The Ultimate Guide to AI-Driven Drug Discovery

Why Drug Findy Desperately Needs a Revolution
Here’s the uncomfortable truth: traditional drug findy is broken. It takes 13-15 years and over $2.5 billion to bring a single new medicine to market, and approximately 90% of drug candidates fail in trials. These aren’t just statistics; they represent patients waiting for cures that never arrive.
The problem is scale. With over 10^60 potential drug compounds to explore, traditional trial-and-error methods are too slow and expensive.
AI-driven drug findy is the solution. By using machine learning and generative AI, we can accelerate target identification, optimize drug candidates, and predict clinical outcomes—cutting development time from over a decade to just a few years.
Key benefits of AI-driven drug findy:
- Speed: Reduces preclinical research from years to months.
- Cost: Dramatically lowers R&D expenses by predicting failures earlier.
- Success Rate: Improves the dismal 10% Phase I approval rate.
- Scale: Explores a vast chemical space impossible for humans to steer.
- Precision: Predicts drug properties like toxicity and efficacy before lab work.
This isn’t theoretical. AI-designed drugs are already in clinical trials. But success isn’t just about algorithms; it’s about data. AI needs massive, diverse datasets—from genomics to electronic health records—to work. It’s about integrating wet-lab biology with dry-lab computation and turning predictions into real medicines.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit. With a background in computational biology and AI, I’ve spent over a decade building federated platforms that provide secure access to the biomedical data that powers this revolution. This guide will show you how AI is changing every stage of the drug development pipeline.
AI-driven drug findy terms to learn:
The Core Components: How AI is Rewriting the Rules of R&D
AI-driven drug findy works by combining powerful computational techniques with vast biological data. This fusion allows researchers to cut years off development timelines and explore chemical possibilities that were previously out of reach.
Let’s break down the core technologies:
- Machine Learning (ML): The workhorse algorithm that learns from data to make predictions. In drug discovery, this can range from predicting whether a compound will be toxic to forecasting how a specific patient might respond to treatment based on their genetic makeup.
- Deep Learning (DL): A subset of ML that uses complex, multi-layered neural networks to spot intricate patterns in massive datasets. This is the technology behind breakthroughs like AlphaFold’s protein structure prediction, which solved a 50-year-old grand challenge in biology.
- Generative AI: Goes beyond prediction to create entirely new biological entities. These models can design novel molecules and protein sequences from scratch, each optimized for specific properties like high efficacy, low toxicity, and good manufacturability.
- Natural Language Processing (NLP): Gives AI the ability to read, interpret, and synthesize information from millions of research papers, patents, and clinical trial notes. This helps researchers uncover hidden connections between genes, proteins, and diseases that would be impossible for humans to find manually.
- Foundation models: These are large, pre-trained models built on vast biological datasets (e.g., all known protein sequences or chemical compounds). They develop a fundamental “understanding” of biology or chemistry that can then be fine-tuned for highly specific drug discovery tasks, such as predicting protein-protein interactions or designing antibodies.
However, none of these technologies matter without high-quality, multimodal data. This includes genomics, proteomics, transcriptomics, and clinical data from real patients. For example, genomic data from sources like the UK Biobank can reveal genetic variants associated with disease risk, pointing to potential drug targets. Proteomic data provides information on protein expression and post-translational modifications, offering a more dynamic view of cellular processes. Transcriptomic data (RNA-seq) measures gene expression levels, helping researchers understand how diseases alter cellular function and how drugs might reverse those changes. Without this rich, multi-layered information, even the most sophisticated algorithm is just expensive code.
Understanding the AI Toolkit
Matching the right technique to the right problem is key.
- Supervised learning learns from labeled examples. For instance, a model trained on thousands of compounds with known binding affinities to a target protein can predict the affinity of a new, unseen molecule, saving valuable lab time and resources.
- Unsupervised learning finds hidden patterns in unlabeled data. This is extremely useful for tasks like discovering new disease subtypes from patient clinical data or grouping molecules by their mechanism of action without prior knowledge.
- Reinforcement learning learns by trial and error, much like a game-playing AI. It can iteratively refine molecular structures, making small changes and receiving a “reward” if the change improves a desired property like target binding or solubility.
- Graph Neural Networks (GNNs) are ideal for molecular data, as they process molecules as graphs (atoms as nodes, bonds as edges). They are particularly powerful because they capture the 3D structure and chemical relationships within a molecule, which are crucial for its biological activity. This is a significant leap from older methods that represented molecules as simple text strings (SMILES).
- Transformers, originally from language processing, excel at handling the sequential data found in proteins (amino acid sequences) and genes (DNA sequences). Initially developed for translating languages, they are now being adapted to understand the ‘language’ of biology. Models like ESM-2 can predict protein structures from their amino acid sequences with incredible accuracy, a task that once took years of dedicated lab work.
Together, these tools allow us to predict molecular properties with remarkable accuracy before synthesizing a single molecule.
The Data Foundation for AI Success
An AI model is only as good as its data. In drug findy, we work with structured data (like genomic sequences) and unstructured data (like scientific papers). The challenge is integrating these sources.
Public databases like ChEMBL are valuable but suffer from inconsistencies, varying standards, and “batch effects” that can mislead AI models. Another problem is publication bias—AI needs to learn from failures, but negative results are rarely published.
This is why data harmonization and standardization are critical. However, the biggest challenge is that most valuable data is locked in institutional silos due to privacy regulations and proprietary concerns. Centralizing this data is a security and compliance nightmare.
The solution is federated data access. Instead of moving data, you bring the AI to the data. Models learn from information across multiple institutions without sensitive data ever leaving its source. This respects privacy while open uping the collective power of global biomedical information.
At Lifebit, our federated AI platform was built to solve this problem. Our Trusted Research Environment enables secure, compliant research across distributed data sources, allowing researchers in London, New York, and beyond to collaborate without compromising data security. The future of medicine depends on open uping data—safely, securely, and at scale.
AI in Action: Revolutionizing the Drug Findy Pipeline
AI-driven drug findy is actively changing every stage of the development pipeline. We’re now exploring the vast 10^60 chemical space, accelerating preclinical research from years to months, and improving the industry’s historically low success rates.
Stage 1: Identifying and Validating Novel Targets
Choosing the right biological target is the most critical first step, as a mistake here dooms the entire project. AI algorithms analyze vast, multimodal datasets—genomics, proteomics, transcriptomics, and clinical records—to link genes and proteins to disease mechanisms. By spotting subtle patterns buried in millions of data points, machine learning helps prioritize the most promising targets, dramatically reducing early-stage failures. For example, AI can identify neoantigens for personalized cancer vaccines or mine scientific literature to uncover hidden connections. A prominent real-world example is BenevolentAI, which used its AI platform to analyze biomedical literature and patient data to identify baricitinib, an existing rheumatoid arthritis drug, as a potential treatment for COVID-19. This hypothesis was rapidly validated in clinical trials, showcasing AI’s power to connect disparate information. This ensures that research efforts are built on a solid, data-driven foundation. Scientific research on AI for target discovery shows how this approach is fundamentally changing success rates.
Stage 2: Designing Better Molecules with AI-driven drug findy
Once a target is validated, AI moves from analysis to creation. Instead of screening existing compounds, de novo drug design powered by generative AI creates entirely novel molecules from scratch. These models can optimize for multiple factors simultaneously: efficacy, stability, manufacturability, and safety.
A key capability is predicting ADMET properties (Absorption, Distribution, Metabolism, Excretion, and Toxicity) early in the process. This includes predicting properties like oral bioavailability (how much of the drug is absorbed), blood-brain barrier permeability (whether it can reach the brain), metabolic stability (how quickly it’s broken down), and potential for liver toxicity (hepatotoxicity). By flagging molecules with poor ADMET profiles computationally, AI prevents resources from being wasted on candidates destined to fail in later, more expensive stages. AI is now designing everything from small molecules to complex biologics like therapeutic proteins and gene therapies. A deep dive into automated chemical design showcases the sophisticated algorithms making this possible.
Stage 3: Accelerating Clinical Trials and Drug Repositioning
Clinical trials are the longest, most expensive, and riskiest phase of development. AI is changing this calculus by making trials smarter and more efficient.
Patient stratification uses AI to analyze patient data (genomics, medical histories) to identify subgroups most likely to respond to a drug. This leads to more targeted trials with higher success rates. AI can also predict clinical trial outcomes, helping researchers “fail faster” on unpromising candidates and reallocate resources.
Furthermore, AI is a game-changer for drug repositioning—finding new uses for already-approved medicines. By analyzing drug data, molecular structures, and disease profiles, AI uncovers unexpected connections, offering a faster, lower-risk path to treatment. This is particularly valuable for rare diseases, where the high cost of de novo discovery is a major barrier. The baricitinib example for COVID-19 is a perfect illustration of successful AI-driven repositioning.
Key AI applications changing clinical trials:
- Predicting drug efficacy and safety profiles before human trials.
- Identifying optimal patient populations to improve success rates.
- Real-time monitoring of patient responses and adverse events.
- Accelerating drug repositioning for new indications.
- Generating synthetic control arms (SCAs) to reduce trial size. An SCA uses AI to model the progression of a disease in a group of patients based on historical clinical trial data and real-world evidence. This can be used in place of a traditional placebo group, especially in rare diseases where recruiting enough patients for a control arm is difficult or unethical. This not only accelerates trials but also allows every enrolled patient to receive a potential treatment.
- Improving data management across complex, multi-site trials.
AI isn’t eliminating human trials, but it’s making them faster, cheaper, and far more likely to succeed.
The Problems Ahead: Challenges and Solutions in AI-Powered Pharma
The potential of AI-driven drug findy is immense, but the path forward has challenges. We’re dealing with complex data, regulatory problems, and the need for new ways of working.
The Data Dilemma: Quality, Accessibility, and Privacy
AI is only as good as its data. In pharma research, data quality is a major issue. Variations in lab protocols create “batch effects” that can mislead AI models. Publication bias is another problem; AI needs to learn from failures, but negative results are often unpublished, creating a skewed view of reality.
Even with perfect data, privacy and security are paramount. Patient data is rightly protected by regulations in Europe, the UK, USA, and beyond, but this can create barriers to the large-scale data sharing AI needs. Furthermore, valuable data is often locked in institutional silos.
At Lifebit, we solve this with a federated approach. Our platform allows AI models to analyze data where it resides, without moving sensitive information. This protects patient privacy while giving researchers secure access to diverse, global datasets. We also focus on data harmonization, as clean, consistent data is essential for reliable AI.
Bridging the Gap: Integrating Biology, Chemistry, and Code
AI isn’t replacing scientists; it’s amplifying them. The real breakthroughs happen when computational predictions meet experimental validation in a process called the ‘lab-in-the-loop’ concept.

In this cycle, an AI model suggests a molecule, chemists synthesize it, and biologists test it. The results—good or bad—are fed back into the model, which learns and improves. This iterative process requires close collaboration between biologists, chemists, and data scientists. The models don’t need to be perfect initially; they just need to generate testable hypotheses that can be refined through rapid experimentation.
Navigating Ethical, Regulatory, and IP Challenges in AI-driven drug findy
As AI becomes central to drug development, new ethical and legal questions arise.
- Algorithmic bias is a serious concern. If AI models are trained on data that isn’t diverse, the resulting drugs may not work for everyone, worsening health disparities. For example, if a model is trained primarily on genomic data from European populations, it may develop drugs that are less effective or have different side effects in individuals of African or Asian descent. Addressing this requires a concerted global effort to build more diverse, representative, and equitably sourced datasets.
- The “black box” problem, where complex AI models can’t explain their predictions, creates regulatory hurdles. Agencies like the FDA and EMA need transparency to approve new drugs. To address this, the field of Explainable AI (XAI) is developing methods like SHAP (SHapley Additive exPlanations) and LIME (Local Interpretable Model-agnostic Explanations). These techniques help researchers understand why a model made a certain prediction (e.g., which part of a molecule is responsible for its predicted toxicity), making the results more trustworthy, auditable, and actionable for chemists and biologists.
- Intellectual property law is struggling to keep pace. Current patent laws were designed for human inventors. When an AI system independently conceives of a novel and useful molecule, it raises complex questions. The DABUS case, where an AI was named as the inventor on patent applications, has been tested in courts worldwide with varying outcomes. The industry and patent offices are now grappling with how to adapt IP frameworks to recognize and protect AI-generated inventions, which is crucial for incentivizing the massive investment required for this technology.
Regulators are adapting. The FDA Modernization Act 2.0 now allows alternatives to animal testing, opening the door for AI to analyze data from more advanced, human-relevant models like organoids. The solution lies in transparency, rigorous validation, and strong ethical frameworks from the start.
The Future is Now: AI’s Impact on Health and the Pharma Industry
The impact of AI-driven drug findy is not a distant promise; it’s reshaping medicine today. We are moving toward a future where medicine is personalized, proactive, and accessible to patients left behind by traditional pharma economics.
Democratizing Findy for Unmet Needs
Roughly 7,000 rare diseases affect over 300 million people, yet 95% have no approved treatment. Traditional drug findy economics fail when the patient population is small. AI changes that calculus completely.
By slashing R&D costs and accelerating timelines, AI makes it viable to develop treatments for rare diseases. The same logic applies to neglected areas like antibiotics, where market incentives are broken, and women’s health, which is chronically underfunded. AI doesn’t just make drug findy faster; it makes it possible to pursue innovation where it’s needed most. This democratization of research fosters global health equity, enabling innovation in underserved regions worldwide.
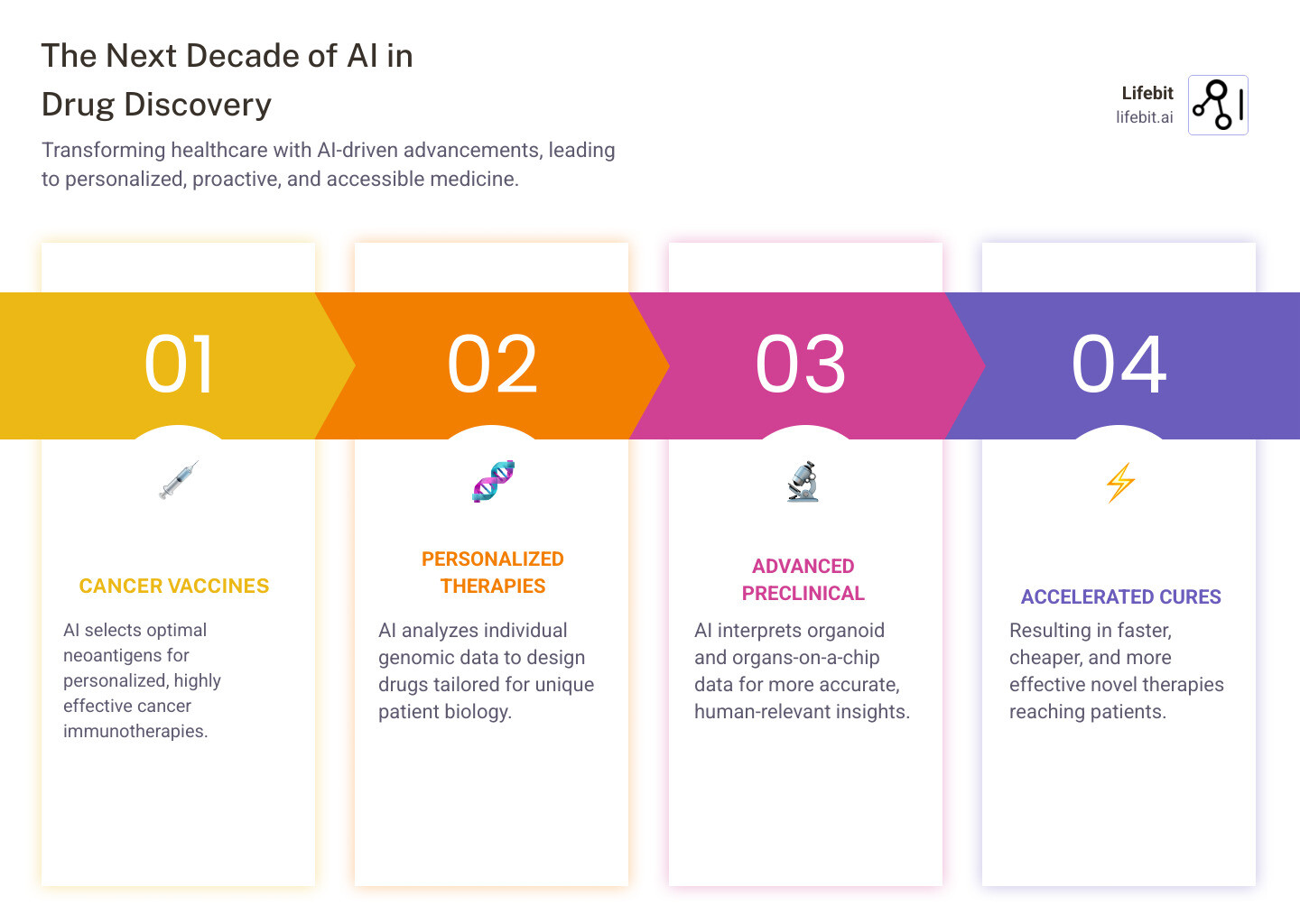
The Next Decade: From AI Models to Patient Cures
The next decade will see even more dramatic advances.
AI is already helping design personalized cancer vaccines that are now in clinical trials. Soon, AI-designed immunotherapies customized to individual tumors will become standard care. This is the beginning of true patient-specific therapies, where drugs are matched to an individual’s unique biology for maximum efficacy and minimal side effects.
This future is enabled by integrating AI with advanced preclinical models like organoids and organs-on-a-chip. As sanctioned by the FDA Modernization Act 2.0, these human-relevant systems provide far more accurate data for AI analysis than animal testing. Meanwhile, protein language models are accelerating the design of biologics, which already represent a large portion of new FDA approvals.
The ultimate impact is simple: faster, cheaper, and more effective medicines for patients. We will see more novel treatments in the next ten years than in the last fifty.
At Lifebit, we’re building the federated AI platform that makes this future possible, enabling researchers across London, New York, and the globe to securely analyze the diverse biomedical data that powers these breakthroughs. This is a fundamental shift in what’s possible for human health.
Frequently Asked Questions about AI-Driven Drug Findy
What is the main advantage of using AI in drug findy?
The biggest advantages are speed, cost, and success rate. AI compresses research timelines from years to months. It cuts costs by helping researchers fail early and cheaply, predicting toxicity computationally before millions are spent on clinical trials. Finally, by identifying more promising drug candidates from the start, AI dramatically improves the odds of developing a drug that works.
Can AI replace human scientists in drug findy?
No. AI is a powerful tool that augments human expertise, but it cannot replace scientific creativity, intuition, and strategic decision-making. The future of AI-driven drug findy is a partnership where AI generates hypotheses at a massive scale, and human scientists design experiments, validate predictions, and provide critical biological understanding. This “lab-in-the-loop” approach combines the best of machine intelligence and human intellect.
Is AI-driven drug findy a reality today?
Yes, absolutely. This isn’t science fiction. Multiple AI-designed drugs are currently in human clinical trials. Nearly every major pharmaceutical company is integrating AI into its R&D pipeline to stay competitive. Regulatory bodies like the FDA are adapting their frameworks for AI-generated therapies. The question is no longer if AI will transform drug findy, but how quickly organizations can adopt it.
Conclusion: A New Era for Medicine
We stand at an inflection point in human health. AI-driven drug findy is fundamentally reimagining how we develop medicines, compressing timelines, slashing costs, and giving patients access to treatments that were previously impossible. We are seeing hope for rare diseases, new paths for antibiotics, and the dawn of personalized cancer vaccines.
But this revolution is only as powerful as the data it can access. Success requires secure, scalable platforms that can open up the power of global biomedical data while respecting patient privacy. It demands infrastructure that breaks down data silos without compromising security.
At Lifebit, our federated AI platform was built for this purpose. Our Trusted Research Environment (TRE) and other solutions enable biopharma companies and public health agencies to conduct compliant, real-time analysis across distributed datasets. We provide the foundation for collaboration at the scale and speed modern drug findy demands.
The future of medicine will be written by human intuition and machine intelligence working in harmony. The question isn’t whether AI will transform drug findy. It already has. The question is: will you be part of the revolution?
Find how to power your research with federated AI and join us in building a healthier future for everyone.

