Finding the Needle in the Bio-Haystack with AI

AI Biomarker Findy: How to Find Life-Saving Targets in Hours, Not Years
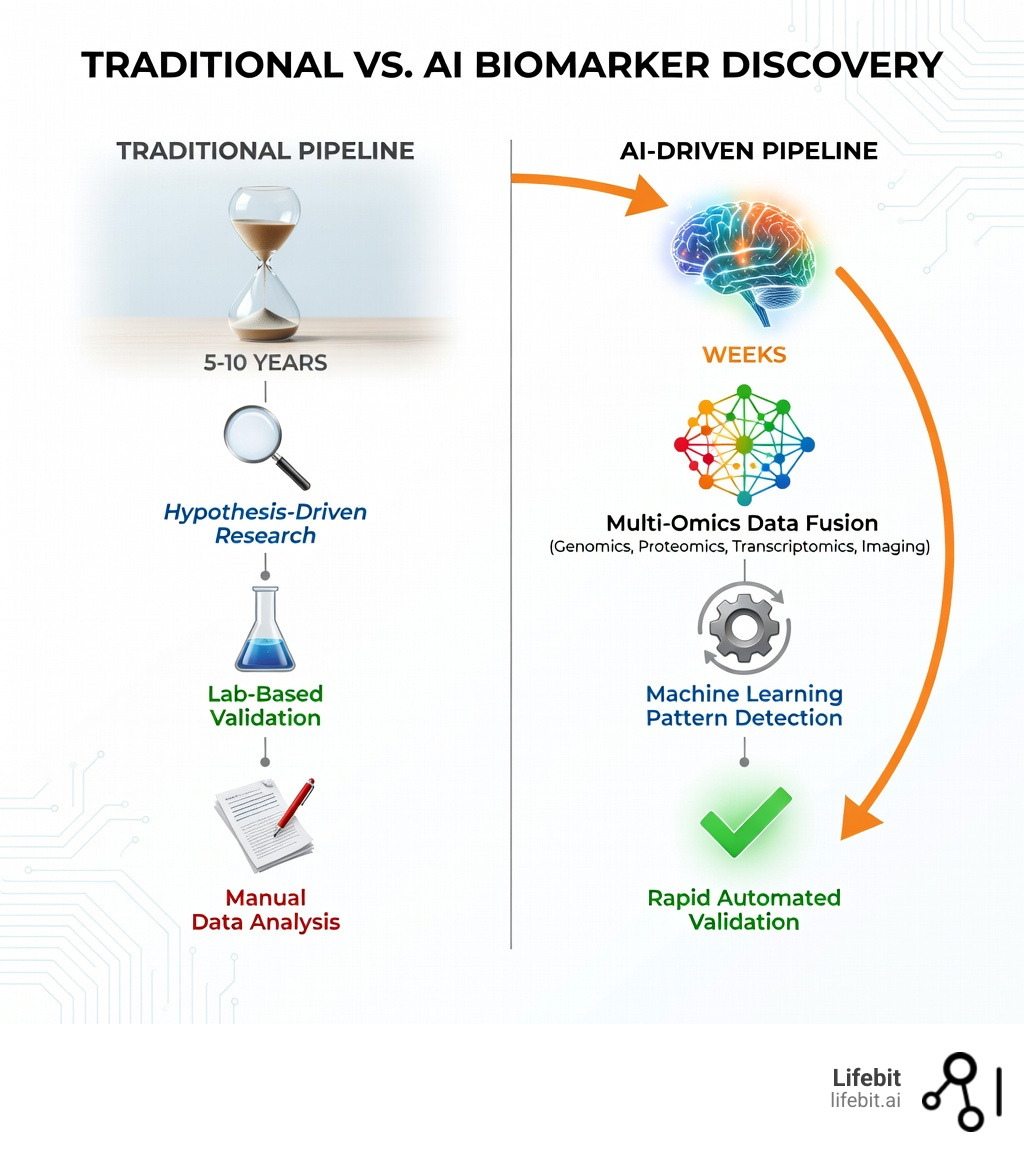
AI biomarker findy is changing how we detect, diagnose, and treat cancer by uncovering hidden patterns in massive datasets that human analysis would miss. Here’s what makes it so powerful:
- Multi-omics integration: AI combines genomic, transcriptomic, proteomic, and imaging data to identify complex biomarker signatures
- Superior accuracy: Machine learning models outperform traditional methods in classifying cancer types and stages, especially for breast, lung, brain, and skin cancers
- Early detection: AI can spot subtle patterns in radiomics, pathomics, and molecular data that signal cancer before symptoms appear
- Personalized treatment: AI-finded biomarkers predict which patients will respond to specific therapies, including immunotherapy
- Faster findy: What used to take years of lab work can now happen in hours through automated pattern recognition
The numbers tell the story. Cancer kills approximately 10 million people every year worldwide, with 70% of those deaths occurring in low- and middle-income countries. Traditional biomarker findy methods are too slow, too expensive, and too limited to address this crisis at scale.
AI changes everything. Modern clinical trials now capture tens of thousands of clinicogenomic measurements per patient. AI-driven models can detect highly precise biomarker signatures linked to different cancer subtypes with accuracy rates above 85%. They analyze millions of data points across imaging, histology, genomics, and electronic health records to find the exact molecular needles in vast biological haystacks.
The shift from hypothesis-driven to data-driven findy means we’re no longer guessing which biomarkers matter. AI systematically explores potential biomarkers in an automated and unbiased way, uncovering novel genes and protein signatures that traditional methods never would have found.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over 15 years building federated platforms that power AI biomarker findy across secure, compliant environments for pharma and public health organizations globally. My work in computational biology and genomics has centered on enabling precision medicine through scalable AI biomarker findy tools that work with real-world data.
Stop Guessing: Why AI Biomarker Discovery is the New Oncology Standard
In the traditional oncology model, biomarkers were often viewed as simple diagnostic “on/off” switches, such as the presence of a specific gene mutation like BRAF or EGFR. Today, we are elevating biomarkers from mere diagnostic tools to indispensable orchestrators of personalized treatment paradigms. This shift is driven by the sheer complexity of cancer biology, which is far too intricate for human clinicians to map using isolated measurements.
The “one size fits all” approach to cancer treatment is effectively dead because it fails to account for intratumoral heterogeneity—the fact that different parts of the same tumor can have different genetic profiles. To achieve true precision, we must move toward multiparameter approaches that incorporate dynamic biological processes and immune signatures. AI biomarker discovery allows us to integrate multi-omics, standardized assay platforms, integrative data analysis, and machine learning to build comprehensive biological signatures that reflect the tumor’s evolution in real-time.
From Data Points to Treatment Orchestrators
Traditional methods often struggle with the heterogeneity of tumors. A single needle biopsy might miss the critical mutations driving a patient’s disease simply because of where the needle was placed. AI-driven models, however, can amalgamate radiography, histology, and genomics to improve diagnostic precision. By identifying these complex patterns, AI helps clinicians predict likely progression, including the potential for recurrence and what outcome can be expected. This is particularly vital in the era of liquid biopsies, where AI can detect trace amounts of circulating tumor DNA (ctDNA) in a blood sample, providing a non-invasive way to monitor treatment efficacy and detect relapse months before it would appear on a standard CT scan.
Hard Computing vs. AI/ML: Choosing the Right Tool
While “hard computing” (traditional rule-based algorithms) has served us well in simple medical imaging, it lacks the flexibility required for the high-dimensional data of modern oncology. Hard computing requires a human to define every rule, which is impossible when dealing with the 20,000+ genes in the human genome and their millions of potential interactions.
| Feature | Hard Computing Algorithms | AI and Machine Learning |
|---|---|---|
| Data Handling | Excellent for structured, low-dimensional data | Superior for high-dimensional multi-omics |
| Flexibility | Static; requires manual rule updates | Dynamic; learns and adapts from new data |
| Pattern Recognition | Limited to pre-defined parameters | Uncovers non-intuitive, non-linear patterns |
| Interpretability | High (White box) | Improving (via Explainable AI/XAI) |
| Scalability | Struggles with “Big Data” complexity | Built for large-scale biomedical data access |
| Discovery Mode | Hypothesis-driven (test what you know) | Data-driven (uncover what you don’t know) |
Deep Learning & NLP: The Tech Slashing Biomarker Discovery Timelines
The “engine room” of ai biomarker discovery consists of several distinct but overlapping technologies. At Lifebit, we see the most significant breakthroughs coming from the fusion of these methods within AI-powered research workflows. By automating the heavy lifting of data processing, these technologies allow researchers to focus on biological interpretation rather than data cleaning.
How Deep Learning Drives AI Biomarker Discovery
Deep learning (DL) is the most potent tool in our arsenal for scaling biomarker discovery workflows. Unlike traditional machine learning, which requires humans to “feature engineer” (tell the computer what to look for), deep learning uses neural networks to automatically identify relevant features. This is critical for identifying “signatures”—groups of genes or proteins that work together—rather than single-point mutations.
One of the most exciting recent developments is AI-driven predictive biomarker discovery with contrastive learning. This approach, exemplified by frameworks like the Predictive Biomarker Modeling Framework (PBMF), allows researchers to distinguish between prognostic biomarkers (which tell us how a disease will progress regardless of treatment) and predictive biomarkers (which tell us how a specific patient will respond to a specific drug). Contrastive learning works by comparing “responders” to “non-responders” in a high-dimensional space, highlighting the subtle molecular differences that dictate drug sensitivity.
Natural Language Processing (NLP) and Clinical Records
Not all biomarkers are molecular. Sometimes, the “biomarker” is a pattern of symptoms, comorbidities, or outcomes hidden in unstructured clinical notes. NLP is revolutionizing how we extract numerical data related to cancer, including tumor grade, size, and behavior from Electronic Health Records (EHRs). Large Language Models (LLMs) can now parse millions of physician notes to identify patients who experienced specific side effects or exceptional responses, linking these real-world outcomes back to their genomic data. This creates a feedback loop that accelerates the validation of new biomarker candidates.
Radiomics and Pathomics: The Visual Revolution
AI is also turning medical images into mineable data, a field known as quantitative imaging.
- Radiomics: Extracts thousands of features from CT, MRI, and PET scans that are invisible to the human eye, such as voxel-level heterogeneity and shape descriptors that correlate with underlying gene expression.
- Pathomics: Uses AI to analyze digital pathology slides at the sub-cellular level. AI can identify the physical distance between cells, which type of cells are present, and how they are organized. For example, AI can quantify the density of tumor-infiltrating lymphocytes (TILs), which is a critical biomarker for immunotherapy success.
Multi-Omics Integration: Detecting Early-Stage Cancer Before Symptoms Appear
The true power of ai biomarker discovery lies in integration. Isolated measurements, such as a single protein level in the blood, are no longer sufficient for early detection because they often lack the specificity required to distinguish cancer from inflammation. By combining imaging and molecular data, we can detect early-stage cancers and identify hard-to-detect tumors that might otherwise be missed until they reach an advanced stage.
Spatial Biology: Location Matters
The complex heterogeneity of tumors makes it challenging to identify new biomarker candidates using traditional “bulk” sequencing, which grinds up a tumor sample and loses all architectural information. Spatial biology techniques now allow us to reveal the spatial context of dozens (or more) markers within a single tissue.
Recent studies suggest that the distribution (rather than simply the absence or presence) of a spatial interaction can actually impact response. For instance, if immune cells are present but “excluded” to the periphery of the tumor, the patient is unlikely to respond to immunotherapy. AI models like HEX and MICA can now generate “virtual” spatial proteomics maps from routine H&E pathology slides, allowing us to see these tumor-immune interactions at single-cell resolution without the need for expensive, specialized hardware. This “virtual staining” could democratize precision medicine in low-resource settings.
The Multi-Omics Marvel
When we pair spatial data with multi-omics profiling (including genomic, epigenomic, and proteomic data), we gain a holistic view of the disease. Epigenomics, in particular, is a rising star in early detection; AI can identify DNA methylation patterns that act as “fingerprints” for specific cancers in the blood long before a tumor is visible on an MRI. For instance, integrated multi-omics recently helped in identifying the functional role of two genes, TRAF7 and KLF4, which are frequently mutated in meningioma. This level of insight is essential for AI-powered target identification and validation, as it allows researchers to see not just that a gene is mutated, but how that mutation ripples through the entire biological system.
Predicting Immunotherapy Response: How AI Digital Twins Forecast Patient Outcomes
The ultimate goal of precision oncology is to move from reactive treatment to proactive management. AI-derived biomarkers are revolutionizing cancer treatment, driving advancements in both therapeutics and prognoses by allowing doctors to simulate treatment outcomes before a single drug is administered.
Forecasting Outcomes with Digital Twins
Predictive models could ultimately facilitate a paradigm shift within oncology. By using a patient’s multi-omic data to create a “Molecular Twin,” AI can integrate clinical, genomic, and proteomic data to predict outcomes for complex cases like pancreatic adenocarcinoma, which has historically been very difficult to treat. These models can process fluorescence imaging data to detect circulating tumor cells and suggest how different patients will respond to specific treatments. This allows for “in silico” testing, where thousands of drug combinations can be simulated to find the one most likely to shrink a specific patient’s tumor.
Immunotherapy: Identifying the Responders
Immunotherapy has been a game-changer, but only about 20-30% of patients respond to it. The rest may suffer from severe side effects without any clinical benefit. AI can pinpoint biomarker signatures that help determine which patients are predisposed to react to checkpoint inhibitors. For example, AI models can analyze the tumor microenvironment to assess tumor-infiltrating CD8 cells and the expression of PD-L1 across different cell types, providing a non-invasive way to predict response to anti-PD-1 or anti-PD-L1 therapies. Furthermore, AI is being used to predict “immune-related adverse events” (irAEs), allowing clinicians to identify patients who might have a dangerous hyper-inflammatory response to treatment, thus improving patient safety.
Solving the Black Box: Secure AI Biomarker Discovery Without Data Risks
Despite the promise, ai biomarker discovery faces significant hurdles. The most prominent is the “Black Box” problem—the difficulty in understanding why an AI model made a specific prediction. If a model identifies a biomarker but cannot explain the biological mechanism, clinicians are hesitant to use it. To build clinical trust, we must prioritize Explainable AI (XAI) and algorithmic transparency, using techniques like SHAP (SHapley Additive exPlanations) to show which genes contributed most to a prediction.
Addressing Bias and Ensuring Equity
If an AI model is trained on data from a single population (e.g., individuals of European descent), it may fail when applied to others, leading to “algorithmic bias.” We must ensure that biomedical AI benefits diverse populations to avoid exacerbating health disparities. This requires:
- Diverse Datasets: Actively seeking data from underrepresented groups in global biobanks.
- Data Harmonization: Using advanced harmonization techniques to ensure data from different sources, recorded in different formats, can be compared accurately.
- Bias Mitigation: Implementing class-imbalance techniques during model training to ensure rare cancer subtypes are not ignored by the algorithm.
Scaling Discovery with Federated Governance
The biggest bottleneck in AI research is often data access. Sensitive patient data is frequently trapped in silos due to privacy regulations like GDPR and HIPAA. This is where federated learning in healthcare becomes vital. Instead of moving data to a central server (which creates security risks), federated learning moves the model to the data.
At Lifebit, we use Trusted Research Environments (TREs) to allow researchers to train their AI models across multiple global datasets simultaneously without the data ever leaving its secure home. This “federated” approach ensures:
- Data Privacy: Sensitive patient information remains behind the hospital’s or biobank’s firewall.
- Equitable Access: Researchers in smaller institutions can collaborate on global datasets without needing massive local storage infrastructure.
- Real-Time Insights: Platforms like our Trusted Data Lakehouse provide the infrastructure needed for high-speed, compliant analysis of petabyte-scale genomic data.
Cut Trial Costs: How AI-Driven Validation Speeds Up Clinical Success
The traditional clinical trial model is often described as slow, expensive, and brittle, with a failure rate of over 90% for new oncology drugs. AI is making trials more adaptive and personalized, allowing research teams to modify trial designs based on accumulating data and ensuring the right patients are enrolled from day one.
Real-World Evidence (RWE) and Synthetic Arms
AI allows us to leverage real-world data from EHRs and insurance claims to validate biomarkers outside the controlled environment of a trial. We can even create “synthetic control arms” using historical patient data. This means that instead of giving half the patients in a trial a placebo, researchers can use AI to predict how a control group would have fared based on thousands of past cases. This can improve survival risk by 15% in retrospective analyses by refining patient selection criteria and ensuring that only those most likely to benefit are enrolled in the active treatment arm.
Case Study: Functional Precision Medicine
Advanced models, including organoids (3D mini-organs grown from a patient’s own cells) and humanized mouse models, are now being integrated with AI. Organoids excel at recapitulating the complex architectures and functions of human tissues and have been used in the identification of biomarkers for drug screening. When AI analyzes the drug responses of these organoids, it can enhance the robustness and predictive accuracy of studies. By testing drugs on a patient’s organoid before the patient themselves, AI can identify the most effective therapy with near-perfect accuracy, paving the way for faster clinical validation and reducing the time it takes for life-saving drugs to reach the market.
Frequently Asked Questions about AI Biomarker Findy
How does AI improve early cancer detection?
AI analyzes vast multi-omics and imaging datasets to uncover complex patterns that human clinicians might miss. By integrating radiomics (imaging) and molecular signatures, AI can identify “signals” of cancer—such as circulating tumor DNA or specific protein expressions—long before physical symptoms appear.
What is the difference between prognostic and predictive biomarkers?
- Prognostic Biomarkers: These provide information about the patient’s overall cancer outcome (e.g., risk of recurrence or overall survival) regardless of the treatment they receive.
- Predictive Biomarkers: These identify individuals who are likely to respond to a specific therapeutic intervention (e.g., determining if a patient will respond to a specific immunotherapy drug). AI is particularly skilled at distinguishing these two types in high-dimensional data.
How does AI handle multi-omics data?
AI uses specialized architectures, such as deep neural networks and contrastive learning, to “fuse” different data types (genomics, proteomics, transcriptomics). It finds non-linear correlations across these layers, creating a unified biological signature that is more accurate than any single-data-type analysis.
Conclusion: The Future of Precision Oncology
The era of finding the “needle in the bio-haystack” has arrived. AI biomarker findy is no longer a futuristic concept; it is a clinical necessity. By moving from hypothesis-driven research to data-driven findy, we are finally beginning to match the complexity of cancer with the power of our analytical tools.
At Lifebit, we believe that the future of oncology depends on secure, federated collaboration. Our platform enables researchers to access global biomedical data in real-time, providing the Trusted Data Lakehouse and AI-driven analytics needed to turn vast datasets into life-saving treatments.
Whether you are looking to scale your biomarker discovery pipeline or leverage real-world evidence for target validation, the tools are now at your fingertips. The bio-haystack is massive, but with AI, the needles are finally coming into focus.
Ready to accelerate your research?
Explore Lifebit’s AI-Powered Target Identification & Translation Solutions

