Revolutionize Your Lab with AI-Powered Drug Discovery Platforms

Why AI Drug Findy Software Is No Longer Optional
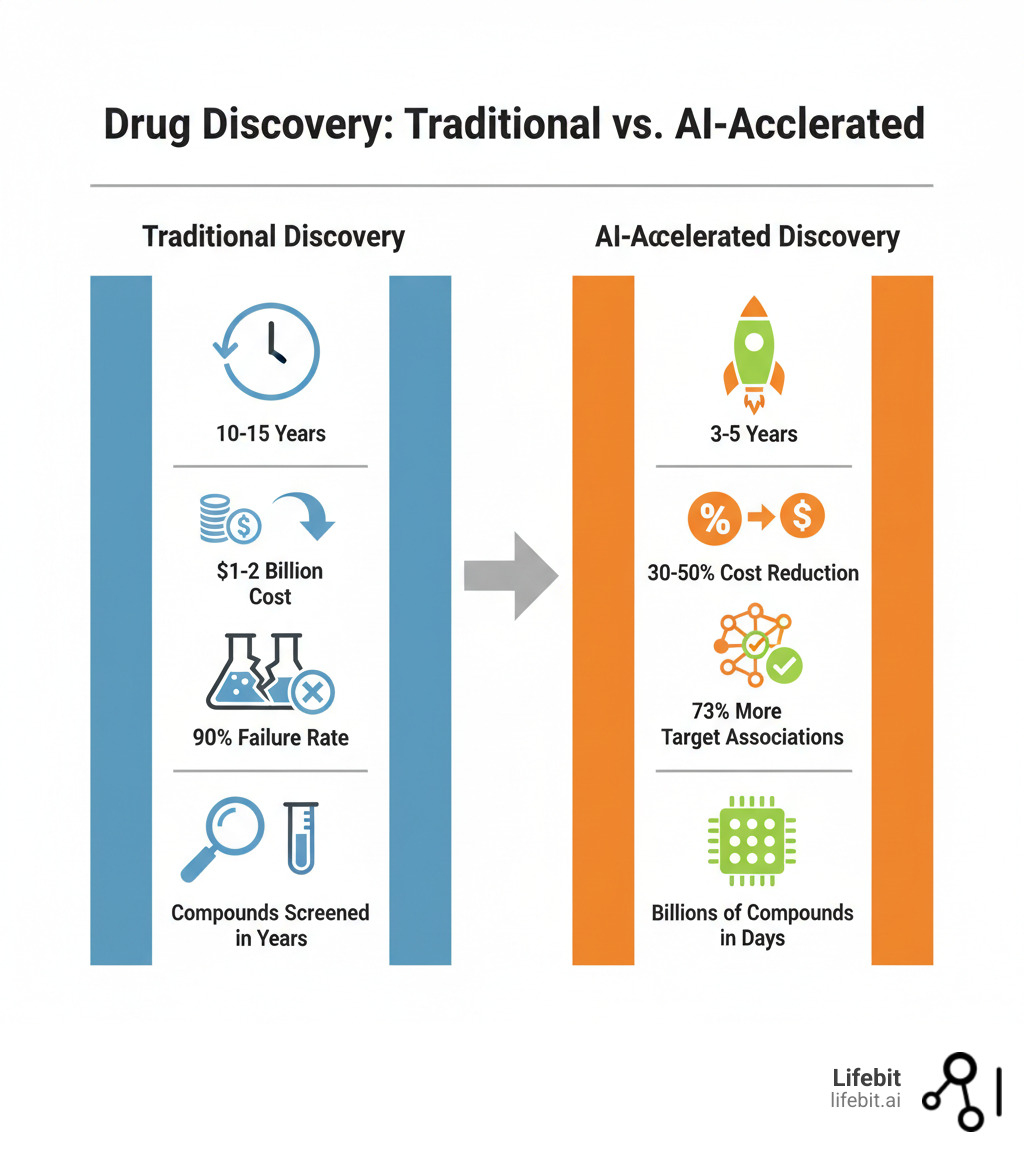
AI drug findy software is redefining how pharma identifies targets, designs molecules, and predicts clinical success. By pairing machine learning and generative AI with vast chemical and biomedical datasets, these platforms compress the traditional 10–15 year timeline and chip away at the $1–2 billion cost per approved drug. From multi-omics-driven target findy to generative de novo design, ultra-fast virtual screening, and early toxicity prediction, AI moves critical decisions upstream.
The contrast is stark. Traditional drug findy suffers a 90% clinical failure rate, much of it predictable earlier. Today, AI can scan billions of virtual molecules in minutes, and advanced deep learning models uncover dramatically more gene–phenotype associations than standard methods. The industry is rapidly shifting toward integrated, automated “drug findy and design” pipelines that blend generative AI with robotics—an evolution already visible as fully AI-finded drugs advance into mid-stage trials.
As Dr. Maria Chatzou Dunford, CEO and Co-founder of Lifebit, I’ve spent over 15 years building platforms that connect AI drug findy software to federated biomedical data, enabling pharmaceutical organizations to open up insights from genomics, clinical trials, and multi-omics datasets without moving sensitive data. In our experience, the true bottleneck isn’t AI capability—it’s scalable access to high-quality, harmonized data.
Basic AI drug findy software glossary:
- AI-driven drug development
- artificial intelligence (AI) enabled data repository services and informatics tools and capabilities
How AI Transforms Drug Findy from a Gamble to a Science
Let’s be honest—traditional drug findy has always been a bit of a crapshoot. You’re searching for a needle in a haystack the size of a small universe, and you’re doing it blindfolded. The numbers are brutal: over a decade of work, $1-2 billion per approved drug, and a 90% failure rate in clinical trials. That’s nine out of ten candidates crashing and burning after years of effort and investment.
Why is it so hard? The chemical universe contains an estimated 10⁶⁰ possible drug-like molecules. Exploring even a tiny fraction of this space through traditional trial-and-error lab work is like trying to taste-test every grain of sand on Earth. AI drug findy software is changing this fundamental equation, turning what was essentially an educated gamble into a predictive science.
AI tackles the core challenges by bringing speed, precision, and predictive power to every stage of the drug findy pipeline. Instead of relying on luck or brute-force screening, we can now make smarter, data-driven decisions from day one. This means fewer dead ends, less wasted time, and significantly better odds of success.
Accelerating Target Identification with Precision
Before you can design a drug, you need to know what you’re aiming at. Target identification—figuring out which protein, gene, or biological pathway to target—is the crucial first step. Traditionally, this meant researchers spending months or years combing through biological pathways and disease mechanisms.
AI drug findy software changes this game entirely by analyzing massive amounts of multi-omics data—genomics (your DNA), proteomics (your proteins), and metabolomics (the small molecules in your cells). By feeding an AI platform mountains of patient genetic sequences, protein expression profiles, and metabolic data, it can spot subtle patterns and correlations that would take human researchers years to uncover.
The results speak for themselves. For example, certain deep neural networks can identify 73% more gene-phenotype associations for complex human diseases compared to standard methods. That’s a massive leap forward in our ability to find promising targets quickly. Scientific research on AI for target discovery is advancing rapidly, giving us clearer views into disease mechanisms and validating disease pathways that are ripe for drug intervention. These platforms help us spot novel targets that we might have overlooked entirely.
Designing Novel Molecules with Generative AI
Once you’ve got a target, the next challenge is designing a molecule that can interact with it effectively. This is where generative AI becomes almost magical. Instead of synthesizing and testing thousands of compounds one by one, generative models can design entirely new molecules from scratch.
These algorithms work similarly to AI that generates realistic images or text, but they’re creating chemical structures. They can explore vast chemical spaces to create novel compounds through de novo design, optimizing for exactly what you need: potency (how strongly the drug binds to its target), selectivity (how well it avoids hitting the wrong targets), and even synthesizability (how easy it is to make the molecule in a lab).
Modern platforms leverage generative AI to explore billions of virtual compounds, generating fresh ideas for chemical structures that might never have occurred to a human chemist. These systems can reduce off-target effects by fine-tuning molecular properties and provide synthesizability scores so you know whether your design can actually be manufactured. The integration of AI with robotics is taking this even further, automating the entire journey from abstract design to tangible compound. A Guide to Generative AI for Medicinal Chemists offers insights into how this technology is becoming an essential part of the modern drug findy toolkit.
Predicting Success and Failure Before the First Experiment
One of the most frustrating realities of traditional drug development is watching a promising compound fail in clinical trials due to unforeseen toxicity or poor absorption. This is where AI offers perhaps its most valuable contribution: predicting success and failure before you even step into the wet lab.
Modern AI drug findy software can accurately forecast a molecule’s ADMET properties—that’s Absorption, Distribution, Metabolism, Excretion, and Toxicity. These predictions let us weed out problematic candidates early, when it’s still cheap and easy to pivot.
By simulating how a molecule will behave in a biological system, AI can assess its physicochemical properties, conduct toxicity screening, and predict bioavailability. AI platforms can predict late-stage outcomes from early-stage data, helping you avoid unnecessary and expensive measurements. These tools provide comprehensive ADMET modules covering everything from intestinal permeability to blood-brain barrier penetration and potential carcinogenicity.
This predictive power is a game-changer for reducing late-stage failures. Instead of finding deal-breaking problems after you’ve already invested years and millions, you can identify them on day one and either fix them or move on to better candidates. We’re talking about fundamentally better odds and smarter use of resources.
The Engine Room: A Breakdown of AI Technologies in Drug Findy
If you’ve ever wondered what’s actually happening inside AI drug findy software, you’re not alone. The terminology can feel overwhelming—machine learning, deep learning, neural networks, generative AI. But you don’t need a PhD to understand the basics. Think of these technologies as different tools in a sophisticated toolbox, each designed for specific tasks in the drug findy process.
Let’s break down the key AI technologies that are changing how we find new medicines and how they work together to accelerate findy.
Machine and Deep Learning: Finding Patterns in a Sea of Data
At the heart of most AI drug findy software sits machine learning (ML) and its more sophisticated relative, deep learning (DL). These technologies excel at one critical task: finding patterns in massive amounts of data that would take humans years to analyze.
Machine learning algorithms learn from existing data to make predictions. In drug findy, this often means using QSAR (Quantitative Structure-Activity Relationship) models. These models study the relationship between a molecule’s chemical structure and its biological activity to guide chemists toward better candidates. Even open-source platforms offer functionalities for developing these models, making the capability more accessible.
Deep learning takes this a step further. Using neural networks with multiple layers—hence “deep”—these algorithms can learn incredibly complex patterns. They’re particularly powerful for analyzing images from high-content screening and understanding intricate three-dimensional molecular structures. Some platforms combine advanced quantum chemical methods with machine learning for molecular design, extracting additional value from compound data.
Supervised learning works when we have labeled data—we know which compounds worked and which didn’t. Unsupervised learning finds hidden patterns in unlabeled data, potentially revealing novel relationships we didn’t even know to look for. Together, ML and DL provide the predictive analytics backbone that helps us screen vast chemical libraries and predict properties.
Generative AI: Creating What Has Never Existed
While machine learning is brilliant at analyzing what already exists, generative AI does something almost magical: it creates entirely new molecules and proteins.
Generative models learn the underlying rules of chemistry and biology, then use this understanding to design novel structures from scratch. This is de novo design at its finest, and it’s opening up chemical space that was previously inaccessible. A Guide to Generative AI for Medicinal Chemists walks through how these tools streamline workflows from hit identification to lead optimization.
Generative AI software leverages de novo molecular design, allowing researchers to explore billions of virtual compounds and generate new ideas for chemical structures. This isn’t just tweaking existing molecules—it’s inventing entirely new ones optimized for potency, selectivity, and synthesizability.
The applications extend beyond small molecules. Generative AI is now used to design improved proteins, with models trained on billions of protein sequences. A major breakthrough in this area is the accurate prediction of 3D protein structures, which is revolutionizing our understanding of protein function. The field is pushing even further with “generative biology,” creating entirely new therapeutic proteins that don’t exist in nature.
Reinforcement learning plays a crucial role here too. These algorithms learn through trial and error, getting “rewards” when they generate molecules with desired properties. It’s like having a tireless chemist who can explore millions of design options, learning what works and what doesn’t, all in silico before a single compound is synthesized.
A Buyer’s Guide to AI Drug Findy Software
Choosing the right AI drug findy software is like finding the perfect research partner—one who understands your needs, keeps your secrets, and helps you move faster. With the market expanding rapidly and full-scale “drug findy and design” pipelines accelerating, this decision has never been more critical.

The landscape is crowded with platforms promising to transform your research. The best choices focus on three areas: the specific functionalities you need, organizational fit, and data security/IP protection.
Key Functionalities of Modern AI Drug Findy Software
The most capable platforms act as comprehensive workbenches with integrated workflows that reduce friction and error. Expect seamless support for both ligand-based and structure-based design, plus robust molecular docking for binding pose and affinity predictions with clear visualization.
At scale, virtual screening is pivotal—top platforms can evaluate vast libraries in minutes. To bridge digital designs to the bench, retrosynthesis planning proposes practical synthetic routes. Rounding out the toolkit, strong cheminformatics/data analysis and AI-guided lead optimization help teams iterate quickly and intelligently.
Evaluating Different Types of AI Drug Findy Software
Start with your research priorities: novel target findy, de novo design, or lead optimization. Some tools specialize; others span end-to-end workflows. Ensure scalability via cloud-native computing and smooth enterprise integration.
Choose a user-friendly interface your chemists and biologists can adopt quickly, and verify custom model training on your proprietary data. Decide between SaaS vs. on-premise to balance accessibility and control. Review licensing options—annual subscriptions, pay-per-use, or open-source hybrids—based on project scope and internal expertise.
The Critical Role of Data Security and IP Protection
In drug findy, your data is existential. The “garbage in, garbage out” principle applies relentlessly: clean, harmonized, and relevant datasets drive valid predictions. Recent industry analysis underscores that robust data quality and validation are central to regulatory success .
Because research data lives across departments and legacy systems, platforms enabling data harmonization and federated access are essential—letting you analyze where the data resides without moving sensitive information. Look for recognized security certifications (for example, ISO 27001) and clear IP terms defining ownership of data and AI-generated outputs. Hybrid architectures can deliver public-cloud scale while keeping private data behind your firewall.
At Lifebit, our federated AI platform is built around these principles. Data remains at its source, protected by your controls, while our platform enables secure, real-time access for analysis—so your research, findies, and competitive edge stay protected.
The Bottom Line: Measuring the ROI of AI in Your Lab
The question isn’t whether AI works—it’s what it’s worth. For pharma, the value of AI drug findy software shows up where it matters: faster timelines, lower costs, and higher success rates.

Cost savings arrive first. By predicting failures early (toxicity, poor bioavailability, off-target effects), AI avoids expensive dead ends. In practice, teams have cut months of work by flagging inactive compounds upfront and reducing assay burden by well over half—while prioritizing molecules that are both potent and synthesizable.
Time efficiency compounds value. Entire pipelines accelerate—from target identification to virtual screening and AI-guided design. The first fully AI-finded and AI-designed drug reaching Phase 2 shows how fast the loop from in silico to in vitro can move when AI orchestrates decisions.
Success rates improve. AI highlights risks early and proposes better candidates, attacking root causes of late-stage failure. Large-scale studies have shown that AI can surface structurally novel hits across diverse targets, expanding chemical space beyond traditional screening.
Beyond metrics, AI lifts R&D productivity by automating repetitive work so scientists focus on strategy and hypotheses. The market reflects this measurable ROI, with rapid growth signaling that the shift is well underway.

Add it up—cost, time, and success—and the ROI is clear. The real question isn’t whether your lab can adopt AI drug findy software. It’s whether you can afford not to.
Frequently Asked Questions about AI in Drug Findy
As we steer the exciting landscape of AI drug findy software, many questions naturally arise. We’ve gathered some of the most common ones to provide clarity and insight into this transformative field.
How does AI actually speed up the drug findy process?
The short answer? AI tackles the bottlenecks that have plagued drug findy for decades, turning what used to take years into a matter of weeks or even days.
First, AI accelerates target identification by analyzing vast multi-omics datasets—genomics, proteomics, metabolomics—to quickly pinpoint and validate promising disease targets. What would take human researchers years of painstaking analysis, AI can accomplish in a fraction of the time by identifying subtle patterns and correlations that might otherwise go unnoticed.
Second, when it comes to molecule design, generative AI can create billions of novel compounds in silico (via computer simulation) in minutes. These aren’t just random molecules—they’re optimized for desired properties like potency, selectivity, and synthesizability. This eliminates the need for countless cycles of physical synthesis and testing, which traditionally consumed enormous amounts of time and resources.
Third, AI drug findy software platforms conduct virtual screening of these enormous chemical libraries, rapidly identifying potential candidates that are most likely to bind to the target. Instead of testing compounds one by one in the lab, we can screen billions virtually and focus our wet lab efforts only on the most promising hits.
Finally, and perhaps most importantly, AI predicts ADMET properties (Absorption, Distribution, Metabolism, Excretion, Toxicity) and potential side effects early in the process. This allows us to eliminate unpromising candidates before costly and time-consuming wet lab experiments or clinical trials begin. By catching potential failures early—whether due to poor bioavailability, toxicity concerns, or off-target effects—AI drastically reduces the overall failure rate and accelerates the journey from concept to candidate.
What kind of data is needed to train AI drug findy models?
Effective AI drug findy software models are hungry learners, and they thrive on diverse, high-quality data. Think of it this way: the better the data we feed into these models, the more accurate and reliable their predictions become.
To build robust and predictive models, we typically need multi-omics data—genomic, proteomic, and metabolomic information. This includes DNA sequences, RNA expression profiles, protein structures, and protein-protein interaction networks. This data is crucial for understanding disease mechanisms and identifying novel targets.
We also rely heavily on chemical structure libraries: large databases of known molecules, their 2D and 3D structures, and their properties. These libraries help AI models understand chemical space and learn what makes a molecule drug-like.
Bioactivity data from past experiments is equally essential. This includes information on how specific compounds interact with biological targets—binding affinities, enzyme inhibition data, receptor activity, and more. The more high-quality bioactivity data available, the better an AI can predict the activity of new molecules.
We also need ADMET data (experimental results on absorption, distribution, metabolism, excretion, and toxicity) to train models that can predict these critical properties for new drug candidates. And when available, clinical trial data—including patient responses, side effects, and trial outcomes—helps AI predict the likelihood of clinical success for new compounds.
The quality and diversity of data are paramount for accurate predictions. As experts note, the importance of robust data for AI success cannot be overstated. Poor-quality or biased data leads to unreliable predictions, which is why data harmonization and federated access to diverse datasets are so critical in modern drug findy.
Is AI going to replace medicinal chemists?
This is a common and understandable concern, but our answer is a resounding “no.” AI drug findy software is not designed to replace medicinal chemists—it’s designed to empower them.
Think of AI as a super-efficient data analyst and a tireless virtual experimenter rolled into one. It can process billions of data points, identify complex patterns, and generate novel molecular designs far faster than any human could. But here’s the thing: AI doesn’t have intuition, creativity, or the deep scientific understanding that comes from years of experience.
What AI does brilliantly is handle the heavy lifting—the massive-scale data processing, pattern recognition, and repetitive screening tasks that would otherwise consume a chemist’s time. This means medicinal chemists spend less time on manual data analysis and designing compounds that are likely to fail, and more time on what they do best: strategic decision-making (interpreting AI-generated insights and formulating hypotheses), creative problem-solving (tackling the truly challenging scientific questions that require human ingenuity), and validation and interpretation (critically evaluating AI predictions and understanding the nuances of biological systems).
AI also frees up chemists to collaborate more effectively with biologists, pharmacologists, and clinicians, integrating diverse perspectives to drive breakthroughs. Rather than replacing medicinal chemists, AI lifts their role, allowing them to operate at a higher, more strategic level. The result? Faster innovation, smarter decisions, and breakthroughs that would otherwise be impossible. AI is the co-pilot, but human expertise remains firmly in the driver’s seat.
Conclusion: Your Next Breakthrough Starts with the Right Data Foundation
The journey of drug findy, once characterized by daunting timelines and astronomical costs, is being fundamentally reshaped by AI drug findy software. We’ve moved from a world of educated guesses and laborious trial-and-error to one where precision, prediction, and rapid innovation are the norm. AI is no longer a futuristic concept; it’s an indispensable tool empowering researchers to identify targets, design molecules, and predict outcomes with unprecedented speed and accuracy.
Looking ahead, the future is incredibly exciting. The vision of “drug findy and design AI factories,” combining generative AI with robotics, is rapidly becoming a reality. Large Language Models (LLMs) are also starting to play a significant role in understanding complex biological texts and generating insights from scientific literature. This future promises a continuous feedback loop between in silico predictions and in vitro validation, accelerating the path from promising molecule to life-changing medicine.
But here’s the truth we’ve learned through 15 years of working with pharmaceutical organizations: the power of this revolution hinges on one critical element—the right data foundation. The most sophisticated AI models are only as good as the data they consume. It doesn’t matter how advanced your algorithms are if they’re working with incomplete, siloed, or poor-quality data.
This is where many organizations hit a wall. Research data often sits trapped in different departments, hospitals, or institutions. Bringing this information together while maintaining security, privacy, and compliance is a massive challenge. Yet it’s absolutely essential. Access to high-quality, harmonized, and securely managed biomedical data is the bedrock upon which all successful AI drug findy software implementations are built.
At Lifebit, we’ve built our federated AI platform specifically to solve this challenge. Our platform provides secure, real-time access to global biomedical and multi-omic data, enabling advanced AI/ML analytics without compromising data privacy or intellectual property. Through components like our Trusted Research Environment (TRE), Trusted Data Lakehouse (TDL), and R.E.A.L. (Real-time Evidence & Analytics Layer), we deliver the data harmonization, federated governance, and analytics capabilities that pharmaceutical research demands. This means your AI models can learn from vast, diverse datasets while your sensitive data stays exactly where it belongs—secure and compliant.
Your next breakthrough isn’t just about the algorithms; it’s about connecting those algorithms to the insights hidden within vast, diverse datasets. It’s about building on a foundation that’s secure, scalable, and designed for the complexities of modern biomedical research.

