Smart Science and Why AI is the New Lab Partner

AI for Clinical Research: How Lifebit Cuts Trial Costs by 40%
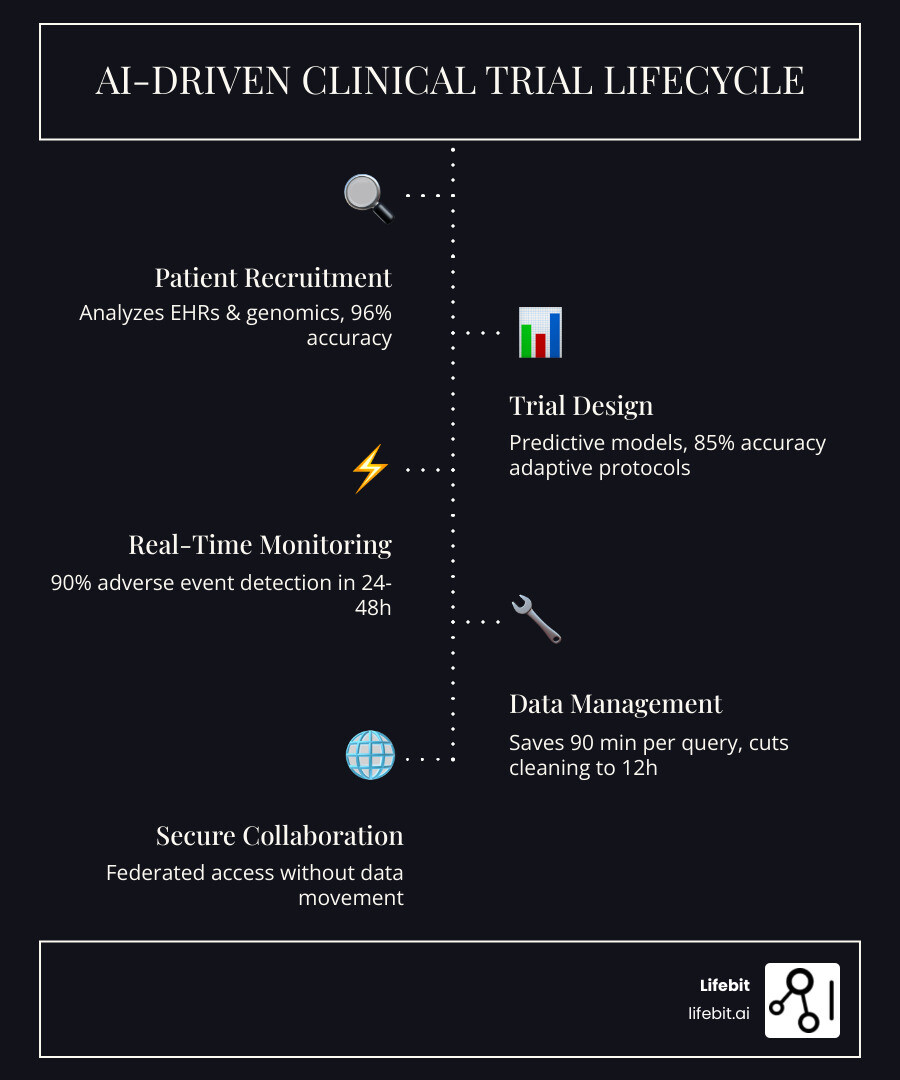
AI for clinical research is transforming how drugs move from lab to patient—cutting trial costs by up to 40%, accelerating timelines by 30–50%, and solving recruitment failures that plague 80% of studies. Here’s what it does:
- Patient Recruitment: AI analyzes EHRs and genomic data to identify eligible participants 3x faster with 96% accuracy
- Trial Design: Predictive models forecast outcomes with 85% accuracy and enable adaptive protocols that adjust in real-time
- Data Management: Automated systems reduce query resolution time by 90 minutes and cut data cleaning from 60–80 hours to 12–16 hours per 100 patients
- Safety Monitoring: Digital biomarkers detect adverse events with 90% sensitivity within 24–48 hours vs. 4–6 weeks for traditional methods
- Cost Reduction: End-to-end AI integration slashes trial costs by up to 40% and shrinks timelines that once stretched across years
Traditional clinical trials burn through billions of dollars and years of time. The average approved drug costs $2.6 billion to develop, with Phase III trials alone consuming $19 million and 6–7 years. Nearly a third of Phase III studies fail due to enrollment issues—86% of all trials miss recruitment schedules. Manual patient screening takes 8 hours per candidate. Data quality problems affect 50% of clinical datasets. And success rates for new drug approvals hover below 12%.
AI changes this equation by automating the tedious work that slows research to a crawl. It reads millions of patient records in seconds, designs smarter trials, monitors safety in real-time, and predicts which protocols will succeed before you spend a dollar on enrollment. Platforms like Lifebit’s federated AI infrastructure let global research teams access diverse datasets—EHRs, genomics, imaging—without moving sensitive data, enabling real-time pharmacovigilance and cohort discovery across secure, compliant environments.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over 15 years building computational biology tools and federated data platforms that power precision medicine globally. My team and I have seen how AI for clinical research eliminates the barriers that once made multi-institutional collaboration impossibly slow—turning months-long queries into minutes and unlocking insights from over 275 million patient records.
Ai for clinical research terms made easy:
The $2 Billion Problem: Why Old-School Trials Waste Time and Money
In the pharmaceutical world, we often talk about “Eroom’s Law”—the frustrating observation that drug discovery is becoming slower and more expensive despite improvements in technology. It is the literal reverse of Moore’s Law in computing. Today, bringing a single drug to market can cost upwards of $2.6 billion, and half of that budget is swallowed by clinical trials.
The statistics are sobering. Scientific research on trial failure rates reveals that only about 12% of drugs entering Phase I ever reach the pharmacy shelf. The primary culprit? Inefficiency. Protocol complexity has skyrocketed, yet many teams still rely on manual data entry and “paper-and-pencil” recruitment methods.
Currently, 86% of trials fail to meet their original recruitment schedules. When a trial stalls, it doesn’t just cost money; it delays life-saving treatments for patients who don’t have time to wait. Manual review of Electronic Health Records (EHRs) is a Herculean task—researchers often spend hours sifting through unstructured notes just to find one eligible candidate. This administrative burden, combined with high Phase III dropout rates, creates a bottleneck that ai for clinical research is uniquely positioned to break.
How Lifebit’s AI for Clinical Research Cuts Enrollment Time by 90%
Patient recruitment is the single biggest “time-waster” in clinical development. Traditional methods are like looking for a needle in a haystack—if the haystack was spread across five different continents and written in three different technical languages.
Lifebit’s approach to ai for clinical research turns this search into a high-speed digital operation. By using Natural Language Processing (NLP) to scan structured and unstructured EHR data, notes, and charts, our platform identifies protocol-eligible patients three times faster than manual review. In real-world settings, such as the Cleveland Clinic, AI systems have demonstrated 170x speed improvements, identifying candidates in minutes rather than hours.
Smarter Trial Design with Lifebit’s AI
Before a single patient is enrolled, AI helps us build a better blueprint. Scientific research on adaptive designs shows that trials capable of adjusting protocols in real-time—based on interim data—are significantly more efficient.
We use AI to optimize trial protocols by simulating thousands of scenarios to find the most effective endpoints and sample sizes. This feasibility analytics ensures that when a study builds, it is anchored in reality. Instead of waiting until the end of a trial to realize a protocol was flawed, AI allows for “go/no-go” decisions much earlier, saving millions in wasted R&D spend.
Digital Twins and Biosimulation: Smaller, Faster, Smarter Trials
One of the most exciting frontiers in ai for clinical research is the use of “Digital Twins.” These are AI-generated forecasts of how a specific patient is likely to respond to a control treatment.
By using digital twins as external comparators, we can reduce the number of human participants needed for a control group. This is a game-changer for rare disease research, where the patient pool is tiny.
| Feature | Traditional Trials | In Silico (AI) Trials |
|---|---|---|
| Patient Pool | Large groups required for control | Smaller cohorts; AI-simulated controls |
| Speed | Years of manual observation | Accelerated via predictive modeling |
| Cost | High ($19M+ for Phase III) | Up to 40% reduction |
| Risk | High participant burden | Lower risk via biosimulation |
Biosimulation allows us to test how a drug might affect various organ systems digitally before it ever enters a human subject. This doesn’t replace human trials, but it ensures that the trials we do run are safer and more likely to succeed.
Stop Waiting Weeks: Get Real-Time Evidence with Lifebit’s R.E.A.L. Layer
The old way of managing trial data involved “data cleaning” marathons. Up to 50% of clinical datasets contain errors or inconsistencies that require manual queries. This “back-and-forth” between investigators and data teams can take weeks.

Lifebit’s Real-time Evidence & Analytics Layer (R.E.A.L.) automates this process. Instead of waiting for a monthly report, we provide real-time monitoring of every data point. This allows for immediate anomaly detection, ensuring that safety signals are caught within 24–48 hours rather than weeks later.
The Future of Data Management with Lifebit AI
Data management is where AI’s “grunt work” capabilities truly shine. Lifebit’s automated systems save up to 90 minutes per query identification and 50 minutes on data transformation, ensuring your data is analysis-ready faster.
We focus on data harmonization—taking messy, fragmented data from different hospitals and turning it into a unified, analysis-ready foundation. This is critical for decentralized trials, where data comes from wearables, home visits, and local labs. By using AI to automate the “boring stuff,” we give your best scientists their time back to focus on high-impact discovery.
Boosting Patient Retention and Medication Adherence
A trial is only as good as its participants’ commitment. Unfortunately, up to 50% of prescriptions in the U.S. are taken incorrectly, and clinical trials are no different. Low adherence can ruin the statistical power of a study.
AI helps us boost retention through:
- Behavioral Monitoring: Identifying patients at risk of dropping out before they actually leave.
- Smart Reminders: Personalized engagement through AI-powered apps that use neuroeconomic principles to nudge compliance.
- Patient Navigation: AI chatbots that answer participant questions 24/7, reducing the burden on site staff.
Eliminate Regulatory Risk: Ethical AI Built for FDA and EU Compliance
Integrating AI into clinical research isn’t just about the tech; it’s about trust. Regulatory bodies like the FDA and the EU (via the AI Act) are rapidly evolving their guidance. We prioritize ethical AI for clinical research by building frameworks that address algorithmic bias.
Scientific research on AI reporting guidelines, such as CONSORT-AI, emphasizes the need for transparency. We ensure that every AI-generated insight is verifiable. Our AI agents produce “traceable” outputs—meaning you can see exactly which data point led to a specific conclusion. This “human-in-the-loop” approach ensures that AI assists, rather than replaces, the expert judgment of a clinician.
AI for Clinical Research: Your Top Questions Answered
How does AI reduce clinical trial costs?
AI eliminates the “hidden costs” of inefficiency. By automating 80% of administrative tasks, shrinking recruitment cycles from months to days, and reducing the need for massive control groups through digital twins, AI can cut overall trial costs by up to 40%. It optimizes resources so that money is spent on science, not paperwork.
Can AI improve patient diversity in trials?
Absolutely. Traditional trials often suffer from “geographic bias,” recruiting only from major academic centers. AI can scan global EHR data to identify diverse cohorts in underserved areas. Furthermore, by analyzing real-world data, we can adjust eligibility criteria to be more inclusive, ensuring that the results of a trial actually apply to the general population.
What are the biggest risks of using AI in trials?
The primary risks are data quality and algorithmic bias. If an AI is trained on non-diverse data, it may produce biased results. There is also the risk of “hallucinations” in generative models. This is why Lifebit insists on rigorous validation, federated governance, and human oversight. AI should be treated as a powerful lab partner, not a solo pilot.
Secure the Future of Your Research with Lifebit’s Federated AI
The era of siloed, slow, and expensive clinical trials is ending. AI for clinical research is no longer a futuristic concept; it is the current standard for teams that want to stay competitive and, more importantly, save lives.
At Lifebit, we believe the secret to better science is secure, real-time access to data. Our federated AI platform allows you to run advanced analytics across a global network of multi-omic and clinical data without the security risks of moving sensitive files. From AI clinical trial optimization to long-term pharmacovigilance, we provide the tech stack that turns big data into big breakthroughs.
Ready to see how AI can transform your research? Let’s build the future of medicine together.
Learn more about our solutions:

