Keep Calm and Collect Data: Patient Registry Software UK Guide

Stop Wasting Data: How Patient Registry Software UK Teams Can Accelerate Research
Patient registry software UK platforms are purpose-built tools that collect, store, and analyse structured clinical data on defined patient populations — enabling everything from NHS commissioning decisions to rare disease research.
When evaluating the right platform for your team, consider these primary deployment models:
- Disease-Specific Registries: Focused on quality improvement and multi-centre support, often available via G-Cloud procurement.
- Medical Device & Safety Platforms: Specialized in tracking implants and regulator-ready PROMs.
- NHS-Integrated Solutions: Built specifically for NHS information governance and N3/HSCN security.
- Research & Genetic Registries: Designed for rare disease research with dynamic consent and participant engagement.
- Federated Research Platforms: The gold standard for large-scale multi-omic research, allowing secure analytics without moving sensitive data.
The UK has a long history of disease-specific registries — the UK CF Registry, the IBD Registry, and more recently the relaunched UK Myotonic Dystrophy Patient Registry — all demonstrating how the right platform can transform patient outcomes, benchmarking, and research at national scale. But with so many options, different regulatory requirements, and NHS integration challenges, choosing the right software is genuinely hard.
I’m Dr. Maria Chatzou Dunford, CEO and Co-founder of Lifebit, and my work building federated genomics and biomedical data infrastructure has given me a front-row seat to the real challenges organisations face when selecting and deploying patient registry software UK-wide. In the sections below, I’ll break down exactly what to look for, how the leading platforms compare, and what the data says about outcomes.
Essential patient registry software uk terms:
What is Patient Registry Software and Why Does the NHS Need It?
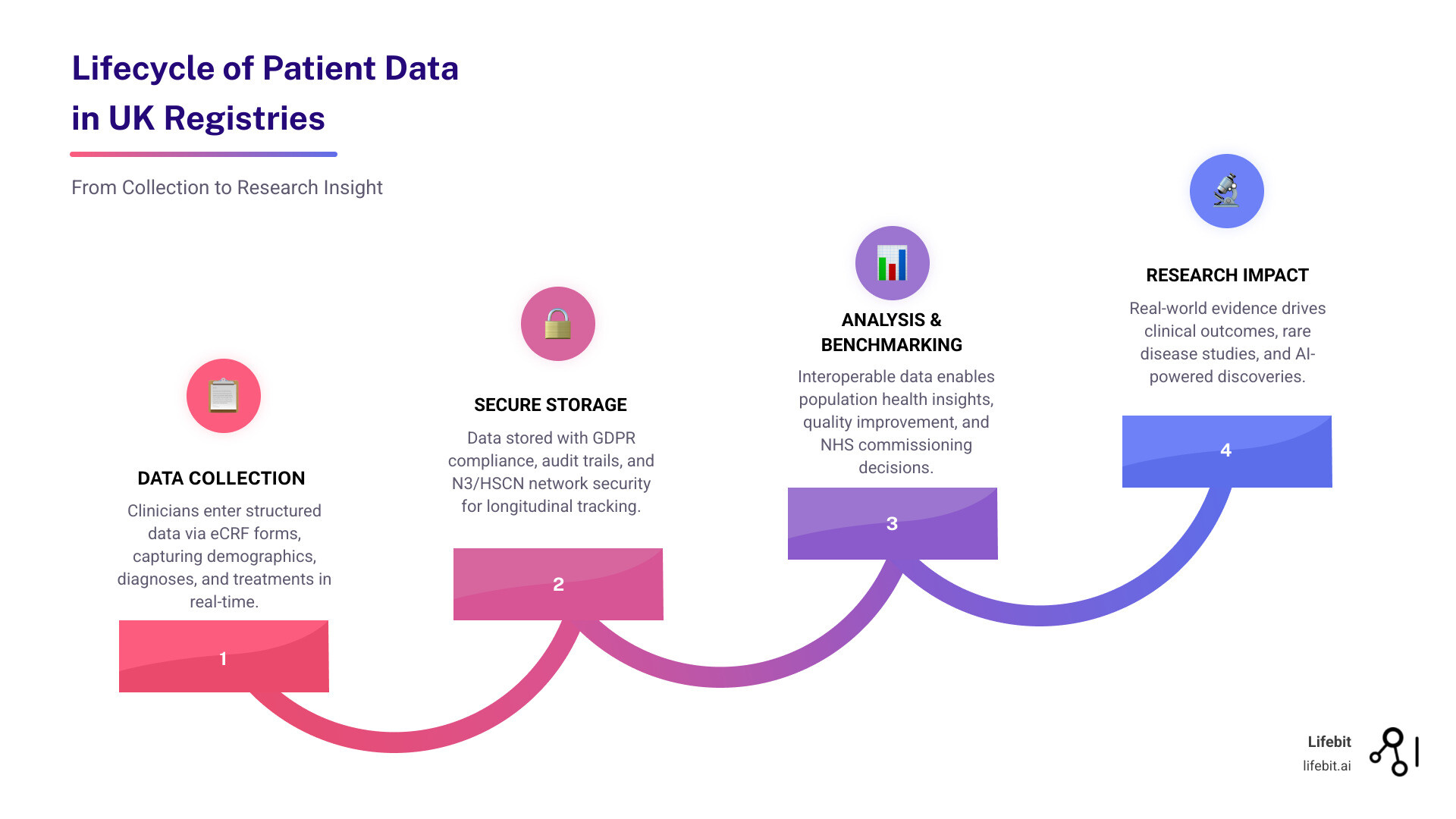
At its core, what are patient registries? They are organised systems that use observational study methods to collect uniform data (clinical and otherwise) to evaluate specified outcomes for a population defined by a particular disease, condition, or exposure. In the UK, this software is the backbone of “data-driven medicine,” serving as the primary mechanism for translating clinical observations into actionable national policy.
The evolution of these systems in the UK has moved from simple spreadsheets and paper-based records to sophisticated, cloud-native platforms. This transition is driven by the NHS Long Term Plan, which envisions a “Learning Health System” where every patient interaction contributes to a broader understanding of disease progression and treatment efficacy. The NHS relies on these registries for several critical functions:
- NHS Commissioning and Resource Allocation: Data from registries like the UK CF Registry helps the NHS decide how to allocate funding and resources for specialist care. By understanding the exact geographic distribution and clinical needs of patients with cystic fibrosis, commissioners can ensure that specialist centres are appropriately funded and located.
- Benchmarking and Quality Improvement: By comparing performance across different trusts, providers can identify gaps in care. For example, the IBD Registry has historically enabled sites to benchmark their service performance against national levels, identifying variations in biologic prescribing or surgical outcomes that might otherwise go unnoticed.
- Population Health Management: Understanding the longitudinal journey of patients allows for better planning of long-term care for chronic conditions. This is particularly vital for the UK’s ageing population, where multi-morbidity is becoming the norm rather than the exception.
- Real-World Evidence (RWE) and Regulatory Oversight: Registries provide a “real-world” look at how treatments perform outside of highly controlled clinical trials. This is vital for the MHRA (Medicines and Healthcare products Regulatory Agency) and NICE (National Institute for Health and Care Excellence) when evaluating the cost-effectiveness and safety of new therapies post-market.
- Post-Market Surveillance for Medical Devices: Following the Cumberlege Review, there has been an increased focus on tracking the long-term outcomes of medical implants. Registry software provides the necessary infrastructure to track every device from implantation to explantation, ensuring patient safety is monitored over decades.
For a deeper dive into how these systems function and the different types of data they can capture, check out our disease registries complete guide.
Essential Features of Patient Registry Software UK Providers Must Have

When we evaluate patient registry software UK options, we look for features that bridge the gap between clinical utility and technical compliance. A registry is only as good as the data it collects and the ease with which clinicians can enter that data without disrupting their existing workflows.
1. Customizable eCRFs and Multi-Centre Support
UK registries often involve dozens of NHS Trusts, each with slightly different local workflows. Software must support multi-centre data entry while allowing for highly customizable electronic Case Report Forms (eCRFs). This ensures that whether you are tracking a rare neuromuscular disease or a common cardiac procedure, the data points are relevant. Advanced platforms now offer “drag-and-drop” form builders that allow clinical leads to update data collection points in real-time as new research priorities emerge.
2. Interoperability and Semantic Standards
The “holy grail” of registry software is the ability to pull data directly from existing Electronic Patient Records (EPRs) like Rio, Epic, or Cerner. Integration with GP Connect and adherence to HL7 FHIR standards are becoming non-negotiable. However, technical connectivity is only half the battle; semantic interoperability is equally important. Leading UK platforms use standardized terminologies like SNOMED CT for clinical terms and ICD-10/11 for diagnoses, ensuring that data collected in Manchester is perfectly comparable to data collected in London.
3. Data Quality and Validation Frameworks
To be useful for research, registry data must be clean. Modern patient registry software includes automated validation rules that flag outliers or missing fields at the point of entry. This reduces the need for retrospective data cleaning, which is often the most expensive and time-consuming part of registry management. Some platforms now incorporate the NHS Data Quality Maturity Index (DQMI) standards to provide real-time feedback to Trusts on the quality of their submissions.
4. PROMs, PREMs, and Patient Engagement
Modern registries must move beyond clinical data to include Patient-Reported Outcome Measures (PROMs) and Patient-Reported Experience Measures (PREMs). Leading platforms excel here by offering:
- Automated Reminders: Using SMS or email to keep patients engaged over years, ensuring high retention rates for longitudinal studies.
- Dynamic Consent: Allowing patients to update their participation preferences as study protocols evolve, which is a core requirement of the UK’s ethical framework.
- Multilingual Support: Ensuring diverse patient inclusion, which is critical for the UK’s multicultural population and for reducing health inequalities.
For more details on the technical specifications required for these systems, see our breakdown of the four key patient registry software requirements.
Navigating UK Regulations: GDPR, NHS Standards, and Security
In the UK, data security isn’t just a feature; it’s a legal mandate. Any patient registry software UK provider must navigate a complex web of regulations to ensure patient trust and legal compliance. The landscape is governed by the “Five Safes” framework: Safe People, Safe Projects, Safe Settings, Safe Data, and Safe Outputs.
- GDPR and the Data Protection Act 2018: Software must be built with “privacy by design.” This includes robust audit trails that track every single access event. In the context of registries, this also means managing the “Right to be Forgotten” while maintaining the statistical integrity of the research dataset.
- The Caldicott Principles: Registry software must support the role of the Caldicott Guardian within an NHS Trust. This involves ensuring that the use of confidential patient-identifiable information is justified, necessary, and kept to the absolute minimum.
- National Data Opt-Out: Since 2022, all health and care organisations in England must comply with the national data opt-out policy. Registry software must have automated mechanisms to check patient records against the opt-out list before data is exported for research purposes.
- N3/HSCN and the DSPT: While the trend is moving toward cloud-based solutions, they must still align with the NHS Data Security and Protection Toolkit (DSPT). This is an online self-assessment tool that allows organisations to measure their performance against the National Data Guardian’s 10 data security standards.
- Cyber Essentials Plus: For many NHS contracts, having basic Cyber Essentials is no longer enough. Providers are increasingly expected to hold Cyber Essentials Plus, which involves an independent technical audit of the provider’s systems to ensure they are resilient against common cyber threats.
- Trusted Research Environments (TREs): The Goldacre Review (2022) recommended a shift away from “data dissemination” (sending data to researchers) toward “data access” (bringing researchers to the data). Modern registry platforms are increasingly being built as TREs, where data stays within a secure environment and only the results of analyses are exported.
You can find more on navigating these hurdles in our clinical registry solutions guide.
Comparing Patient Registry Software UK Options and Pricing Models
Choosing the right deployment model significantly impacts both the budget and the administrative burden on your IT team. In the UK, procurement is often streamlined through the G-Cloud framework, which allows public sector bodies to buy cloud-based services without a full tender process.
| Model | Description | Best For |
|---|---|---|
| Cloud-Based (SaaS) | Hosted by the provider; accessible via web browser. | Rapid deployment, multi-centre collaboration, and lower upfront costs. |
| On-Premise | Installed on the Trust’s local servers. | Organizations with strict internal data sovereignty requirements or legacy infrastructure. |
| Managed Service | A hybrid where the provider manages the infrastructure and security on behalf of the client. | Large-scale national registries requiring high uptime and dedicated support. |
Pricing and Total Cost of Ownership (TCO)
The cost of patient registry software UK varies wildly based on scale. For instance, high-end clinical platforms listed on the G-Cloud 14 framework can start at approximately £30,000 per unit per year, but this is often just the baseline. When calculating the TCO, organisations must also consider:
- Implementation and Configuration: Setting up custom eCRFs and workflows can cost between £10,000 and £50,000 in one-off fees.
- Data Migration: Moving data from legacy spreadsheets or older SQL databases into a modern, structured format is often the most underestimated cost.
- Integration Fees: Connecting the registry to an EPR via APIs or HL7 FHIR interfaces may require additional development time from both the registry provider and the EPR vendor.
- Training and Support: Ensuring that clinical staff are proficient in using the system is vital for data quality.
Public sector buyers should consult the Pricing document and the SFIA rate card for detailed breakdowns of day rates for implementation specialists. Many providers offer a “shared development” model where the costs of new features are shared across multiple NHS Trusts, making high-end software more accessible to smaller research groups.
Real-World Impact: UK Case Studies and Future Trends for 2025
The real power of patient registry software UK is seen in its application across diverse clinical fields, from rare diseases to national screening programmes.
The Myotonic Dystrophy Success
In May 2025, the UK Myotonic Dystrophy Patient Registry was relaunched using a modern, user-friendly clinical platform. The pilot showed that an intuitive interface could significantly boost engagement from both patients and clinicians. By allowing patients to upload their own genetic reports and track their symptoms over time, the registry has become a vital tool for clinical trial readiness. This success has paved the way for other neuromuscular registries, such as FSHD and SMA, to migrate to similar advanced platforms that prioritize the patient experience.
The UK CF Registry and Modular Therapies
Sponsored by the Cystic Fibrosis Trust, this registry is a global gold standard. It collects data from over 99% of consenting patients across the UK. Its ability to provide longitudinal insights was instrumental in the introduction of life-changing modular therapies like Kaftrio. By providing the NHS with clear evidence of the drug’s real-world effectiveness, the registry helped secure long-term funding for these treatments.
The National Congenital Anomaly and Rare Disease Registration Service (NCARDRS)
Managed by NHS England, NCARDRS demonstrates the scale at which registry software must operate. It tracks thousands of different conditions across the entire population, providing the data needed to evaluate screening programmes and investigate potential environmental clusters of disease. This requires a platform capable of handling massive datasets while maintaining granular security controls.
Future Trends: Multi-Omics and Federated AI
As we look toward 2025 and beyond, the trend is moving away from “data silos” and toward federated data access. Advanced platforms now allow researchers to access multi-modal data—including clinical, genomic, and imaging data—from millions of patients globally without the data ever leaving its secure home. This is particularly important for rare diseases, where no single country has enough patients to conduct statistically significant research. For those working in these specialized fields, our rare diseases registry complete guide explains how these technologies are helping to solve the “small n” problem by enabling international collaboration while respecting national data sovereignty.
Frequently Asked Questions about Patient Registry Software
How much does patient registry software UK cost?
Pricing typically follows a subscription model based on the number of users or the volume of patients. For high-end clinical platforms, expect to pay around £30,000 per year per registry unit. However, bespoke research platforms may have different structures based on the number of participants or the complexity of the questionnaire builder. Always check the Terms and conditions for hidden costs like data egress fees, API maintenance, or mandatory training modules.
Can patient registry software UK integrate with NHS EPR systems?
Yes, but it requires the software to support modern interoperability standards like HL7 FHIR and APIs. Integration with systems like Rio, Cerner, or GoodX is essential for reducing manual data entry and ensuring that the registry reflects the most up-to-date clinical information. Achieving high-quality patient registries establishment often depends on how well the registry “talks” to the hospital’s existing digital infrastructure. Many modern platforms now use “middleware” to map local hospital codes to national standards automatically.
How does patient registry software UK support rare disease research?
Registries are the lifeblood of rare disease research because they allow for the collection of data from small, geographically dispersed cohorts over long periods. Features like automated reminders and digital screening help maintain high retention rates, which are crucial for longitudinal studies. Furthermore, by using standardized data models like OMOP, UK registries can participate in international research networks, effectively increasing the sample size for rare conditions. Learn more about the four benefits of patient registries for rare disease to see how they drive drug discovery.
Who owns the data in a UK patient registry?
In the UK, the “Data Controller” is typically the organisation that established the registry (e.g., an NHS Trust, a University, or a Charity). The software provider acts as the “Data Processor.” It is vital that the registry’s governance framework clearly defines who can access the data and for what purposes, ensuring that patient consent is always respected. Most registries now include a “Data Access Committee” to review research requests and ensure they align with the registry’s mission.
Conclusion
The UK is at a turning point in how it manages healthcare data. From the closure of legacy systems like the IBD Registry to the rise of AI-powered federated platforms, the landscape of patient registry software UK is evolving rapidly toward greater security and deeper insights.
At Lifebit, we believe that the future of research lies in secure collaboration. Our federated AI platform enables researchers to access and analyse complex biomedical data in-situ, ensuring the highest levels of security and compliance while accelerating the journey from data to discovery. By embracing these next-generation tools, UK healthcare providers can ensure that patient data isn’t just “collected”—it’s put to work to save lives.
Discover how Lifebit is powering the next generation of secure, federated research registries.

