An Essential Guide to Clinical Research SaaS Providers in USA
Cut Trial Costs 30–70%: The SaaS Shift Reshaping U.S. Studies Now
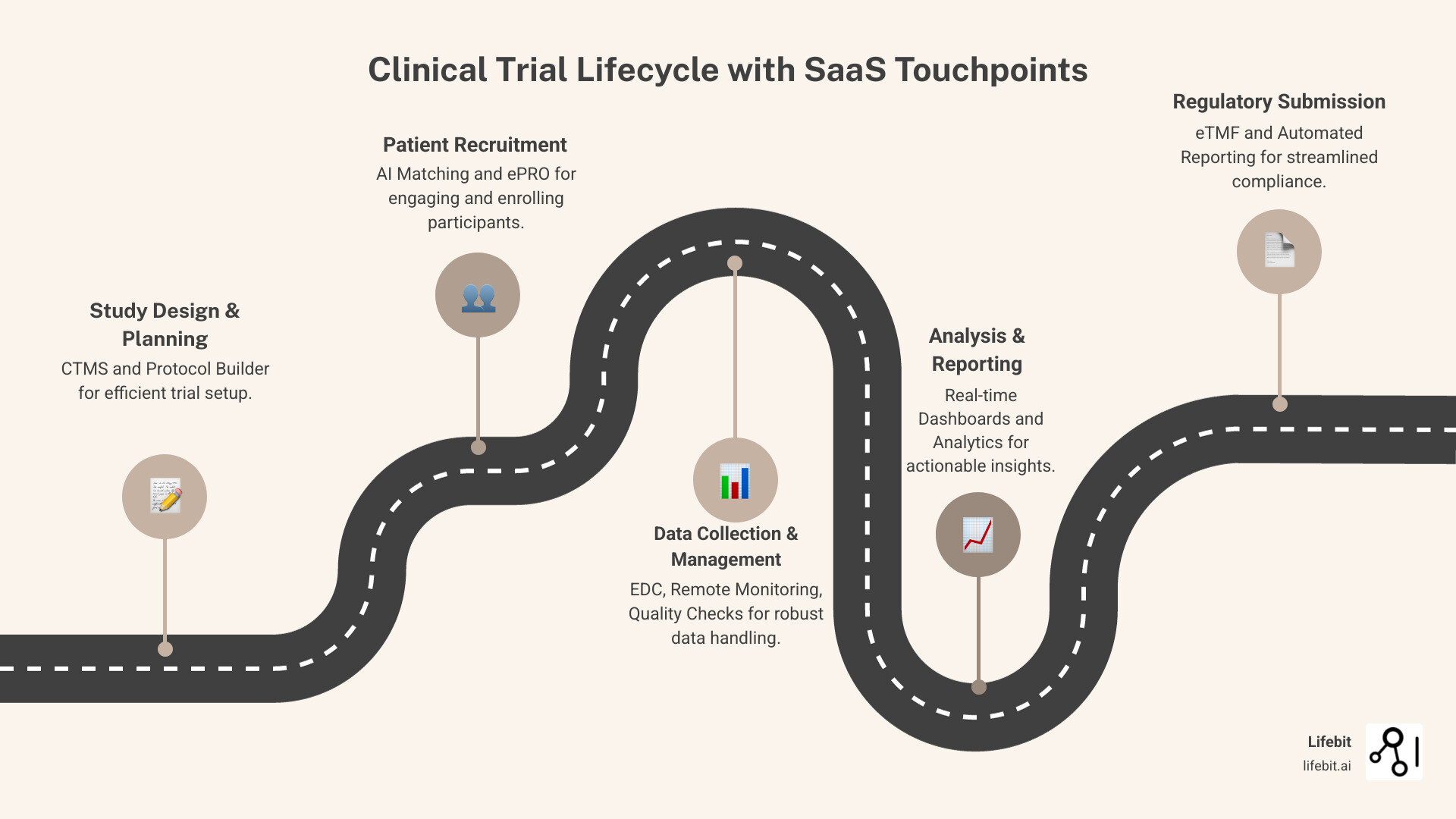
The clinical trial landscape is at a breaking point. With $300 billion spent annually on R&D and drug development timelines stretching longer each year, the industry can no longer rely on fragmented, paper-based systems. Traditional on-premise tools are too slow, too expensive, and too rigid for modern research needs.
Cloud-based SaaS platforms have emerged as the proven solution. Organizations migrating from legacy systems report 30-70% reductions in total cost of ownership, 40% faster study startup times, and dramatic improvements in data quality. The global CTMS market alone is projected to grow from $2.14 billion in 2024 to $9.67 billion by 2035a 14.7% annual growth ratewith North America accounting for the largest share.
The shift to SaaS isn’t just about moving to the cloud. It’s about reimagining clinical trial design, execution, and management through intelligent automation, real-time collaboration, and AI-driven insights. As decentralized trials grew over 380% from 2010 to 2022, the need for unified platforms that connect patients, sites, and sponsors has never been more urgent.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where I’ve spent 15 years building genomics and biomedical data platforms that power global healthcare through federated data analysis. My experience developing cutting-edge tools for precision medicine has given me unique insight into what separates the best clinical research SaaS technology providers in the USA from the restand how these platforms are accelerating the path from findy to approval.
What are the best clinical research saas technology providers in the USA? terms to learn:
Launch Trials 40% Faster: Why U.S. Leaders Are Ditching On‑Prem for SaaS
For decades, clinical research relied on traditional on-premise systems—expensive, slow, and inflexible. This model required organizations to purchase costly software licenses, procure and maintain dedicated servers, and hire large IT teams just to keep the lights on. Today, that paradigm is obsolete. Instead of buying software and hardware, organizations can now access powerful, validated research tools instantly through the cloud. This shift to Software as a Service (SaaS) is a fundamental change in how we run clinical trials, moving from a capital-intensive infrastructure model to a flexible, operational one.
The numbers tell a compelling story. Organizations switching to SaaS platforms report 30-70% reductions in total cost of ownership. This isn’t just about the subscription fee; it’s about eliminating the hidden costs of on-premise solutions: server hardware procurement and replacement cycles, electricity and cooling for data centers, physical security, software licensing and annual maintenance fees, and the salaries of dedicated IT staff for patching, upgrades, and troubleshooting. Studies also show 40% faster startup times with cloud-based platforms. This acceleration comes from streamlined processes like rapid site activation, intuitive drag-and-drop eCRF builders that don’t require custom programming, and on-demand user training, allowing trials to launch in weeks instead of months.
Beyond speed and cost, SaaS platforms improve the work itself. Data quality soars because real-time data capture and automated validation rules catch errors at the point of entry, not months later during database lock. Collaboration becomes seamless, allowing a research coordinator in Boston, a data manager in Bangalore, and a medical monitor in San Francisco to review the same patient data simultaneously. SaaS platforms scale to meet your needs—from a small Phase I study to a global Phase III trial—without requiring infrastructure overhauls. Vendors handle all updates and patches behind the scenes, so you always have the latest features and security protections without any downtime or complex validation projects.
Core Benefits of SaaS in Clinical Research
The advantages of cloud-based platforms reshape how research teams work and make decisions, fostering a more proactive and efficient trial environment.
- Centralized Data Access: Everyone works from a single source of truth. All study information—patient demographics, adverse events, lab results, protocol deviations—lives in one secure cloud environment. This eliminates version control issues and the risk of teams working with outdated information, ensuring decisions are based on the most current data available.
- Real-Time Visibility: Dashboards update instantly, showing current enrollment rates by site, data query aging, and emerging safety signals. This allows teams to respond to issues immediately. For example, if a site is under-enrolling, resources can be reallocated in real-time. If a safety trend is detected, it can be investigated within hours, not after waiting for weekly or monthly reports.
- Elimination of Data Silos: Modern SaaS platforms are designed to break down the walls between clinical operations (CTMS), clinical data (EDC), and regulatory documentation (eTMF). This integration lets information flow where it’s needed without manual data entry or reconciliation, providing a holistic view of trial progress and performance.
- Built-in Security and Compliance: Leading platforms are designed from the ground up to meet stringent requirements like 21 CFR Part 11 and HIPAA. They often provide more robust, multi-layered security—including advanced threat detection and disaster recovery—than individual organizations can afford to build and maintain on their own.
- Agility for Protocol Changes: In research, change is constant. A new scientific insight or a regulatory request can necessitate a protocol amendment. Cloud-based platforms allow for rapid updates to electronic forms, randomization schemes, or data collection workflows, which can be deployed across all sites simultaneously with minimal disruption.
ROI and Cost-Efficiency of SaaS
The financial case for SaaS in clinical research is overwhelming and extends far beyond the initial subscription cost.
- Reduced IT Overhead: SaaS vendors handle all infrastructure, maintenance, security, and support, freeing up your internal IT teams to focus on strategic initiatives rather than routine system administration.
- Predictable Pricing: Subscription-based models (often per-study or per-user) replace massive upfront capital expenditures with predictable operating costs. This allows for better budgeting and financial planning, with the ability to scale costs up or down based on your research pipeline.
- Faster Time to Market: The ultimate ROI is accelerating the path to approval. Shaving months off development timelines by launching studies 40% faster and cleaning data in weeks instead of months gets life-saving treatments to patients sooner and extends the patent life of a commercialized product.
- Shorter Data Cleaning Timelines: Automated validation rules, cross-form logic checks, and AI-powered anomaly detection catch data entry issues as they happen. This dramatically reduces the number of queries and the time required for manual data review, allowing database lock to happen faster and with higher confidence.
- Automation of Manual Tasks: Automating repetitive, low-value tasks like generating monitoring reports, tracking site payments, and managing document workflows saves thousands of hours of manual labor per study. This allows your highly skilled team to focus on science and strategy, not administration. As healthcare SaaS market research shows, faster time-to-market for new therapies benefits patients who need access to innovative treatments—and that’s the ROI that matters most.
End Data Silos: Choose U.S. Platforms That Unite EDC, CTMS, and eTMF
The United States is the epicenter of the clinical trial software revolution. As the world’s largest market for these solutions, U.S. providers benefit from proximity to major pharmaceutical companies, the highest concentration of R&D investment, and a demanding regulatory environment overseen by the FDA. This creates a hothouse for innovation, where the best providers are constantly redefining eClinical technology to meet the demands of increasingly complex and global trials.
One of the most significant shifts is the move away from fragmented “point solutions” toward integrated platforms that unify multiple functions. In the past, a sponsor might use separate vendors for EDC, CTMS, and eTMF, leading to data silos, manual reconciliation, and a disjointed view of the trial. True efficiency comes from seamless data flow across all aspects of a trial. When your EDC, CTMS, and eTMF are interconnected on a single platform, you eliminate redundant data entry, reduce human error, and gain a holistic, real-time view of your trial data.
Another defining trend is the explosive growth of decentralized clinical trials (DCTs), which grew over 380% from 2010 to 2022. This is a fundamental reimagining of how we bring research to patients, moving away from a purely site-centric model. Leading U.S. SaaS providers are at the forefront of this shift, building robust capabilities for remote monitoring, telehealth, eConsent, and wearable device integration, making trials more accessible, patient-centric, and efficient.

Types of Leading SaaS Solutions
The clinical research software landscape is diverse, with solutions tailored to different organizational needs, trial complexities, and budgets.
- Enterprise-Grade Unified Platforms: These are the comprehensive, end-to-end solutions for large pharmaceutical companies and CROs managing complex, multinational trial portfolios. They integrate everything—EDC, CTMS, eTMF, RTSM, ePRO, safety, and analytics—under a single umbrella for unprecedented control and visibility.
- Integrated EDC-Centric Suites: These platforms are built around a robust Electronic Data Capture core, with other modules like a lightweight CTMS and eTMF tightly integrated. This approach recognizes that high-quality data collection is the heart of any trial and is often a perfect fit for small to mid-sized biotechs.
- Site-Focused Management Systems: Designed specifically for research sites, hospitals, and academic medical centers, these solutions excel at participant recruitment, visit scheduling, financial management, and site-level regulatory document management. They prioritize ease-of-use for busy site staff and often integrate with institutional EHR systems.
- Specialized Niche Solutions: These providers focus on one function and do it exceptionally well. Examples include advanced eConsent platforms with multimedia capabilities, dedicated Randomization and Trial Supply Management (RTSM) for complex adaptive trials, or AI-powered patient recruitment tools that mine real-world data.
Key Features: What to Demand from Clinical Research SaaS Technology
When evaluating platforms, focus on the core modules that drive efficiency, ensure data integrity, and maintain compliance.
- Clinical Trial Management System (CTMS): The operational backbone of a trial. A modern CTMS goes beyond simple tracking. Look for features like automated milestone tracking, site selection and activation workflows, integrated monitoring visit report management, automated site payment calculations and processing, and comprehensive subject tracking from screening through study completion.
- Electronic Data Capture (EDC): The interface for collecting patient data. Modern EDCs feature intuitive, drag-and-drop electronic case report form (eCRF) builders, real-time data validation and edit checks to prevent errors, streamlined query management workflows, and integrated medical coding with dictionaries like MedDRA and WHODrug.
- Electronic Trial Master File (eTMF): A digital repository for all essential trial documents. A leading eTMF provides more than just storage; it offers TMF Health metrics (completeness, timeliness, quality), version control, secure audit trails, and workflow automation to ensure you are always inspection-ready. It should also integrate with a site portal to streamline document submission from investigators.
- Randomization and Trial Supply Management (RTSM): Manages patient randomization and the investigational product supply chain. Essential for blinded studies, an RTSM must handle complex randomization schemes (e.g., stratified, dynamic), manage drug supply across global sites to prevent stockouts, and support emergency unblinding procedures with a full audit trail.
- Electronic Patient-Reported Outcomes (ePRO/eCOA): Allows patients to report outcomes directly via apps or web portals. This improves data quality by capturing information in real-time (reducing recall bias), increases compliance compared to paper diaries, and lessens the burden on patients. This includes everything from simple symptom diaries to validated quality-of-life questionnaires.
A truly comprehensive suite also offers strong integration capabilities through Application Programming Interfaces (APIs). In today’s increasingly complex and decentralized trial landscape, platforms that can securely integrate data from multiple sources—EHRs, central labs (LIMS), medical imaging systems (PACS), and patient wearables—while maintaining compliance are essential for success.
Fail Part 11, Fail the Trial: The Compliance and Security Checklist You Need
In clinical research, compliance and security are not features; they are the foundation upon which everything is built. We are dealing with sensitive patient data and the scientific evidence that could determine the approval of a new therapy. When evaluating SaaS providers, these criteria must be at the very top of your list, as a failure in this domain can invalidate an entire trial.
In the USA, the regulatory framework centers on several key pillars. Any electronic system used to collect, manage, or submit clinical data must comply with the FDA’s 21 CFR Part 11 for electronic records and electronic signatures. Good Clinical Practice (GCP) guidelines, specifically the ICH E6(R2) addendum, set the international ethical and scientific quality standards for trial conduct. And because we handle sensitive patient health data, strict adherence to the Health Insurance Portability and Accountability Act (HIPAA) is the absolute baseline for any operations in the U.S.
The best providers don’t just check a box for compliance; they build it into their platform’s DNA. Compliance should be an automated, inherent function of the system, not an afterthought that requires manual workarounds.

Ensuring 21 CFR Part 11 Compliance
21 CFR Part 11 defines the criteria under which electronic records and signatures are considered as trustworthy, reliable, and legally equivalent to paper records. In practice, this requires a specific set of technical and procedural controls:
- Secure Audit Trails: The system must generate a detailed, unalterable, computer-generated log that records every action related to data creation, modification, or deletion. This audit trail must capture the who (user ID), what (the change made), when (a date/time stamp), and why (the reason for the change, if applicable). This is your primary evidence for regulatory inspectors.
- Electronic Records and Signatures: Every electronic signature must be uniquely linked to its signer and permanently bound to the record. The system must automatically capture the printed name of the signer, the date and time of the signature, and the meaning of the signature (e.g., “approval,” “review,” or “authorship”).
- Role-Based Access Controls: The system must enforce granular permissions automatically, ensuring that only authorized personnel can access sensitive data or perform critical functions like data modification or query closure. Users should only see and be able to do what their specific role requires.
- System Validation: The sponsor is ultimately responsible for ensuring the system is fit for purpose. However, leading SaaS vendors provide comprehensive validation documentation packages, including installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ) evidence, to significantly reduce the validation burden on your team.
Navigating HIPAA and Data Privacy
Beyond FDA regulations, protecting patient privacy is paramount. HIPAA establishes national standards for the protection of Protected Health Information (PHI).
- Key HIPAA Rules: Providers must demonstrate compliance with the Privacy Rule (governing the use and disclosure of PHI), the Security Rule (defining safeguards to protect electronic PHI), and the Breach Notification Rule (requiring notification to individuals and HHS in the event of a data breach).
- Business Associate Agreements (BAA): Before you can entrust a SaaS vendor with PHI, you must have a signed BAA in place. This is a legal contract that obligates the vendor to maintain the same level of HIPAA compliance as your own organization.
Security Certifications and Global Compliance
Your data must be protected from ever-evolving cyber threats. The best providers stay ahead of threats through rigorous, independently audited security practices.
- ISO 27001: This is the international standard for information security management. Certification demonstrates a provider’s commitment to a comprehensive security program, including risk assessment, threat mitigation, and continuous improvement.
- SOC 2: A SOC 2 Type 2 report is an independent audit that assesses how effectively a provider’s controls for security, availability, processing integrity, confidentiality, and privacy operate over an extended period. Demand to see this report.
- Data Encryption: Data must be encrypted both in transit (as it moves across networks) and at rest (while it is stored on servers) using strong, modern cryptographic standards to prevent unauthorized access even in the event of a physical breach.
- Navigating Global Compliance: For trials with sites outside the U.S., compliance becomes more complex. Your SaaS provider must have a strategy for regulations like Europe’s General Data Protection Regulation (GDPR), which imposes strict rules on data processing and cross-border data transfers. Ask potential vendors how they handle data residency requirements and their legal basis for transferring data internationally.
At Lifebit, our federated AI platform embeds these security and compliance principles into every layer. Our Trusted Research Environment (TRE) enables secure, compliant collaboration across global datasets, ensuring sensitive data remains protected within its local jurisdiction while being accessible for powerful, real-time insights.
Decentralize Now: AI Moves That Turn Months of Cleaning into Weeks
The clinical research landscape is racing forward, powered by decentralized trials and the integration of artificial intelligence.
Decentralization is a fundamental shift. The volume of decentralized trials grew over 380% from 2010 to 2022. Instead of requiring patients to travel to sites, we’re bringing trials to them through remote monitoring, telehealth, and direct-to-patient services. Modern SaaS platforms are the backbone of this revolution, providing patient-facing apps, integrating with wearables and sensors, and facilitating secure telehealth consultations. This shift makes trials more accessible, accelerates enrollment, and improves retention by meeting patients where they are.
The Impact of AI and Predictive Analytics
AI and predictive analytics are evolving clinical research from reactive data management to proactive, intelligent decision-making.
- Predicting Patient Dropout: AI algorithms can now predict with 85% accuracy which patients are at risk of leaving a study, allowing for targeted retention strategies.
- Optimizing Site Selection: AI can identify high-performing sites by analyzing vast datasets on past enrollment, data quality, and operational efficiency, helping allocate resources more strategically.
- Automating Data Cleaning: Machine learning algorithms detect anomalies and inconsistencies that human reviewers might miss, shortening data cleaning timelines from months to weeks.
- Accelerating Patient Recruitment: Natural language processing helps researchers find the right patients faster by translating complex criteria into sophisticated database queries across multiple data sources, including Real-World Evidence (RWE).
The integration of Real-World Evidence from electronic health records, claims databases, and patient registries provides richer context for trial results, supporting better trial design and more robust regulatory submissions.
At Lifebit, we’re pushing this frontier with our federated AI platform. Instead of moving sensitive patient data, our technology brings the analytics to the data. This allows researchers to run sophisticated AI models across distributed datasets in real time, generating insights while keeping patient information secure within its original environment. It’s the kind of secure, intelligent collaboration that the future of clinical research demands.
Pick the Right SaaS, Save Months: Your Buyer’s Checklist
Choosing a clinical research SaaS provider is a critical partnership that will shape your trial efficiency, data quality, and compliance posture for years to come. The right choice is not a one-size-fits-all solution; it depends on your organization’s size, complexity, therapeutic focus, and long-term goals.
- Large Pharmaceutical Companies & CROs need comprehensive, integrated suites (EDC, CTMS, eTMF, RTSM, Safety) with proven scalability for large, global trials. Key requirements include robust APIs for integration with enterprise systems, advanced analytics and reporting capabilities, support for complex study designs, and 24/7 global support.
- Biotech Startups & Mid-Sized Pharma require user-friendly, core EDC and CTMS functionality that can be deployed quickly and can scale as the company grows. Flexible, study-based pricing models are often preferred over large enterprise commitments. Strong CRO partnership support and responsive, high-touch customer service are critical.
- Academic Research Organizations (AROs) & Research Sites prioritize budget-friendly, easy-to-use solutions with intuitive interfaces that minimize the training burden on busy site staff and researchers. Integration with university EHR systems and grant management software is a major plus.
Selection Criteria: How to Identify the Best Clinical Research SaaS Technology Provider
Beyond organizational fit, several universal criteria separate the best providers from the rest. Use these points to build your evaluation scorecard and ask pointed questions during vendor demos.
- Integration Capabilities: A platform’s value multiplies when it connects seamlessly with your existing ecosystem. Ask the vendor: “Can you demonstrate your API library? What pre-built connectors do you have for common systems like EHRs, LIMS, and safety databases? What is the process and cost for a custom integration?”
- Scalability and Global Support: The platform must grow with you, from a single-site Phase I study to a multi-thousand-patient global Phase III trial. Ask the vendor: “What is the largest study you have supported in terms of patients, sites, and data volume? Can you provide performance metrics under load? What are your support hours across different time zones?”
- User Experience (UX): An intuitive, role-based interface is critical for adoption by sites, monitors, and data managers. A platform that is too complicated will lead to user frustration, data entry errors, and low adoption, wasting the investment. Ask the vendor: “Can we have access to a sandbox environment to test the interface for our key roles? What is the average training time required for a site coordinator or CRA? Can you share user satisfaction scores or case studies?”
- Total Cost of Ownership (TCO): Look beyond the initial subscription price. Factor in all associated costs, including implementation and validation services, integration fees, training, support, and any potential charges for study amendments, data storage, or number of users. A platform with a higher subscription that accelerates timelines and reduces manual effort often delivers a far better ROI. Ask the vendor: “Please provide a detailed pricing sheet that breaks down all potential costs. Are there extra fees for building reports, API calls, or long-term data archival?”
- Regulatory and Industry Expertise: Your vendor is a key compliance partner. They must have deep, demonstrable expertise in 21 CFR Part 11, GCP, HIPAA, and evolving global regulations like GDPR. Ask the vendor: “Can you walk us through your validation package? How do you stay current with changing regulations? Can we speak with your Quality Assurance lead?”
- Service and Support Quality: When a critical issue arises mid-study, prompt, knowledgeable support is non-negotiable. Evaluate the availability (e.g., 24/7), guaranteed response times (SLAs), and expertise of the support team. Ask the vendor: “What are your support SLAs for critical issues? Can you provide references from current customers who can speak to their support experience? What professional services are available for complex implementations?”
- Financial Stability and Product Roadmap: You are entering a long-term partnership. Choose a financially stable company with a clear, innovative product roadmap. This demonstrates a commitment to future development and ensures your platform will evolve with industry trends like AI and decentralization. Ask the vendor: “Can you share your product roadmap for the next 18-24 months? How is your company funded, and can you share information on your financial stability?”
Structuring Your Evaluation: The RFP Process
For a thorough evaluation, a structured Request for Proposal (RFP) process is invaluable.
- Define Requirements: Assemble a cross-functional team (Clinical Ops, Data Management, IT, QA) to document your must-have and nice-to-have features.
- Create a Scorecard: Build a weighted scorecard based on the selection criteria above to objectively compare vendors.
- Shortlist Vendors: Send the RFP to 4-6 potential vendors and use their responses to shortlist the top 2-3 finalists.
- Conduct Scripted Demos: Have the finalists demonstrate how their system solves your specific use cases and pain points.
- Check References: Conduct thorough reference checks with companies similar to yours.
The clinical research landscape is shifting toward intelligent, federated platforms that enable secure collaboration. Platforms like Lifebit’s next-generation solution are pioneering real-time insights through federated AI analytics, showing what the future of clinical research technology looks like: secure, intelligent, and truly interconnected.
Act Now: The SaaS Stack That Gets Therapies to Patients Faster
The digital change of clinical research is no longer optionalit’s the foundation for future success. Cloud-based SaaS platforms consistently outperform legacy systems, delivering 30-70% reductions in TCO, 40% faster study startups, and dramatically cleaner data.
The market is consolidating around integrated eClinical suites that unify CTMS, EDC, and eTMF into single, powerful platforms. These systems are enabling new research models, particularly decentralized trials, which grew over 380% in the last decade.
Technology without trust is worthless. The best providers build their platforms on a bedrock of compliance (21 CFR Part 11, GCP, HIPAA) and security (ISO 27001, SOC 2), ensuring research integrity and patient safety.
The future is already here. It’s intelligent, with AI predicting patient dropout with 85% accuracy. It’s decentralized, bringing trials to patients. And it’s data-driven, with platforms that generate insights to accelerate decision-making.
Choosing a partner isn’t just about features; it’s about finding a partner who shares your vision. The next generation of clinical research requires federated governance, advanced AI analytics, and the ability to harmonize data across global sources without compromising security.
Platforms enabling secure, federated AI analytics, like Lifebit’s next-generation solution, are paving the way for real-time insights and faster drug development. The clinical trials of tomorrow are being designed today. The question is: are you equipped with the right technology partner to power them?
Learn how Lifebit’s federated platform powers next-generation clinical research

