Why Diversity in Clinical Trials is Essential for Modern Medicine

The High Cost of Ignoring Diversity in Clinical Trials
Diversity in clinical trials is one of the most critical — and most neglected — factors in modern drug development. When the people tested in a trial don’t reflect the people who will actually use the treatment, the results can be incomplete, misleading, or even dangerous.
Here is a quick snapshot of where things stand today:
| Group | U.S. Population Share | Clinical Trial Representation |
|---|---|---|
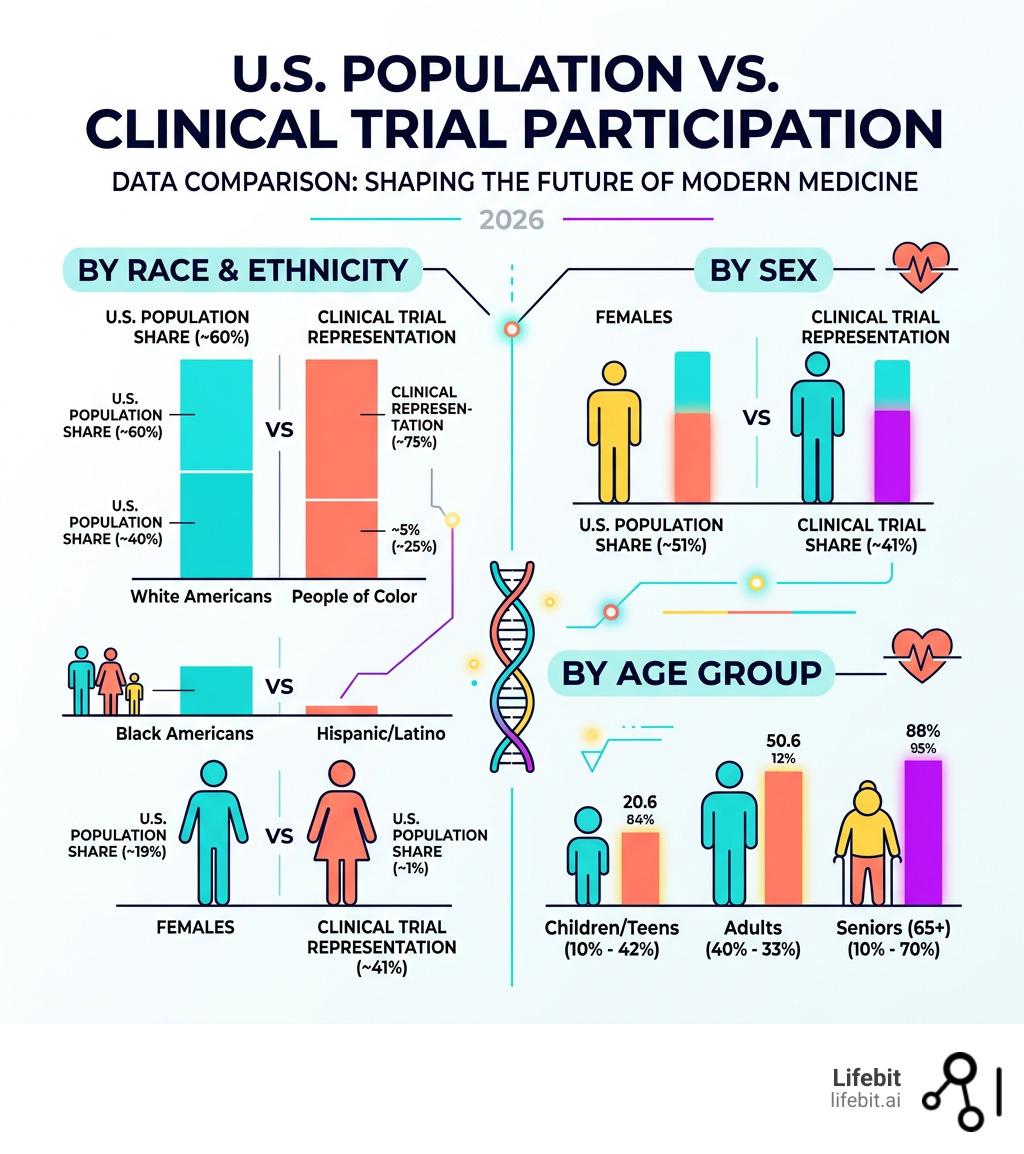
| White Americans | ~60% | 75% of participants |
| People of Color | ~40% | ~25% of participants |
| Black Americans | ~13% | ~5% of participants |
| Hispanic/Latino | ~19% | ~1% of participants |
| Females | ~51% | ~41% of participants |
These gaps are not just statistics. They have real consequences for drug safety, treatment efficacy, and health equity across entire populations.
Consider this: in 2020, 75% of the roughly 32,000 participants in trials for 53 FDA-approved drugs were White — even though racial and ethnic minorities make up nearly 40% of the U.S. population. At the same time, Black individuals account for 12.4% of pancreatic cancer diagnoses but only 8.2% of pancreatic cancer trial participants. The science is only as good as the people it includes.
The problem runs deep. It is rooted in historical trauma, logistical barriers, regulatory gaps, and a research system that has historically been designed around a narrow slice of humanity. Fixing it requires action at every level — from trial design and site selection to data infrastructure and community trust.
This guide walks through why the problem exists, what the regulations now require, and — most importantly — what actually works to build more inclusive trials.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, a federated genomics and biomedical data platform built to make global, diverse health data accessible for research at scale. My background in computational biology, bioinformatics, and precision medicine has shown me how the lack of diversity in clinical trials creates blind spots that no amount of computing power can fix if the underlying data is biased from the start. In the sections that follow, we will break down the barriers, the mandates, and the practical solutions that can finally move this field forward.
Diversity in clinical trials terms you need:
Why Diversity is the Foundation of Safe Medicine
Science tells us that people don’t all respond to medications in the exact same way. Biological variation, driven by genetics, age, sex, and even environmental factors, influences how our bodies metabolize drugs. If a clinical trial population is homogeneous, we risk missing critical safety signals or efficacy gaps that only appear in specific groups. This is not just a matter of social justice; it is a matter of fundamental biological accuracy.
The Science of Pharmacogenomics and Metabolism
Pharmacogenomics explores how an individual’s genetic makeup affects their response to drugs. For instance, variations in the cytochrome P450 (CYP450) enzyme system—responsible for metabolizing the majority of prescribed medications—can differ significantly across ethnic groups. A dosage that is therapeutic for a person of European descent might be toxic or completely ineffective for someone of East Asian or African descent due to these enzymatic differences. Without diverse representation, these metabolic nuances remain hidden until the drug is released to the general public, often leading to adverse drug reactions (ADRs) that could have been predicted.
For example, this study highlights the importance of understanding the disparity of minority representation to create an industry benchmark for diversity. Without this data, we are essentially guessing how a drug will perform in the real world. Variations by therapeutic area also play a huge role; while women may be well-represented in geriatric trials, they are often underrepresented in cardiovascular disease studies, despite heart disease being a leading cause of death for all genders.
Oncology and the Precision Medicine Gap
We’ve seen similar disparities in oncology. As mentioned, Black Americans are disproportionately affected by pancreatic cancer but are significantly less likely to be included in the trials testing new treatments for it. This lack of representation undermines the generalizability and quality of medical research. When we develop “targeted therapies” based on genomic markers found primarily in one demographic, we are effectively creating a tiered healthcare system where precision medicine only works for the majority. To dive deeper into how we can bridge these gaps, read more about breaking barriers in clinical trials.
The Scientific Risk of Homogeneous Data
When 75% of trial participants are White, as seen in the 2020 FDA drug snapshots, we develop a “one-size-fits-all” approach to medicine that actually fits very few. A lack of diversity means we might not catch group-specific side effects until a drug is already on the market. This creates a massive health equity gap where minority populations may be prescribed medications that haven’t been adequately tested for their specific biological profiles. This can lead to lower adherence rates, as patients from underrepresented groups may experience unexpected side effects that were never documented during the trial phase.
Gender Diversity and the Pregnancy Gap
Gender diversity has evolved, but significant gaps remain. While women represented an average of 51% of participants in FDA-approved drug trials between 2014 and 2021, they are still underrepresented in areas like cardiology and infectious diseases. This is particularly concerning given that women often experience different symptoms and physiological responses to cardiovascular events than men.
The most glaring omission, however, involves pregnant and lactating individuals. Historically excluded due to safety concerns post-thalidomide, they remain drastically underrepresented today. In fact, a study of six registries found that only 0.32% of active trials focused on pregnant individuals. This exclusion creates a “data desert” where clinicians are forced to make prescribing decisions for pregnant patients based on anecdotal evidence or data extrapolated from non-pregnant populations. The PRGLAC task force is currently working to address these research gaps, but the industry has a long way to go. You can learn more about Pfizer’s diversity initiatives to see how major players are beginning to tackle these historical exclusions.
Breaking Down the Barriers to diversity in clinical trials

Understanding why people don’t participate is the first step to fixing the problem. Barriers to diversity in clinical trials are often a mix of historical trauma, logistical hurdles, and systemic exclusion. For sponsors, the challenge is shifting from a passive recruitment model to one that actively meets patients where they are. You can find more insights on this in our guide to clinical trial recruitment strategies.
Overcoming Historical Mistrust and the Tuskegee Legacy
We cannot talk about diversity without acknowledging the deep-seated mistrust caused by historical atrocities. The Tuskegee Syphilis Experiment, where Black men were deceived and denied treatment for decades, led to 128 deaths and a legacy of fear. Similarly, the unauthorized use of Henrietta Lacks’ cells and the Puerto Rican Pill Trials — where women were given high-dose contraceptives without informed consent — have left lasting scars.
This mistrust is not just a relic of the past; it is reinforced by modern-day experiences of medical gaslighting and unequal treatment in the healthcare system. Rebuilding trust requires more than just a brochure; it requires long-term, transparent engagement with the communities that have been most harmed. Modern research must be built on the foundation of Good Clinical Practice (GCP) and rigorous IRB/IEC oversight to ensure ethical standards, but it must also involve community leaders in the oversight process itself.
Solving Logistical and Financial Participation Hurdles
For many, the “cost” of a clinical trial isn’t just medical — it’s the cost of taking time off work, finding childcare, and traveling to a distant site. These “hidden costs” disproportionately affect low-income individuals and those living in rural areas. If a trial requires ten visits to a metropolitan hospital three hours away, it is effectively excluding anyone without a flexible job or reliable transportation.
With 68 million people in the U.S. speaking a language other than English at home, language barriers are another massive hurdle. If the informed consent process is only available in English, or if the trial staff cannot communicate effectively with the participant, the trial is not truly inclusive.
To improve inclusivity, we must:
- Provide financial reimbursement: This should cover not just travel, but also lost wages and childcare expenses.
- Offer flexible visit windows: Implementing evening or weekend hours can accommodate working professionals.
- Decentralized trial options: Using local labs or mobile health clinics reduces the need for travel.
- Language Access: Ensure all materials are available in multiple languages and written in plain, culturally sensitive language.
- Geographic Expansion: Expand trial sites into rural areas and community health centers rather than relying solely on large academic medical centers.
The Digital Divide in Modern Recruitment
As trials move toward digital recruitment and electronic data collection, we must be wary of the “digital divide.” Not everyone has access to high-speed internet or the latest smartphone. If a trial requires a participant to use a complex app to report symptoms, it may inadvertently exclude older adults or those in lower socioeconomic brackets. Hybrid models that offer both digital and analog options are essential for maintaining a diverse participant pool.
Regulatory Mandates: Navigating FDORA and FDA Action Plans
The days of “voluntary” diversity goals are coming to an end. With the passage of the Food and Drug Omnibus Reform Act (FDORA) in 2023, the FDA now has the authority to mandate diversity in clinical trials. This is a game-changer for health equity, shifting the responsibility from the patient to the sponsor.
Implementing Mandatory Diversity Action Plans (DAPs)
Trial sponsors are now required to submit Diversity Action Plans (DAPs) for Phase 3 drug trials and pivotal device studies. These plans are not mere suggestions; they are formal requirements that must be submitted early in the development process. These plans must specify:
- Enrollment Goals: Specific targets disaggregated by race, ethnicity, sex, and age group.
- Rationale: A scientific justification for these goals, based on the epidemiology of the disease being studied.
- Implementation Strategy: A detailed explanation of exactly how the sponsor intends to monitor and achieve these goals, including site selection and community outreach efforts.
While waivers exist, they are granted only under very specific criteria, such as when a disease is so rare that demographic-specific recruitment is impossible. For a detailed breakdown, see the FDA Guidance on Diversity Action Plans.
How Regulatory Changes Impact Trial Design
These mandates are forcing a shift in how trials are designed from day one. Under FDASIA Section 907, there is an increased focus on the inclusion of demographic subgroups and the public availability of that data through tools like Drug Trials Snapshots. This transparency holds sponsors accountable to the public and to regulators.
We are seeing a move toward broader eligibility criteria. Historically, trials often used overly restrictive criteria that excluded people with common comorbidities—conditions like hypertension or diabetes that are more prevalent in certain minority populations. By broadening these criteria, sponsors can create a trial population that better reflects the real-world population likely to use the drug post-approval. Furthermore, the FDA now encourages the use of decentralized clinical trials (DCTs) as a primary method for meeting DAP goals, recognizing that bringing the trial to the patient is the most effective way to ensure diversity.
Global Implications of FDA Mandates
While FDORA is a U.S. law, its impact is global. Most major pharmaceutical companies operate internationally, and the FDA’s rigorous standards often set the tone for other regulatory bodies like the EMA (European Medicines Agency). We are beginning to see a harmonization of diversity requirements, as global researchers realize that diverse data is simply better science. This regulatory pressure is driving investment in research infrastructure in previously underserved regions, including Africa and Latin America, to meet these new global standards.
Proven Strategies to Improve Clinical Research Inclusivity
So, how do we actually move the needle? It starts with implementation science — using evidence-based strategies to integrate diversity into every stage of the research lifecycle. It is no longer enough to simply “hope” for a diverse cohort; sponsors must be intentional and data-driven. For more on the technological side of this, check out our guide on digital patient recruitment.
Practical Tools for diversity in clinical trials
Tools like the 3CTN EDI Framework and Toolkit provide a roadmap for sites to identify local barriers and implement inclusive best practices. This includes everything from improving access for Indigenous populations to ensuring that trial staff reflect the demographics of the community they are serving. Representation among the research staff is a powerful trust-builder; patients are more likely to participate when they see themselves reflected in the doctors and nurses conducting the study.
Building Sustainable Community Partnerships
Companies like Sanofi are leading the way by investing $18 million in Historically Black College and University (HBCU) medical schools to build long-term research capacity. This isn’t just about one trial; it’s about creating a sustainable ecosystem. Successful strategies include:
- Faith-Based Outreach: Partnering with churches and mosques to host health fairs and educational sessions about clinical research.
- Cultural Humility Training: Providing implicit bias training for all research staff to ensure that every potential participant is treated with respect and dignity.
- Bidirectional Feedback: Establishing community advisory boards where members have a say in trial design, including the review of consent forms and the scheduling of visits.
- Patient Navigators: Employing dedicated staff members whose sole job is to help participants navigate the complexities of the trial, from transportation to insurance paperwork.
The Role of Site Selection in Diversity
One of the most effective ways to increase diversity is to change where trials are conducted. Traditionally, trials have been clustered around a few elite academic institutions. However, these institutions often serve a demographic that does not reflect the broader population. By moving trials into community hospitals, retail pharmacies (like CVS or Walgreens), and local clinics, sponsors can tap into a much more diverse patient pool. Data-driven site selection tools now allow sponsors to analyze the demographic makeup of a 20-mile radius around a potential site, ensuring that they are placing their research in the heart of the communities they need to reach.
Incentivizing Diversity at the Site Level
Sponsors can also incentivize diversity by building it into their contracts with research sites. This might include providing additional funding for community outreach coordinators or offering bonuses for meeting specific diversity enrollment milestones. When the financial incentives of the site align with the diversity goals of the sponsor, the likelihood of success increases dramatically.
The Role of Data and Technology in Scaling Inclusivity
At Lifebit, we believe technology is the key to unlocking global diversity. When we can’t bring the patients to the data, we must bring the research to the patients through federated architecture. This allows us to access diverse, multi-omic data from across the globe without compromising security or privacy. Explore how these innovations in clinical trial recruitment are changing the landscape.
Unlocking Global Genomic Diversity
Currently, 78% of people included in genomic studies are of European ancestry. This creates a massive “genomic blind spot.” By expanding our reach into regions like the Middle East, Africa, and Latin America, we can identify genetic variants that are completely missed in standard reference genomes. For example, certain genetic markers for cardiovascular disease or drug metabolism may be prevalent in African populations but virtually non-existent in European ones. Access to this diverse real-world evidence is essential for the future of precision medicine, ensuring that the “targets” we identify for new drugs are relevant to the entire human population.
Federated Learning: A Privacy-First Approach
One of the biggest hurdles to global data sharing is data sovereignty and privacy laws (like GDPR). Federated architecture solves this by allowing researchers to analyze data where it resides—whether that’s in a hospital in Brazil or a genomic center in Nigeria—without the data ever leaving its home jurisdiction. This “data-to-code” model ensures that sensitive health information remains secure while still allowing for the large-scale, diverse analysis required for modern drug development.
Using Digital Tools to Reduce Participant Burden
Digital tools like electronic informed consent (eConsent), wearable health monitors, and mobile health professionals can significantly reduce the burden on participants.
- eConsent: Allows participants to review trial information at their own pace, in their own language, and discuss it with their families before signing.
- Wearables: Devices that track heart rate, sleep, or activity levels can replace many in-person clinic visits, making participation easier for those with busy schedules.
- Telehealth: Virtual check-ins allow investigators to monitor safety and answer questions without requiring the participant to travel.
By allowing for flexible visit windows and remote data collection, we make it possible for people who don’t live near a major academic medical center to participate in life-saving research. This “decentralized” approach is not just a convenience; it is a fundamental tool for equity.
AI and Bias Mitigation
Artificial Intelligence (AI) also plays a role in identifying and mitigating bias. AI algorithms can be used to scan electronic health records (EHRs) to identify potential participants who meet trial criteria but might have been overlooked by traditional recruitment methods. However, we must be careful: if the underlying EHR data is biased, the AI will be too. At Lifebit, we focus on ensuring that the datasets used to train these algorithms are as diverse as the populations they are meant to serve, creating a virtuous cycle of inclusive data and unbiased insights.
Frequently Asked Questions about Clinical Trial Diversity
Why are pregnant and lactating individuals often excluded from trials?
Historically, this group was excluded due to extreme caution following the thalidomide tragedy in the 1950s and 60s. However, this has led to a lack of data on how most drugs affect pregnant people. Today, the PRGLAC task force and the FDA are pushing for more inclusive protocols that balance safety with the need for evidence-based care during pregnancy.
What are the current statistics on minority underrepresentation in the U.S.?
While People of Color make up about 40% of the U.S. population, they represent only about 25% of clinical trial participants. Black Americans account for 13% of the population but only 5% of participants, while Hispanic and Latino individuals make up 19% of the population but only 1% of participants in many trials.
How do FDA Diversity Action Plans (DAPs) affect trial sponsors?
DAPs require sponsors to set and meet specific enrollment goals for underrepresented groups. Failure to submit a DAP or to provide an adequate rationale for enrollment goals can lead to delays in trial approval or the need for post-market studies to fill data gaps.
Conclusion: The Future of Inclusive Research
The industry is at a turning point. We are moving away from a time when diversity in clinical trials was an afterthought and toward a future where it is a regulatory and scientific requirement. By setting measurable goals and investing in community-led partnerships, we can finally ensure that “modern medicine” works for everyone, regardless of their race, gender, age, or zip code.
At Lifebit, we are proud to provide the federated data infrastructure that makes this inclusive future possible. By securely connecting researchers with diverse global data, we are helping to close the gaps in our understanding of human health.

