Clinical Research IT Your Essential Guide to Informatics and Innovation

Why Clinical Trials Are Broken—And How Next-Gen Tech Can Save Time, Money, and Lives
Clinical research information technology is changing how new medicines are developed. Simply put, it’s the application of digital tools and systems to manage, analyze, and secure data throughout the entire clinical trial process.
This technology helps:
- Streamline Data Collection: Automating how information is gathered from patients and devices.
- Improve Data Management: Storing, organizing, and ensuring the accuracy and integrity of vast datasets.
- Boost Operational Efficiency: Speeding up trial timelines and reducing manual work.
- Improve Collaboration: Connecting researchers, sites, and patients more effectively.
- Ensure Compliance & Security: Meeting strict regulatory standards and protecting sensitive patient data.
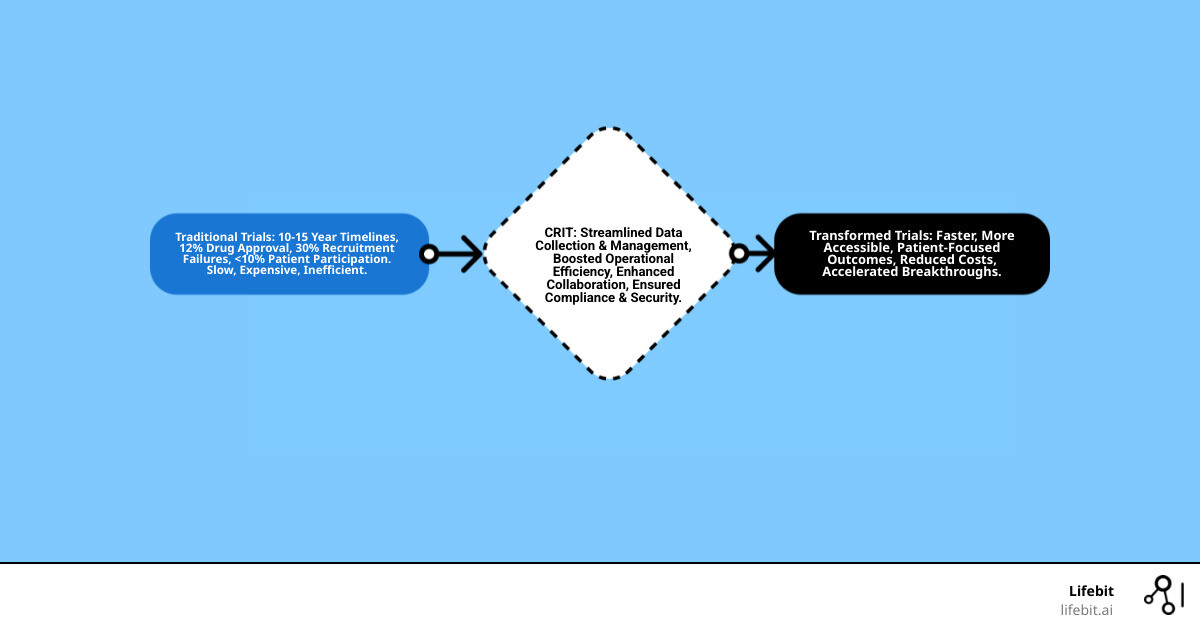
The journey from a new findy to a life-saving treatment is often broken. It’s a long, complex road. Clinical trials, the bridge between theory and practice, can take 10 to 15 years or more. Only about 12% of drugs entering trials ever make it to market.
Recruitment is a major hurdle. It causes 30% of study failures, with fewer than 10% of U.S. patients enrolling in trials. This slow, expensive process desperately needs a change.
That’s where advanced clinical research information technology steps in. It’s the key to making trials faster, more efficient, and more accessible.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit. With a PhD in Biomedicine and expertise in computational biology, AI, and high-performance computing, I’ve spent over 15 years building solutions in clinical research information technology to transform global healthcare. This guide will explore how these next-gen tools are fixing clinical trials, making them smarter and more patient-focused.
The Digital Toolkit: Essential Building Blocks for Modern Clinical Research
In today’s fast-evolving healthcare landscape, clinical research information technology (CRIT) has become the indispensable backbone of the entire research lifecycle. It’s no longer just about collecting data; it’s about intelligent data management, ensuring research integrity, and driving operational efficiency at every stage. We’ve moved beyond clipboards and paper charts to sophisticated digital ecosystems that can handle the complexity of modern studies.
Core Components of Clinical Research Information Technology
At its heart, clinical research information technology comprises several interconnected systems, each playing a crucial role in supporting the research lifecycle (Q1). Think of it as a well-orchestrated symphony, where each instrument contributes to the overall harmony of a successful trial.
-
Electronic Data Capture (EDC) Systems: These are the digital workhorses for collecting patient data directly from study sites. Secure web applications like REDCap allow researchers to build and manage online surveys and databases, offering automated export procedures for seamless data downloads to statistical packages like SPSS, SAS, Stata, and R. We can use these systems for both internal and external users, ensuring consistency and accuracy from the get-go.
-
Electronic Health Records (EHR) Integration: Imagine the power of linking patient care data directly to research. Systems like Epic EHR, when integrated with a Clinical Research Management System, allow physicians to receive up-to-date information on their patients participating in clinical trials. This integration is vital for tracking study subjects, maintaining billing compliance, and leveraging real-world data (RWD) for research. For instance, at NYU Langone Health, this integration helps in identifying patients for trials and managing their participation seamlessly.
-
Clinical Trial Management Systems (CTMS): These comprehensive software platforms are designed to manage the planning, performing, and reporting functions of clinical trials. They handle everything from participant contact information and tracking deadlines to milestone management. A CTMS helps reduce delays by providing centralized data access for experts, facilitating site identification and recruitment, and offering control over subject enrollment. The goal is to move from fragmented systems to an integrated technology suite that streamlines workflows and improves usability.
-
Scalable Data Storage and Management (e.g., DataCore): Modern clinical research generates massive amounts of data—from genomics to imaging to patient-reported outcomes. Robust systems like DataCore provide enterprise-level support for electronic data capture, storage, management, integrity, and extraction. This ensures that data is not only securely housed but also readily available for analysis and sharing, crucial for big data analysis in fields like cancer biology and population health.
-
Unified Research Portals (e.g., Research Navigator): To simplify the administrative burden on researchers, single sign-on portals like Research Navigator are invaluable. These platforms allow investigators to manage their entire research portfolio, submit protocols, and access information related to ongoing clinical trials and studies. This centralization empowers researchers to focus more on the science and less on navigating disparate systems.
-
Laboratory Information Management Systems (LIMS): For studies involving biological samples, LIMS are essential for managing laboratory operations, tracking samples, and submitting requests for core services. This ensures the integrity and traceability of laboratory data, a critical component of research quality.
These core components, when effectively integrated, provide a robust infrastructure for managing the entire research lifecycle, from study design and patient recruitment to data analysis and regulatory submission.
Empowering Research Teams and Slashing Administrative Overhead
One of the most significant impacts of clinical research information technology is its ability to empower site staff and drastically reduce administrative burden (Q7). We know that traditional trials are fraught with manual processes, which are time-consuming and prone to error.
With CRIT, we see:
- Automated Workflows: Repetitive tasks, such as scheduling, data entry, and report generation, can be automated. This frees up valuable time for research staff to focus on patient care and more complex aspects of the study.
- Reduced Manual Data Entry: Instead of transcribing data from paper forms, EDC systems allow direct data input, minimizing errors and speeding up data availability. This means less time chasing down missing information and more time analyzing results.
- Real-time Patient Tracking: Integrated systems provide immediate insights into patient progress, adherence, and safety. Clinical Research Management Systems, for example, help researchers track study subjects efficiently.
- Improved Communication and Collaboration: Platforms often include secure messaging and document-sharing features, allowing seamless communication between study coordinators, investigators, and sponsors. This ensures everyone is on the same page, reducing delays and misunderstandings.
- Streamlined Billing and Compliance: Integration with financial systems helps maintain billing compliance, a complex and often error-prone area in clinical trials. This reduces administrative headaches and ensures financial transparency.
By leveraging these technologies, we can move away from the inefficiencies that plague traditional trials, allowing our research teams to operate with greater agility, accuracy, and focus.
From Paper to Platform: How Digital Innovation Is Changing Trials
The change of clinical research from paper-based processes to digital platforms is nothing short of revolutionary. This shift is not just about digitizing existing workflows; it’s about fundamentally rethinking how trials are designed and conducted. We’re now seeing the power of integrating everyday technologies and cutting-edge innovations to boost operational speed and improve the quality of research.

Decentralizing Trials: Bringing Research Directly to Patients
One of the most profound shifts driven by clinical research information technology is the rise of decentralized clinical trials (DCTs) (Q8). This model leverages technology to enable remote participation, effectively bringing research directly to patients. The benefits are immense:
- Overcoming Geographic and Socioeconomic Barriers: Traditionally, clinical trial participation has been limited by a patient’s proximity to a research site. This often excludes individuals from rural areas or those with mobility issues, childcare responsibilities, or demanding jobs. DCTs, facilitated by telemedicine technologies and mobile health apps, allow individuals to participate from the comfort of their homes.
- Increasing Participant Diversity: The lack of diversity in clinical trials is a well-documented problem, with fewer than 10% of patients in the U.S. enrolling, and even lower rates among some racial/ethnic communities. By removing physical barriers, DCTs can significantly increase diversity among clinical trial participants, ensuring that study populations better reflect the real-world patient population. This is crucial for understanding how therapies work across different demographics.
- Telemedicine and Virtual Visits: The COVID-19 pandemic acted as a powerful catalyst for the adoption of telehealth. Weekly virtual health care visits for Medicare beneficiaries in the U.S. soared from 13,000 before the pandemic to 1.7 million in April 2020. This demonstrated the feasibility and acceptance of remote interactions, paving the way for virtual study visits, remote monitoring, and electronic informed consent (eConsent).
- Real-World Data Collection: Beyond scheduled visits, DCTs use wearable devices and other digital tools for continuous, remote monitoring and data collection. This provides a richer, more frequent dataset than traditional trials, offering insights into how treatments affect participants in their daily lives.
By decentralizing study activities, we can improve trial results, improve participant convenience, and ultimately accelerate medical breakthroughs.
The Power of AI, Blockchain, and Mobile Health
The integration of advanced digital technologies like Artificial Intelligence (AI), blockchain, and mobile health (mHealth) is fundamentally changing clinical trial design and operations (Q2). These technologies are not just incremental improvements; they are game-changers.
-
Artificial Intelligence (AI): AI is revolutionizing nearly every aspect of clinical research. We’re using AI for:
- Patient Identification and Recruitment: AI algorithms can analyze vast datasets, including EHRs, to identify eligible patients for trials much faster and more accurately than manual methods. This helps address the critical challenge of recruitment delays, which cause about 30% of study failures.
- Trial Design Optimization: AI can simulate trial outcomes, predict potential challenges, and optimize study protocols, leading to more efficient and effective designs.
- Data Analysis: From image analysis for diagnostic devices to identifying patterns in complex multi-omic data, AI and Machine Learning (ML) algorithms can process and interpret data at speeds and scales impossible for humans. For instance, AI has been used to analyze billions of molecules for rapid drug development, as seen during the COVID-19 pandemic.
- Pharmacovigilance: AI can monitor for adverse events and identify safety signals in real-time, improving patient safety.
-
Blockchain Technology: This distributed ledger technology offers unprecedented levels of data security, transparency, and integrity in clinical trials.
- Secure Data Logging: Blockchain data can be time-stamped and immutably logged, meaning any attempts to alter data create a new, traceable record rather than erasing the old one. This significantly reduces the risk of data manipulation.
- Participant Control: A unique aspect is the potential for participants to retain partial control of the data they submit via a blockchain platform, deciding whether to share that information with researchers for future trials. This fosters trust and empowers patients.
- Improved Data Quality: By providing a verifiable audit trail, blockchain improves the trustworthiness and reliability of clinical trial data.
-
Mobile Health (mHealth) and Wearable Devices: With approximately 8 billion mobile phone subscriptions worldwide, mHealth is a pervasive force.
- Continuous Data Collection: Wearable devices (smartwatches, fitness trackers) and mobile apps can passively and continuously collect a wealth of real-world data, including heart rate, blood pressure, sleep patterns, and activity levels. This provides a more comprehensive picture of a patient’s health than periodic clinic visits.
- Patient Engagement and Adherence: Mobile apps can deliver interventions, provide reminders, and facilitate communication, improving patient engagement and adherence to study protocols.
- Remote Monitoring: Clinical-grade remote telemetry devices can monitor acute cardiac activity, and specialized equipment can log glucose levels for diabetes studies or conduct sleep studies from home, all synchronizing data automatically.
These technologies enable a shift towards more dynamic, real-time, and patient-centric trials, promising to accelerate the development of much-needed therapies.
Navigating the Maze: The Biggest Challenges in Clinical Research IT
While the promise of clinical research information technology is immense, its implementation is not without problems. We often face significant challenges and limitations when integrating these powerful tools into existing research ecosystems. Understanding these barriers is the first step toward addressing them effectively (Q3).
- Implementation Barriers: Adopting new IT systems can be complex, requiring substantial upfront investment in hardware, software, and training. Existing workflows may need to be redesigned, and staff resistance to change can be a factor.
- Data Silos: Many institutions operate with disparate systems that don’t communicate effectively, creating isolated “data silos.” This fragmentation hinders a holistic view of patient data and complicates data sharing.
- High Costs: Developing, implementing, and maintaining cutting-edge IT systems can be expensive, especially for smaller research organizations or academic centers with limited budgets.
- System Complexity: Advanced systems can be complex to use, requiring specialized technical expertise. This can lead to a steep learning curve for research staff and potential user adoption issues.
- User Adoption: Even the most sophisticated technology is useless if researchers and staff don’t adopt it. Factors like ease of use, perceived benefit, and adequate training are critical for successful integration.
The Critical Need for Data Standardization and Interoperability
One of the most significant challenges in clinical research information technology is the lack of universal data standardization and interoperability (Q4). Imagine trying to read books written in a dozen different languages without a translator—that’s often the reality of clinical data.
- Data Fragmentation and Inconsistent Formats: Data collected from various sources (EHRs, wearables, lab systems) often comes in different formats and uses different terminologies. This “data fragmentation” makes it incredibly difficult to combine, analyze, and compare information across studies or even within a single large trial.
- The Importance of Open Standards: This is where data standardization bodies like the Clinical Data Interchange Standards Consortium (CDISC) play a crucial role. CDISC creates definitions, models, and specifications for data representation, ensuring that data is consistently structured and understood.
- Fast Healthcare Interoperability Resources (FHIR): A key advancement in this area is FHIR (Fast Healthcare Interoperability Resources), a standard published by HL7® for healthcare data exchange. FHIR provides a common language and set of resources for exchanging electronic health information. Its modular design and use of modern web standards make it easier to implement and integrate across different systems. FHIR aims to ensure that electronic health records are available, findable, and understandable as patients move through the healthcare ecosystem.
- Seamless Data Exchange: By adopting standards like CDISC and FHIR, we can achieve true interoperability, allowing for seamless data exchange between different IT systems. This open ups the full value of research data, enabling researchers to conduct more robust analyses, share findings more easily, and accelerate the translation of findies into clinical practice. Without interoperability, our data remains locked in silos, limiting its potential.
Ethical, Regulatory, and Privacy Headwinds
The rapid evolution of clinical research information technology brings with it a complex web of ethical, regulatory, and privacy considerations that researchers must carefully steer (Q6). As we gather more data, more frequently, and from more diverse sources, safeguarding participant rights and data security becomes paramount.
- Informed Consent in the Digital Age (eConsent): Traditional paper-based informed consent processes are giving way to electronic consent (eConsent). While eConsent can improve participant understanding and streamline the process, it raises questions about digital literacy, accessibility, and ensuring that participants truly grasp the implications of data sharing in a digital environment.
- Data Privacy (HIPAA, GDPR): Protecting sensitive patient data is a non-negotiable ethical and legal requirement. Regulations like HIPAA in the USA and GDPR in Europe set stringent standards for how personal health information is collected, stored, shared, and used. Any clinical research information technology system must be built with these regulations at its core, ensuring anonymization and robust access controls.
- Security Risks: The more data we digitize and share, the greater the potential for cyber threats. Robust cybersecurity measures, including encryption, multi-factor authentication, and regular audits, are essential to protect against data breaches and unauthorized access.
- Regulatory Compliance: Regulatory bodies like the FDA and EMA are continually updating their guidance for the use of digital technologies in clinical trials, including electronic records, eConsent, and real-world data. Researchers must stay abreast of these evolving requirements to ensure their studies are compliant.
- The Digital Divide: While mobile technology is widespread (8 billion subscriptions globally), a “digital divide” persists. Individuals from underserved groups and rural areas in the U.S. are less likely to have home broadband access or own a smartphone. This can exacerbate existing disparities in clinical trial participation and raises ethical concerns about equitable access to research opportunities. We must actively work to bridge this divide to ensure that digital trials are inclusive.
Addressing these ethical, regulatory, and privacy considerations requires a proactive approach, including early engagement with Institutional Review Boards (IRBs), adherence to data standards, and a commitment to patient-centric design.
What’s Next? The Future of Clinical Research Informatics
The field of informatics is evolving at a breathtaking pace, driven by the increasing complexity and data demands of modern clinical research (Q9). We are moving into an era where clinical research information technology will not only manage data but actively generate insights, predict outcomes, and facilitate entirely new models of scientific inquiry.
The future landscape of medical research will be shaped by the continued integration of advanced analytics, real-world evidence (RWE), and collaborative data-sharing paradigms. We are seeing a shift from simply collecting data to intelligently leveraging it through sophisticated computational methods.
Future Trends in Clinical Research Information Technology
Looking ahead, several key trends and potential advancements in clinical research information technology will redefine how we conduct medical research (Q10):
- Hyper-automation and AI-driven Trial Design: We will see increasing automation not just of individual tasks, but of entire processes within clinical trials. AI will move beyond patient identification to actively design trial protocols, predict patient responses, and optimize resource allocation. This will dramatically reduce the time and cost associated with bringing new therapies to market.
- Digital Biomarkers: Wearable devices and mHealth apps will become even more sophisticated, enabling the collection of “digital biomarkers”—objective, quantifiable physiological and behavioral data collected using digital health technologies. These biomarkers will offer continuous, real-world insights into disease progression, treatment efficacy, and patient safety.
- Patient-Centric Platforms: Future IT systems will be designed with the patient at the very center, offering intuitive interfaces for data input, real-time feedback, and greater control over their health information. This fosters trust and engagement, crucial for long-term trial success.
- Registry-Based Trials: Leveraging existing patient registries and electronic health records (EHRs) for research will become more common. This approach, often referred to as “pragmatic trials,” allows for more efficient recruitment and the collection of real-world outcomes data, bridging the gap between clinical trials and everyday clinical practice.
- The Rise of Federated Analysis for Secure, Global Collaboration: This is a particularly exciting area. Our own work at Lifebit focuses on a next-generation federated AI platform that enables secure, real-time access to global biomedical and multi-omic data. Instead of moving sensitive data, we bring the computation to where the data resides. This approach, known as federated learning, allows researchers to analyze vast datasets across different institutions and geographies (e.g., in the USA, UK, Europe, Canada, Singapore, Israel, and other regions across 5 continents) without compromising patient privacy or data sovereignty. With built-in capabilities for harmonization, advanced AI/ML analytics, and federated governance, platforms like ours power large-scale, compliant research and pharmacovigilance across biopharma, governments, and public health agencies. This means we can accelerate breakthroughs and save lives by truly open uping the power of global health data.
These trends signify a move towards a more interconnected, intelligent, and patient-empowered future for clinical research.
Conclusion: The Fast Track to Smarter, More Inclusive Clinical Research
We stand at the precipice of a new era in medical findy, one profoundly shaped by clinical research information technology. We’ve seen how these digital tools are systematically dismantling the traditional barriers that have long plagued clinical trials—from their prohibitive length and cost to the persistent challenges of patient recruitment and data integrity.
The power of integrated, standardized data, facilitated by interoperable systems like FHIR and robust data management platforms, is undeniable. It allows us to move from fragmented insights to a holistic understanding of disease and treatment. The decentralization of trials, powered by mobile health and telemedicine, is not just a convenience; it’s a fundamental shift towards more inclusive research that reaches diverse populations globally. And the intelligent application of AI and blockchain promises unprecedented efficiency, security, and ethical data governance.
The future of clinical research is collaborative, decentralized, and patient-centric. It’s a future where cutting-edge clinical research information technology acts as the catalyst, changing complex biological data into life-saving therapies with unprecedented speed and precision.
At Lifebit, we are proud to be at the forefront of this change. Our federated AI platform is specifically designed to enable secure, real-time access to global biomedical data, accelerating breakthroughs by allowing researchers to collaborate and analyze data across diverse datasets without compromising privacy. We believe that by embracing these innovations, we are not just improving clinical trials; we are setting the fast track to a healthier, more equitable future for all.
Find how our federated AI platform can empower your research and accelerate the development of life-changing medicines.
Learn more about Lifebit’s Federated Biomedical Data Platform
Explore our insights on Health Data Analytics
Understand the future of Decentralized Clinical Trials

