The FDA’s Center for Drug Evaluation and Research (CDER) established an AI Council in 2024 to oversee the surge in AI-related drug submissions. Between 2016 and 2023, the FDA reviewed over 500 drug applications that included AI components. They are looking for more than just “cool tech”; they want transparency, traceability, and rigorous oversight.
The Challenge of Explainable AI (XAI)
One of the biggest hurdles in regulatory approval is the “Black Box” problem. If an AI identifies a new drug target, regulators need to know why. This has led to the rise of Explainable AI (XAI). Techniques like SHAP (SHapley Additive exPlanations) are now being used to break down which specific biological markers led an AI to its conclusion. Without this level of interpretability, AI-driven insights are often dismissed by clinical reviewers who require a mechanistic understanding of disease biology.
Key regulatory focus areas include:
- Data Bias: Ensuring AI models aren’t trained on limited datasets that exclude certain ethnicities or genders, which could lead to drugs that are less effective for minority populations.
- Explainability: Moving away from “black box” models. Regulators need to know the logic behind the prediction.
- Data Integrity: Adhering to ALCOA principles (Attributable, Legible, Contemporaneous, Original, and Accurate) to ensure that AI-generated data hasn’t been tampered with.
For companies operating in the US, UK, Europe, and Singapore, staying compliant means following the latest Artificial Intelligence for Drug Development | FDA guidances and understanding ai in drug discovery a review of us eu policy guidance.
Ensuring Security in Pharmaceutical AI Solutions
At Lifebit, we believe that the biggest hurdle to AI adoption isn’t the math—it’s the data access. Biomedical data is sensitive, siloed, and highly regulated. You can’t just move 500 terabytes of genomic data to the cloud whenever you want to run an analysis. This is where Federated Learning becomes essential.
Federated Learning allows models to be trained across multiple decentralized servers (like different hospitals or research centers) without the raw data ever leaving its original location. This preserves patient privacy while allowing the AI to learn from a much larger and more diverse dataset than any single institution could provide. It solves the “data silo” problem that has historically slowed down multi-omic research.
This allows for:
- Secure Collaboration: Multiple organizations can “train” an AI model on their combined datasets without ever actually sharing the raw patient data with each other.
- Multi-Omics Integration: Combining genomics, proteomics, and clinical data within a Trusted Research Environment (TRE).
- FAIR Data Principles: Making sure data is Findable, Accessible, Interoperable, and Reusable.
By using a federated approach, we help biopharma companies and governments maintain HIPAA and SOC 2 compliance while still open uping the insights hidden in their ai for genomics complete guide.
Frequently Asked Questions about AI in Pharma
How much time does AI save in drug findy?
AI can shorten the drug findy process from the traditional 5-6 years to just one year. By automating target identification and lead optimization, it can reduce the overall time to develop a drug by up to four years, potentially saving $26 billion in R&D costs globally.
Is AI compliant with FDA regulations?
Yes, but it must be implemented within a validated quality system. The FDA and EMA have released guiding principles for “Good Machine Learning Practice.” Regulators expect AI models to be transparent, well-documented, and subject to rigorous human oversight (the “human-in-the-loop” model).
What are the biggest challenges of AI implementation?
The “Big Three” challenges are data quality, regulatory compliance, and cultural resistance. AI is only as good as the data it’s trained on; if the data is disjointed or biased, the results will be too. Additionally, integrating AI into legacy pharmaceutical workflows requires a significant shift in how teams collaborate.
The $410B Future of Pharmaceutical AI Solutions
The trajectory is clear: the pharmaceutical AI market is projected to grow at a staggering 42.68% CAGR, adding $15 billion in value by 2029. But the real value isn’t just in the dollars—it’s in the lives saved.
We are moving toward a future where “untreatable” diseases are mapped in weeks, where clinical trials are safer and more inclusive, and where the 95% failure rate in drug development becomes a relic of the past. At Lifebit, our mission is to provide the federated infrastructure that makes this possible, ensuring that the world’s most sensitive data can power the world’s most important findies.
Ready to see how federated AI can accelerate your R&D? Explore the Lifebit Federated Biomedical Data Platform and join the revolution in Pharmaceutical AI solutions.

