Why Your Research Needs Better Trial Technology Now

Why Trial Technology Is the Difference Between a Drug That Saves Lives and One That Never Ships
Trial technology — the tools, platforms, and systems used to design, run, and present clinical or legal trials — is no longer optional. It is the deciding factor in whether a drug reaches patients or dies in development. In the modern era, the “trial tech stack” has evolved from simple electronic databases to complex, interconnected ecosystems that leverage artificial intelligence, federated data structures, and real-world evidence.
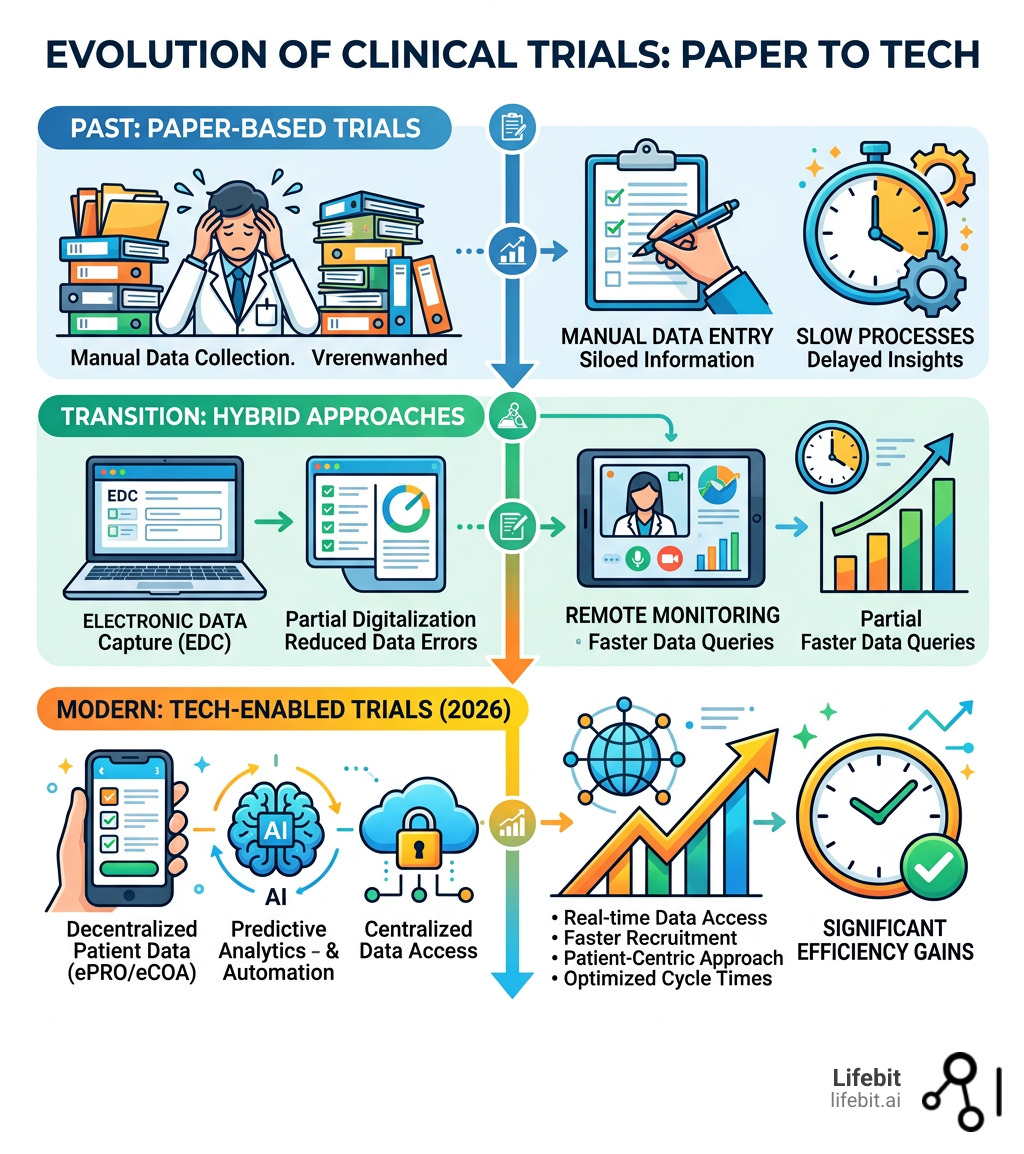
Historically, the pharmaceutical industry was slow to adopt digital transformation. For decades, the gold standard was paper-based Case Report Forms (CRFs) and physical site visits. However, as the complexity of protocols has increased—often requiring multi-omic data, longitudinal tracking, and global patient cohorts—the old methods have become a liability. Today, trial technology is the infrastructure upon which the entire life sciences industry rests. Without it, the sheer volume of data generated by a single Phase III trial would be impossible to manage, let alone analyze for regulatory submission.
Here is a quick breakdown of the most important trial technology categories:
| Category | What It Does | Key Benefit |
|---|---|---|
| Decentralized Trial Platforms | Remote recruitment, data capture, and monitoring | 21x faster recruitment |
| Electronic Data Capture (EDC) | Replaces paper-based data collection | Fewer errors, faster builds |
| Clinical Metadata Repositories (CMDR) | Centralizes and standardizes trial metadata | CDISC compliance, reusable assets |
| AI and Predictive Analytics | Automates protocol authoring and enrollment modeling | Shorter cycle times, lower cost |
| Courtroom Trial Technology | 3D animations, drones, electronic exhibit databases | Better juror comprehension |
| Federated Data Platforms | Secure, in-situ analysis of siloed EHR and genomics data | No data movement, full compliance |
The numbers paint a stark picture. Clinical trials can take 10 to 15 years to complete — and even then, only about 12% of drugs that enter trials ever receive FDA approval. Development costs now average $2.6 billion per successful drug, and up to 50% of trials fail to meet their enrollment targets. These failures are rarely due to the underlying science of the molecule; more often, they are the result of operational inefficiencies that could have been mitigated with the right technology.
That is not a pipeline problem. It is a technology problem.
Manual processes, disconnected systems, and poor data standardization are quietly killing trials before they have a chance. The COVID-19 pandemic made this impossible to ignore — 80% of trials were disrupted, forcing the industry to adopt remote and digital tools almost overnight. Many of those tools never went away. They became the new baseline, shifting the industry from “electronic” (simply digitizing paper) to “digital” (reimagining the process through technology).
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, and I have spent over 15 years at the intersection of computational biology, AI, and federated data infrastructure — building the kind of trial technology that lets researchers access and analyze global patient data without ever moving it. In this guide, I’ll walk you through the trial technology landscape so you can make smarter decisions about the tools your research depends on.
Trial technology terms at a glance:
- AI-Powered Clinical Trials: Real-World Examples Transforming Research in 2025
- Clinical research AI
- Advanced analytics healthcare
Overcoming the $2.6 Billion Failure: Challenges in Traditional Processes
Traditional clinical trials are buckling under their own weight. According to Cancer Research UK, trials often take 10 to 15 years to complete, a timeline that is simply too slow for patients waiting for life-saving treatments. When we look at the clinical trial complete guide, the bottleneck is almost always manual. The financial implications are staggering: every day a drug is delayed from reaching the market represents between $600,000 and $8 million in lost opportunity cost for the sponsor.

For decades, the industry relied on manual data entry, physical binders, and siloed spreadsheets. This “old way” creates three massive points of failure that modern trial technology is designed to solve:
Enrollment Deadlocks: Up to 50% of trials fail to meet their recruitment targets, and 80% of trials fail to finish on time due to enrollment issues. Without the right trial technology, finding eligible participants is like looking for a needle in a haystack. Traditional methods rely on site-specific databases, which are often outdated or limited in scope. Modern technology allows for “omnichannel” recruitment, tapping into social media, Electronic Health Records (EHR), and patient advocacy networks simultaneously.
Data Fragmentation and Silos: When data is trapped in Excel files or legacy systems, there is no “single source of truth.” This leads to errors that can disqualify an entire study during regulatory review. In a typical trial, data comes from multiple sources: labs, imaging centers, wearable devices, and site visits. Without an integrated technology platform, reconciling this data takes months of manual effort, increasing the risk of transcription errors and data loss.
Protocol Complexity and Site Burden: Modern protocols are 70% more complex than they were a decade ago, requiring more procedures and more data points per patient. This places an immense burden on clinical sites. If the trial technology is not user-friendly, site staff spend more time fighting with software than caring for patients, leading to high site turnover and poor data quality.
| Feature | Manual Trial Workflow | Tech-Integrated Workflow |
|---|---|---|
| Data Entry | Paper-based / Manual Excel | Automated EDC / Wearables |
| Recruitment | Site-specific / Local | Omnichannel / Global |
| Monitoring | On-site visits | Remote / Real-time |
| Compliance | Post-hoc cleanup | Built-in CDISC standards |
| Data Visibility | Delayed (weeks/months) | Instant / Real-time Dashboards |
By the time a drug reaches the finish line, the investment is astronomical. We must move away from these fragile, manual processes if we want to lower that $2.6 billion barrier and ensure that scientific breakthroughs aren’t lost in a sea of administrative red tape.
Why Decentralized Trial Technology is the New Industry Standard
If the traditional model is broken, Decentralized Clinical Trials (DCTs) are the fix. By moving the trial to the patient rather than forcing the patient to the site, we solve the recruitment crisis and improve the overall quality of the data collected. DCTs leverage a suite of trial technology including eCOA (Electronic Clinical Outcome Assessment), ePRO (Electronic Patient-Reported Outcomes), and wearable sensors to capture data in the patient’s natural environment.
The results are hard to ignore:
- 21x Faster Recruitment: Using unified platforms allows researchers to reach patients across 95+ countries simultaneously. By removing geographical barriers, a trial for a rare disease can find participants globally rather than being limited to the radius of a few specialized clinics.
- 28% Greater Retention: When patients can participate via mobile apps and wearables from their own living rooms, they don’t drop out. The “patient burden” is the number one reason for attrition; technology minimizes this by reducing the need for travel and time off work.
- 3x More Diversity: Technology enables us to reach underrepresented populations who may not live near a major academic research center. This is critical for ensuring that new treatments are safe and effective for all demographic groups, a requirement that regulatory bodies like the FDA are increasingly enforcing.
Following decentralized clinical trials guidance is no longer just a “nice to have”—it is a competitive necessity. Platforms that offer Unified Technology allow for a seamless journey for both the researcher and the participant, ensuring that data flows securely and accurately from a wearable device directly into the study database without manual intervention.
The Role of Wearables and IoT in Modern Trials
One of the most significant shifts in trial technology is the use of medical-grade wearables. These devices provide a continuous stream of data—heart rate, sleep patterns, glucose levels, and physical activity—rather than the “snapshot” data obtained during a monthly clinic visit. This continuous monitoring allows for much more sensitive detection of drug efficacy and safety signals. For example, a wearable might detect a subtle cardiac arrhythmia that occurs only at night, which would be missed during a standard daytime ECG at a doctor’s office.
Maximizing ROI with Hybrid Trial Technology
Not every trial can be 100% remote. Complex procedures like MRIs or biopsies still require physical facilities. That’s where hybrid trial technology shines. By combining on-site visits with remote monitoring, sponsors can achieve a seven-fold increase in ROI, particularly in Phase II and III trials. This model reduces site overhead and allows for clinical trial technology trends 2025 like “site-less” monitoring, where data managers oversee hundreds of participants through real-time dashboards rather than traveling to physical clinics. This hybrid approach offers the flexibility needed to handle complex therapeutic areas like oncology while still reaping the efficiency gains of digital tools.
Streamlining Data with CMDRs and Metadata Standardization
One of the biggest “silent killers” of clinical research is the lack of data standards. According to the US Congressional Budget Office, drug development costs average $2.6 billion, and a huge portion of that is wasted on re-formatting data for regulatory submissions. When data is collected in different formats across different sites, the “data cleaning” phase at the end of a trial can take six months or longer.
This is where Clinical Metadata Repositories (CMDRs) come in. A CMDR acts as a central library for all your trial “ingredients”—your Case Report Forms (CRFs), data variables, and edit checks. By enforcing CDISC compliance (Clinical Data Interchange Standards Consortium) from day one, you ensure that your data is submission-ready the moment the trial ends.
We are celebrating the role of data in clinical trials because, with a CMDR, you can:
- Reuse approved metadata: Don’t build every study from scratch. Use a library of pre-validated templates that have already passed regulatory scrutiny. This can reduce study setup time by up to 60%.
- Automate study builds: One team reported building three unique studies in just three months—from protocol to go-live—using standardized metadata. This speed is impossible with manual, siloed processes.
- Ensure regulatory alignment: FDA and PMDA both require CDISC standards (specifically SDTM and ADaM formats). Building them into your tech stack from the start eliminates the “data mapping” nightmare that usually adds months to a timeline.
The Technical Architecture of a CMDR
A modern CMDR is more than just a database; it is a governance tool. It allows organizations to maintain a “Single Source of Truth” for metadata across the entire enterprise. When a change is made to a standard variable (e.g., how “blood pressure” is recorded), that change can be propagated across all ongoing and future trials with a single click. This level of control is essential for maintaining data integrity in large-scale, global programs where hundreds of researchers are interacting with the data.
Automating Workflows via Trial Technology Integration
The real magic happens when your CMDR integrates with your Electronic Data Capture (EDC) system. This integration allows for “auto-build” capabilities, where the EDC forms are automatically generated based on the approved metadata in the CMDR.
This creates a loop of current systems and technology in clinical trials that provides real-time insights. Instead of waiting weeks for a data manager to spot an outlier, the system flags it instantly based on pre-defined edit checks. This seamless collaboration between technology and operations is what allows modern biopharma to move at the speed of light, reducing the time from “First Patient In” to “Last Patient Out.”
Beyond the Lab: Modern Courtroom Trial Technology and Presentation
While we often focus on the clinic, trial technology is also revolutionizing the legal side of research and litigation. In a courtroom, where a juror’s understanding determines the outcome, how you present your data is just as important as the data itself.
Modern legal teams are now using:
- 3D Animations and Simulations: These are used to explain complex biological mechanisms or “reconstruct” events. While an animation is demonstrative, a simulation is substantive evidence based on scientific principles.
- Drones and Thermal Imaging: Drones provide an intermediate perspective for site surveillance (like a construction defect site), while thermal imaging can detect environmental leaks or product defects.
- Electronic Exhibit Databases: Gone are the days of fumbling through paper binders. Software like OnCue allows for “hot-seating,” where a technician pulls up any document or deposition clip instantly.
Using Trial Technology – Jury Consultant services ensures that the story remains the focus. Technology should never overshadow the narrative; it should make the narrative undeniable. Whether it’s a medical malpractice case or an intellectual property dispute over a new drug, visual storytelling through trial technology increases juror retention and comprehension significantly.
AI and Predictive Analytics: The Future of Protocol Authoring
The next frontier of trial technology is Generative AI and Machine Learning. We are moving toward a world where AI doesn’t just analyze data—it helps design and write the trial itself, significantly reducing the risk of human error in the planning stages.
- Automated Protocol Authoring: AI can scan thousands of previous trials to suggest the most efficient inclusion/exclusion criteria. By identifying criteria that have historically led to recruitment failures in similar drugs, AI helps researchers avoid costly protocol amendments—which currently cost an average of $450,000 each.
- Synthetic Control Arms: This is perhaps the most disruptive application of AI in trial technology. By using historical trial data and real-world evidence (RWE) to create “virtual” control groups, researchers can reduce or even eliminate the need for a placebo group. This is a game-changer for orphan drug development and rare diseases where the patient population is too small to split into two groups.
- Predictive Enrollment Modeling: Machine learning algorithms can analyze demographic data, local disease prevalence, and historical site performance to predict which geographic sites will actually deliver patients. This prevents the “50% failure rate” we see in traditional recruitment by focusing resources on high-potential areas.
- Generative AI for Regulatory Submissions: New tools are emerging that can draft Clinical Study Reports (CSRs) by pulling data directly from the EDC and CMDR. This reduces the writing time from months to weeks, ensuring that the data is presented in a format that is optimized for regulatory reviewers.
Our ai clinical trials ultimate guide explores how these tools are being used right now. For example, ai-powered clinical trials real-world examples transforming research in 2025 show how predictive analytics can slash months off a recruitment timeline by identifying high-potential patient cohorts in real-time. As these technologies mature, we expect to see “in-silico” trials, where the initial safety and efficacy of a drug are tested on digital twins before a single human patient is ever enrolled.
Frequently Asked Questions about Trial Technology
How did COVID-19 change the adoption of trial technology?
The pandemic was a “forced pilot” for the entire industry. With an 80% disruption rate to ongoing trials, sponsors had no choice but to adopt remote monitoring and decentralized tools. This shift proved that these technologies were not only viable but superior in terms of data quality and patient safety. Today, returning to 100% manual, site-based processes is seen as a major operational risk and a step backward in patient-centricity.
What role does metadata standardization play in regulatory approval?
It is the foundation of a successful submission. Regulatory bodies like the FDA and PMDA require data in specific formats (CDISC). If you don’t use trial technology to standardize your metadata from the beginning, you will spend millions of dollars and several months “fixing” your data at the end of the trial. This delays your submission and potential market entry, costing lives and revenue.
How do 3D animations improve courtroom outcomes?
Humans are visual learners; we retain 65% of information presented visually compared to only 10% of information presented orally. In complex litigation involving medical or technical data, a 3D animation acts as a “bridge” between expert testimony and juror understanding. It creates a mental reference point that jurors can take with them into deliberations, making your case much more memorable and persuasive.
Is trial technology compliant with data privacy laws like GDPR?
Yes, modern trial technology is built with “privacy by design.” Platforms like Lifebit use federated data architectures, which allow researchers to analyze data without ever moving it from its original secure location. This ensures that patient-level data remains protected and compliant with local regulations like GDPR in Europe or HIPAA in the United States, while still allowing for global collaboration.
Can small biotech firms afford advanced trial technology?
While the initial investment can seem high, the cost of not using technology is much higher. Many modern platforms offer scalable, cloud-based solutions that allow smaller firms to pay only for what they use. By reducing the need for large internal data management teams and shortening the time to market, the technology often pays for itself within the first phase of a trial.
Conclusion: Maximizing Research Impact with Lifebit
The era of slow, manual, and disconnected trials is over. To succeed in today’s landscape, you need trial technology that doesn’t just store data but actively works to accelerate your insights.
At Lifebit, we provide a next-generation federated AI platform that allows you to access global biomedical and multi-omic data securely. Our Trusted Research Environment (TRE) and Real-time Evidence & Analytics Layer (R.E.A.L.) empower your team to collaborate across hybrid data ecosystems without the risks of data movement. Whether you are running a large-scale clinical trial or conducting pharmacovigilance, our platform ensures your research is compliant, scalable, and—most importantly—fast.
Ready to transform your research? Explore the Lifebit platform today.

