Definitive Guide: Precision Health in Biopharma with Adv. Data Catalogs

Why Multi-Modal Omics Data Is Holding Back Precision Medicine
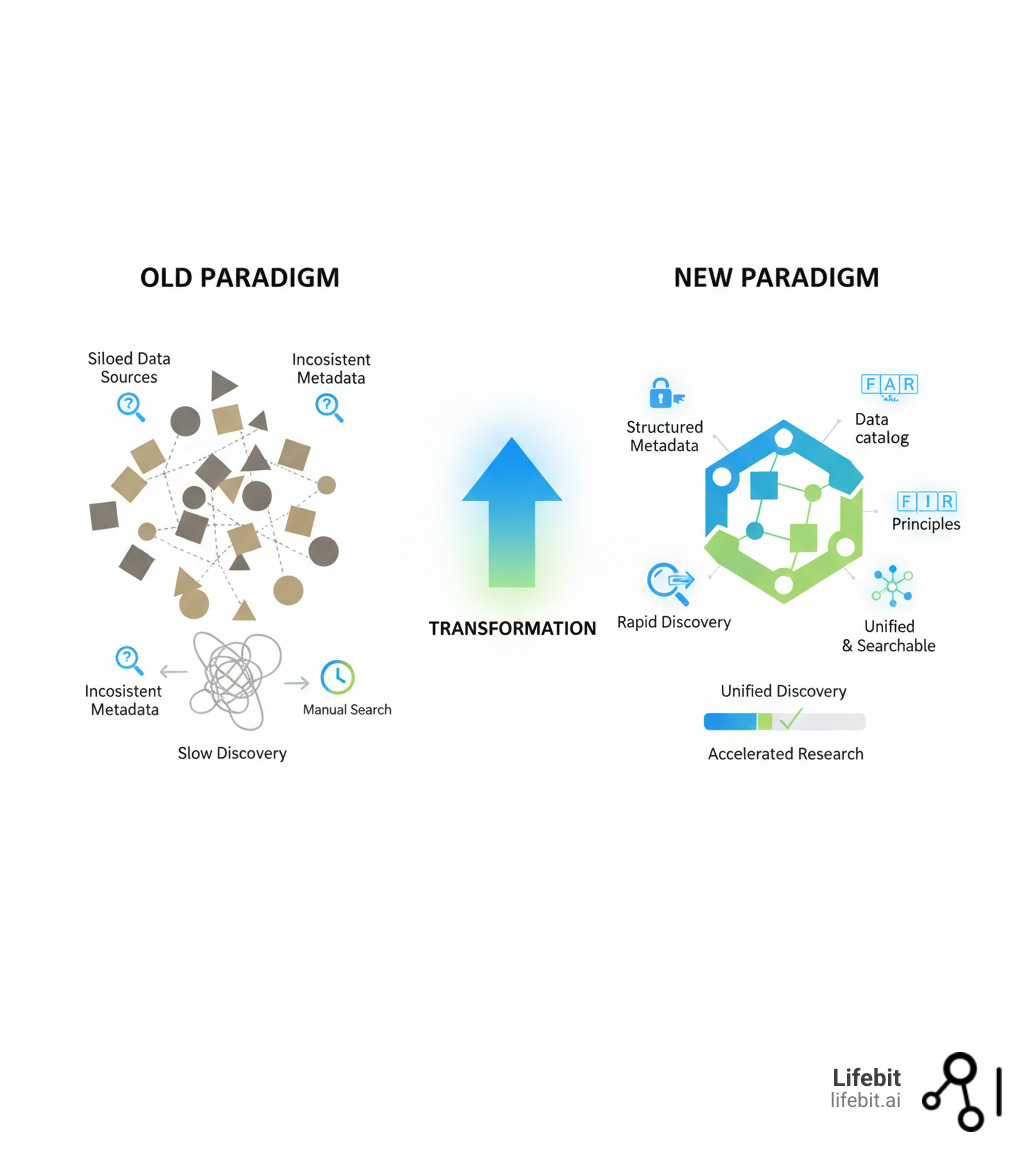
Open uping the potential of precision health with advanced data catalogs addresses a critical bottleneck: biopharma organizations are drowning in genomic, proteomic, metabolomic, and clinical data—but can’t find, access, or trust it fast enough to accelerate drug findy and personalized treatments. The sheer volume and complexity make these datasets nearly impossible to manage with traditional methods.
Researchers grapple with unprecedented data volumes, but historical reliance on ad-hoc systems, scattered databases, and inconsistent metadata has led to wasted time and missed opportunities. Scientists spend more time searching for data than analyzing it. Critical datasets sit locked in silos. Drug development slows.
Key benefits of solving this challenge with advanced data catalogs include:
- 10-20% lower conversion costs and faster R&D cycles
- Order-of-magnitude time savings in data findy (from weeks to hours)
- Improved collaboration with secure, controlled access for internal and external partners
- Compliance by design with granular controls and audit trails for GxP, HIPAA, and GDPR
- Accelerated biomarker findy through structured, FAIR-compliant metadata
The scale of this challenge is immense, with leading platforms managing over 125 petabytes of complex multiomic data for tens of thousands of users globally. Modern advanced data catalogs are changing the game. By applying structured metadata and enabling rapid, semantic search, these catalogs transform data chaos into a streamlined, intuitive experience for researchers.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, with over 15 years of experience in computational biology, AI, and genomics. Throughout my career—from my work at the Centre for Genomic Regulation to leading Lifebit’s federated data platform—I’ve seen how the right data strategy can accelerate findy, reduce costs, and ultimately deliver better patient outcomes. In this guide, I’ll walk you through the challenges, the solutions, and the strategic benefits that make advanced data catalogs a must-have for any biopharma organization serious about precision health.
The Core Challenge: Managing Multi-Modal Omics Data at Scale
Here’s the uncomfortable truth: biopharma is sitting on a goldmine of data—but most organizations can’t actually use it.
The flood of multi-modal omics data from genomics, proteomics, metabolomics, and clinical records represents an unprecedented opportunity. We’re talking about vast genomic sequences, intricate protein structures, and rich clinical datasets that could revolutionize drug findy. This isn’t just one type of data; it’s a complex tapestry woven from different biological layers. Genomics data from whole-genome sequencing (WGS) and whole-exome sequencing (WES) arrives in massive BAM or VCF files. Transcriptomics data (RNA-Seq) reveals gene expression patterns. Proteomics data from mass spectrometry identifies and quantifies proteins, while metabolomics captures a snapshot of cellular processes. Each layer has its own format, its own analytical pipeline, and its own dialect of metadata. Without a unifying system, they are like puzzle pieces from different boxes—impossible to assemble into a coherent picture of disease.
But historically, organizations have cobbled together ad-hoc systems and scattered databases. It’s like trying to organize a library where every book is in a different language and stored in a different building. The result? Valuable data remains locked in silos. Researchers can spend weeks or even months just trying to locate the right dataset—time that should be spent on analysis and innovation. It’s a massive drain on resources and a fundamental barrier to scientific findy. Imagine a computational biologist tasked with identifying potential drug targets for a specific subtype of non-small-cell lung cancer. The required data exists, but it’s scattered. Genomic data is on a local server in one department, clinical trial data with patient outcomes is in a separate, access-controlled database, and proteomics data from a recent experiment is on a collaborator’s cloud drive, described only in an email. The scientist spends the first three weeks just negotiating access, locating files, and manually trying to match patient IDs across these systems. This is not science; it’s digital logistics, and it’s a primary reason why the promise of data-driven medicine has been so slow to materialize.
The life sciences sector is projected to generate more data than any other industry, with some platforms already managing over 125 petabytes of complex datasets. These numbers are a warning signal that traditional approaches won’t cut it. The challenges extend far beyond volume. Each omics modality has unique attributes and varying data formats. Without standardization, integrating these diverse datasets is nearly impossible—yet integration is crucial for understanding disease mechanisms holistically.
Researchers end up struggling with isolated resources, insufficient compute capabilities, and a fundamental lack of trust in data quality. When you can’t find, trust, or integrate data, the promise of precision medicine remains frustratingly out of reach.
Why Traditional Data Management Fails
Traditional data management in biopharma wasn’t built for the scale and complexity of multi-modal omics data. The old playbook fails for several key reasons.
Data silos are the most obvious problem, creating isolated “islands” of information that prevent a comprehensive view of patient data. This is compounded by inconsistent metadata, where different teams use different naming conventions, making it impossible to confidently find and reuse datasets. The metadata becomes unstructured and dispersed, creating a major barrier to findy.
This leads to poor data findability, turning scientists into digital archaeologists who spend more time searching for data than analyzing it. Furthermore, ad-hoc systems often lack the granular access controls and audit trails necessary for compliance with HIPAA, GDPR, and 21 CFR Part 11, creating significant security risks. Finally, legacy platforms create scalability bottlenecks, unable to handle the exponential growth of omics data without performance issues and skyrocketing costs. Together, these shortcomings erode data quality and integrity, stalling precision medicine initiatives.

The Modern Solution: From Data Chaos to a Centralized Findy Engine
There’s a better way forward. Modern advanced data catalogs are powerful engines for scientific findy that transform the overwhelming task of managing diverse datasets into something intuitive. Instead of hunting through scattered databases, researchers can finally find exactly what they need—fast.
At the heart of this modern approach are the FAIR principles: Findability, Accessibility, Interoperability, and Reusability. A robust data catalog provides the practical framework to make them a reality across petabytes of multi-modal omics data.
This is powered by automated metadata enrichment, which uses technologies like Large Language Models (LLMs) to accelerate data wrangling and harmonization. Imagine LLMs expanding cryptic headers and generating descriptive metadata, all without hours of manual effort. This automation is essential for managing the exponential growth of genomic, proteomic, and clinical datasets.
The real game-changer is establishing a single source of truth for all your biopharma data. By providing a semantically rich, unified view, advanced data catalogs let researchers quickly pinpoint relevant datasets, understand their context, and trust their provenance. This centralized approach slashes data search time from weeks to hours, redirecting valuable resources toward meaningful findy and accelerating innovation.
Key Functionalities of a Biopharma-Ready Data Catalog
Not all data catalogs are created equal. For biopharma, you need specialized features designed to handle the unique complexities of multi-modal omics data.
- Automated metadata ingestion: Intuitive loader applications and AI-assisted tools automatically extract and enrich metadata from diverse sources, ensuring consistency at scale without manual data entry. This is crucial for handling unstructured sources like clinical notes or lab reports. For example, AI models can parse a pathology report associated with a tissue sample, automatically tagging the dataset with terms like ‘adenocarcinoma’ or ‘high-grade tumor,’ making it instantly findable for oncology researchers.
- Powerful semantic search: Faceted search capabilities allow researchers to explore data across multiple dimensions (disease, omics type, cohort). This goes far beyond simple keyword matching. By leveraging domain-specific ontologies like the Gene Ontology (GO), Human Phenotype Ontology (HPO), and MeSH, the catalog understands the relationships between biological concepts. A search for ‘neoplasms’ will also return datasets tagged with ‘cancer’ or specific tumor types. This semantic understanding uncovers hidden correlations and connections that simple text searches would miss entirely.
- Full data lineage tracking: Traces the entire lifecycle of each dataset—from origin to analysis—providing a comprehensive audit trail for validation and regulatory compliance. This means capturing not just the source of the raw data, but also the specific software versions (e.g., BWA for alignment, GATK for variant calling), parameters, and reference genomes used in its processing. This ‘digital chain of custody’ is non-negotiable for creating reproducible science and for submitting data to regulatory bodies.
- Version control for reproducibility: Functions as a versioned data registry, tracking changes to ensure analyses can be reliably replicated, a cornerstone of credible scientific research.
- Seamless integration: Connects effortlessly with existing bioinformatics tools, AI/ML platforms, and Trusted Research Environments (TREs). Our federated AI platform at Lifebit includes built-in capabilities for harmonization and advanced analytics in a secure environment.
- Granular access controls: Precise, role-based permissions ensure only authorized individuals can access specific datasets, safeguarding privacy while maintaining research agility.
- Customizable metadata schema: A flexible and extensible framework supports business-specific concepts and accommodates the evolving needs of different research areas.
- Data harmonization capabilities: Integrates disparate datasets into unified, consistent resources, which is essential for overcoming interoperability challenges in federated research.
Together, these functionalities transform data management from a bottleneck into a competitive advantage.
Open uping the Potential of Precision Health with Advanced Data Catalogs
The strategic implementation of a sophisticated data catalog is more than a technical upgrade—it’s a fundamental shift in how biopharma organizations approach R&D. When you move to intelligent data management, you open up capabilities that touch every aspect of the drug development pipeline.
Every hour a researcher spends searching for data is an hour lost from actual science. Every dataset sitting unused in a silo is a potential breakthrough that never happens. Advanced data catalogs address these challenges head-on. By making data findable, accessible, interoperable, and reusable (FAIR), we empower researchers to move faster and develop therapies that are truly personalized. The ultimate goal isn’t just operational efficiency—it’s accelerating breakthroughs that deliver better patient outcomes.
The impact is measurable. Organizations see 10-20% lower conversion costs and faster R&D cycles. They experience order-of-magnitude time savings in data findy, with search times dropping from weeks to hours. The real strategic benefit is changing data from a liability into your most valuable strategic asset.
How Catalogs Accelerate Precision Health Breakthroughs in Biopharma R&D
Advanced data catalogs accelerate the work that matters most: bringing new treatments to patients faster.
It starts with structured metadata. When every dataset has rich, standardized descriptors, researchers can quickly understand its content, origin, and quality. This enables lightning-fast data findability, allowing scientists to locate specific patient cohorts or omics profiles in moments using intuitive semantic search.
The time savings are dramatic, with researchers achieving order-of-magnitude reductions in search time. This speed directly impacts biomarker identification. Precision health relies on finding novel biomarkers, and advanced catalogs make it exponentially easier to find and integrate the diverse datasets (genomics, proteomics, clinical data) necessary for this findy.
The cumulative effect is shorter drug development cycles. Every stage benefits: target identification, preclinical development, clinical trial recruitment, and post-market surveillance. Research increasingly highlights the critical need for frameworks that support large-scale multiomics data integrity and interoperability. Without a proper cataloging infrastructure, the promise of precision medicine remains just a promise.
Scientific research on data integration challenges
The Power of Collaboration: Breaking Down Barriers with Advanced Data Catalogs
In the era of multi-modal omics, collaboration faces unprecedented technical and organizational barriers. Advanced data catalogs are instrumental in breaking them down.
They solve collaboration challenges by providing secure data sharing within a unified platform. Sensitive information can be shared with precise controls, ensuring all stakeholders—from London to New York, across the USA, UK, Canada, Singapore, Israel, and Europe—can access essential data seamlessly and securely.
Our platform includes a Trusted Research Environment (TRE), a secure, controlled space where internal and external collaborators can engage in analysis with pre-loaded tools while maintaining stringent access controls and full audit trails. This allows innovation to flourish without compromising intellectual property or data privacy.
Perhaps most powerfully, our federated governance capabilities enable secure, real-time access to global biomedical data without requiring data movement. This federated approach respects data residency and privacy laws, which is crucial for international research. Teams can perform analyses across distributed datasets, effectively eliminating organizational silos and enabling more comprehensive insights than any single dataset could provide. When collaboration becomes seamless, breakthroughs become inevitable.

Ensuring Ironclad Security and Compliance in a Regulated Industry
In biopharma, protecting sensitive patient data isn’t just good practice—it’s the law. A single breach can derail years of research, trigger massive fines, and erode patient trust. That’s why security and compliance must be baked into every layer of your data infrastructure, not bolted on as an afterthought.
Advanced data catalogs are engineered with this reality at their core. They help biopharma organizations steer the complex regulatory landscape while enabling the speed and collaboration that precision health demands.
Granular access controls are the first line of defense. Our platform ensures access is managed at the most precise level, tied to user roles and project requirements. This means a researcher in London can’t accidentally access oncology data from New York, safeguarding both privacy and intellectual property.
Controlling access is only half the battle. Detailed audit trails record every interaction with data—every search, download, and analysis. This creates an indisputable record for regulatory inspections and internal audits. Robust data governance ties it all together, ensuring policies for data quality, ownership, and lifecycle management are consistently applied.
For global operations, data sovereignty is a critical concern. Our federated AI platform respects these requirements by allowing data to remain in its original jurisdiction while still being findable and usable for analysis. You get the benefits of global collaboration without the legal headaches of cross-border data transfers. These features ensure that open uping the potential of precision health doesn’t come at the expense of security or compliance.
Meeting Regulatory Mandates
Compliance is an ongoing commitment. Advanced data catalogs are built with “compliance by design,” integrating regulatory requirements into the platform’s architecture from day one.
Our systems are designed to meet the stringent requirements of global standards like 21 CFR Part 11, HIPAA, and GDPR. This includes data integrity controls, secure user authentication, time-stamped audit trails, and the full suite of safeguards required to protect patient health information (PHI). For organizations operating in the EU or UK, our solutions are GDPR compliant from the ground up, with features like consent management and data minimization.
GxP data integrity requirements span the entire drug development lifecycle. Our platform provides the necessary controls and documentation to demonstrate GxP compliance, ensuring data is reliable and accurate at every stage. At the heart of this are the ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, and more). An advanced data catalog provides the framework to enforce these principles systematically. Attributable: Every action is tied to a specific user and timestamped in the audit trail. Legible: Data and metadata are stored in a human-readable and computationally accessible format for the required retention period. Contemporaneous: Actions are recorded as they happen. Original: The catalog maintains the primary record or a certified true copy, with full lineage tracing back to the source. Accurate: Version control and validation checks ensure that data transformations are error-free and correctly recorded. By embedding ALCOA+ into the data’s lifecycle, the catalog transforms compliance from a manual, periodic audit into an automated, continuous process. This allows you to innovate with confidence, knowing your data management practices are robust, secure, and fully compliant.
The Strategic Payoff: Driving ROI and Better Patient Outcomes
The adoption of an advanced data catalog isn’t just a technical upgrade; it’s a strategic shift that delivers measurable returns impacting both the bottom line and patient lives.
Biopharma data analytics is now a strategic imperative. Companies that successfully implement these solutions are already seeing tangible benefits: 10-20% lower conversion costs and 10-15% better cost of quality through optimized workflows. In an industry where bringing a single drug to market can cost over $2 billion, these efficiency gains translate into significant savings and shorter development timelines.
- R&D Efficiency: The most immediate payoff comes from giving time back to scientists. When researchers spend hours instead of weeks finding data, that time is returned to actual analysis and findy, compounding across every project.
- Lower Data Management Costs: While there is an upfront investment, the long-term savings are substantial. Automated metadata management, reduced data redundancy, and optimized cloud storage make the infrastructure leaner and less expensive to maintain.
- Faster Drug Target Identification: By rapidly integrating genomic, proteomic, and clinical data, patterns emerge faster. Hypotheses get tested quicker, and promising targets surface months earlier than in a fragmented data environment.
- Development of Personalized Treatments: This is the very heart of precision health. Advanced data catalogs provide the unified view of diverse data types needed to stratify patient populations, identify predictive biomarkers, and design more effective therapies with fewer side effects. This is where data becomes actionable knowledge that changes lives.
- Gaining a Competitive Edge: The life science analytics market is projected to reach $37.20 billion by 2030. Organizations that can extract insights from their data faster don’t just keep pace—they pull ahead. They bring novel therapies to market quicker, secure patents earlier, and establish leadership in emerging therapeutic areas. The strategic payoff is measured not just in dollars saved, but in the acceleration of life-changing treatments reaching patients who need them.
Conclusion
The core message is clear: adopting an advanced data catalog isn’t just a nice-to-have—it’s essential for survival and leadership in today’s data-driven biopharma landscape.
Biopharma organizations are overwhelmed by multi-modal omics data locked in silos, tangled in inconsistent metadata, and impossible to find. This slows drug development and delays breakthroughs. The solution is a modern, biopharma-ready data catalog that transforms chaos into clarity.
By providing automated metadata ingestion, powerful semantic search, and granular access controls, these catalogs slash search times from weeks to hours, accelerate biomarker identification, and help deliver personalized treatments to patients faster. They enable seamless internal collaboration and secure external partnerships across continents—from London to New York, throughout the USA, UK, Canada, Singapore, Israel, and Europe.
With ironclad security and built-in compliance for 21 CFR Part 11, HIPAA, GDPR, and GxP, you can innovate with speed and confidence. The ROI is clear: lower costs, massive time savings, and faster findy. But beyond the numbers, the future of precision medicine depends on our ability to turn raw data into actionable insights. Data catalogs are the cornerstone of that future.
Our federated AI platform is built for exactly this challenge. With components like the Trusted Research Environment (TRE), Trusted Data Lakehouse (TDL), and R.E.A.L. (Real-time Evidence & Analytics Layer), we deliver real-time insights and secure collaboration across hybrid data ecosystems. Built-in harmonization, advanced AI/ML analytics, and federated governance mean you can tackle large-scale research and pharmacovigilance with confidence.
Ready to see what’s possible? Discover the benefits of a structured data catalog and explore how our solutions provide biomedical data access while using cloud computing in healthcare to accelerate your precision health journey. The data revolution is here—it’s time to lead it.

