What Real World Study Actually Means

Why the Real-World Study Is Reshaping Modern Medicine
A real-world study is a type of research that examines how medical treatments, devices, or interventions actually perform in everyday clinical practice — not in the tightly controlled conditions of a laboratory trial. For decades, the medical community relied almost exclusively on the “ivory tower” of clinical research, where every variable was meticulously managed. However, as healthcare moves toward a value-based model, the need to understand how treatments perform in the “messy” reality of diverse patient populations has become paramount.
Here is a quick answer if that is all you need:
| Term | Definition |
|---|---|
| Real-World Study (RWS) | Research conducted in routine clinical settings, without strict trial controls |
| Real-World Data (RWD) | Health information collected during everyday patient care (EHRs, claims, registries) |
| Real-World Evidence (RWE) | Clinical conclusions drawn by analyzing RWD |
| Key difference from RCTs | RWS reflects broader, diverse patient populations — not just those who qualify for a controlled trial |
Clinical trials tell us what a drug can do under ideal conditions. Real-world studies tell us what it actually does for patients in the complex reality of everyday healthcare. This distinction is often referred to as the “efficacy-effectiveness gap.” Efficacy is the performance of an intervention under ideal circumstances, while effectiveness is its performance in real-world clinical practice.
That gap matters enormously. Most clinical treatment guidelines are built on randomized controlled trial (RCT) results — yet RCTs routinely exclude older patients, people with multiple conditions (comorbidities), and other high-risk groups. For example, a drug for heart failure might be tested on 50-year-olds with no other health issues, but in the real world, the average patient might be 75 with diabetes and kidney disease. A real-world study fills that blind spot, capturing outcomes across the full spectrum of patients who actually receive treatment.
The stakes are high. Regulatory bodies like the FDA and EMA are actively expanding their use of real-world evidence for drug approvals and post-market safety monitoring. This shift is driven by the realization that traditional trials are becoming increasingly expensive and time-consuming, sometimes taking over a decade and costing billions of dollars to bring a single drug to market. Meanwhile, biopharma companies report that 9 out of 10 RWE investments deliver measurable business outcomes — yet only 9% consider their programs truly best-in-class, largely due to data infrastructure challenges, such as data silos and lack of standardization.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, and over 15 years of work in computational biology, federated data platforms, and AI-driven biomedical research have given me a front-row seat to how real-world study programs succeed — and where they break down. In this guide, I will walk you through everything you need to understand about real-world studies: what they are, where the data comes from, what the evidence actually shows, and how to overcome the barriers that hold most programs back.
Real-world study further reading:
Defining the Real-World Study: Beyond the Controlled Lab

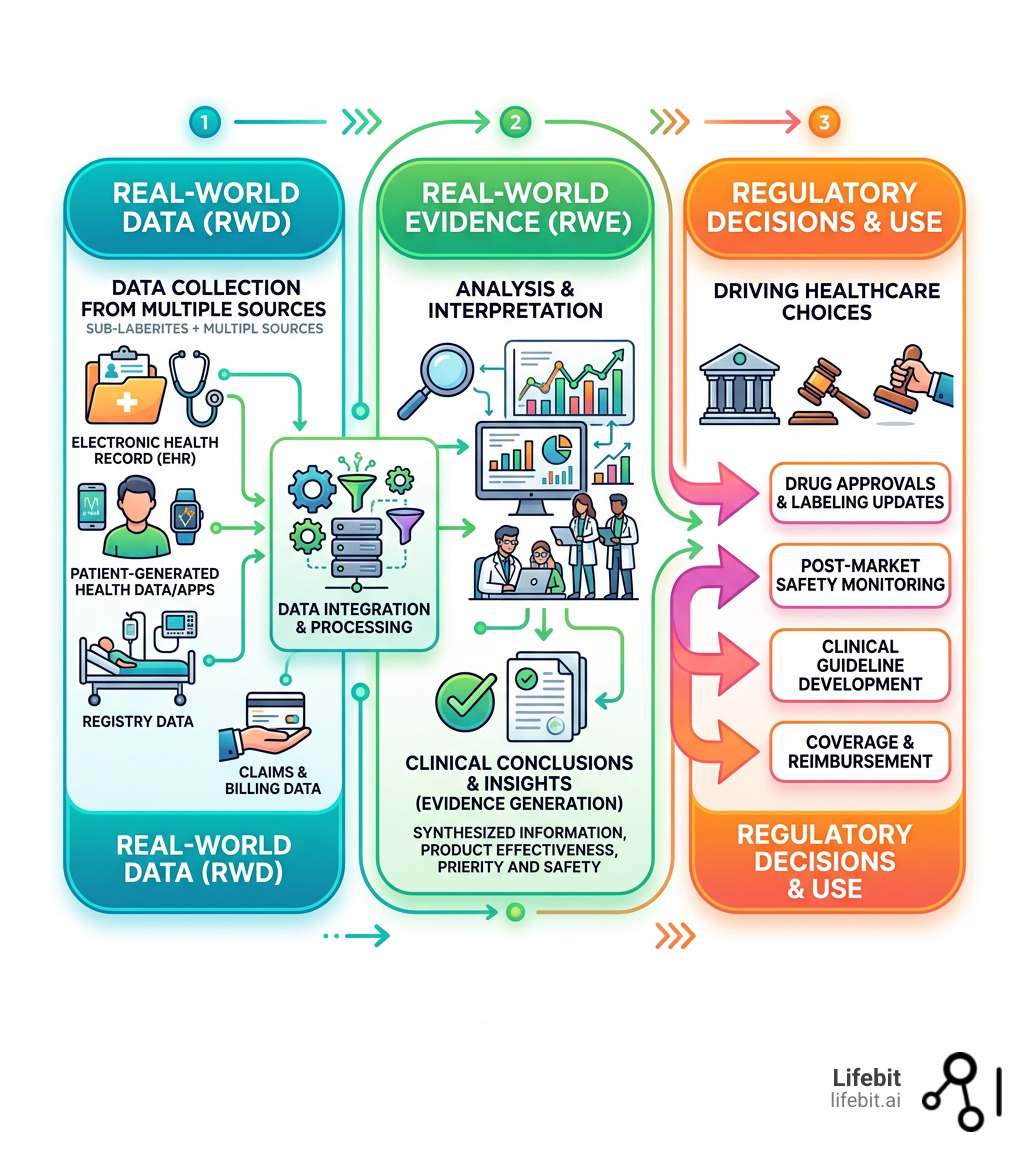
To understand a real-world study, we first have to distinguish between the ingredients and the finished product. At the heart of this field are two acronyms: RWD and RWE.
Real-world data (RWD) is the raw information relating to patient health status or the delivery of health care. It is collected routinely from various sources outside of a clinical trial. This includes everything from the notes a doctor types into a computer during a check-up to the records of which prescriptions were filled at a local pharmacy. It also encompasses patient-generated data from wearables, social media health forums, and even environmental data that might impact health outcomes.
Real-world evidence (RWE) is what we get when we apply rigorous analysis to that data. It is the clinical evidence regarding the usage and potential benefits or risks of a medical product. In short, RWD is the “what happened,” and RWE is the “what does it mean for future patients?” Generating RWE requires sophisticated statistical methods to account for the lack of randomization, ensuring that the conclusions drawn are scientifically valid.
A real-world study is typically observational in design. Unlike a traditional experiment where researchers decide exactly who gets which pill, an RWS looks at what doctors are already doing. It reflects the lived reality of patients and healthcare providers, capturing how treatments perform in the “wild.” This includes observing how patients actually take their medication — whether they skip doses, take it with food as directed, or stop taking it altogether due to side effects.
For a deeper dive into these terms, you can explore our real-world data definition or check out our guide on real-world data vs real-world evidence.
How a Real-World Study Differs from Traditional RCTs
The “Gold Standard” of medical research has long been the Randomized Controlled Trial (RCT). While RCTs are excellent for proving a drug works (efficacy), they often lack generalizability. This is because RCTs are designed to eliminate “noise” — the very variables that define real-world patients.
In an RCT, patients are hand-picked based on strict inclusion and exclusion criteria. They are often younger, have no other diseases, and take no other medications. In a real-world study, the population is “unselected.” We see patients with multiple comorbidities (like diabetes and heart disease), different ethnic backgrounds, and varying levels of adherence to their medication. This diversity is crucial for understanding how a drug interacts with other treatments and how it performs across different genetic profiles.
| Feature | Randomized Controlled Trial (RCT) | Real-World Study (RWS) |
|---|---|---|
| Setting | Highly controlled research environment | Routine clinical practice |
| Population | Homogeneous (strict criteria) | Heterogeneous (broad, diverse) |
| Objective | Efficacy (can it work?) | Effectiveness (does it work in real life?) |
| Randomization | Essential | Usually absent (observational) |
| Cost/Time | Extremely high and slow | Often lower and faster |
| Data Source | Case Report Forms (CRFs) | EHRs, Claims, Registries |
While most real-world studies are non-randomized, there are exceptions like pragmatic clinical trials (PCTs). PCTs introduce some elements of randomization into routine care settings, aiming to combine the scientific rigor of randomization with the real-world relevance of clinical practice. Research on RWE vs RCT differences highlights that these two methods are not rivals; they are partners that help us see the full picture of human health. RCTs establish the baseline possibility of benefit, while RWS establishes the actual utility in the population.
Key Advantages of the Real-World Study Approach
Why are we seeing such a massive shift toward these studies? It comes down to what traditional trials miss.
- Rare Event Detection: Because RCTs involve a limited number of people (often a few thousand) for a short time, they often miss rare side effects that might only occur in 1 in 10,000 patients. A real-world study can track hundreds of thousands of patients over years, making it much better at picking up “needle in a haystack” safety issues that only emerge after a drug is on the market.
- Long-term Safety & Persistence: Clinical trials are often short-lived. We can see if patients actually keep taking a drug for five or ten years in the real world, not just the twelve weeks required by a trial. This is vital for chronic conditions like hypertension or osteoporosis.
- High-Risk Groups: We finally get data on how treatments affect the elderly, pregnant women, or those with kidney impairment — groups often excluded from Phase III trials due to safety concerns or the desire to keep the study population simple.
- Natural Disease History: RWD helps us understand how a disease progresses when left untreated or managed with standard care. This provides a “historical control” that can be used to compare new treatments, especially in rare diseases where finding enough patients for a traditional control group is impossible.
- Cost-Efficiency: Utilizing existing data sources is often more resource-efficient than building a multi-million dollar trial from scratch. By leveraging data already being collected by hospitals and insurers, researchers can answer questions in months rather than years.
Learn more about the benefits of real-world data in clinical research to see how these advantages are being put into practice.
Primary Sources and Global Infrastructure for Evidence
Where does all this “real-world” information actually live? It isn’t in one giant folder. Instead, it’s scattered across a digital ecosystem, often in different formats and languages. Primary sources of RWD include:
- Electronic Health Records (EHR): The digital version of a patient’s paper chart. EHRs contain rich, longitudinal data including clinician notes, laboratory results, and imaging reports. However, much of this data is “unstructured,” requiring advanced AI and Natural Language Processing (NLP) to extract meaningful insights.
- Medical Claims & Billing Data: Records from insurers that show what procedures were performed and what drugs were prescribed. While less clinically detailed than EHRs, claims data is highly structured and provides a complete picture of a patient’s journey across different healthcare providers.
- Product and Disease Registries: Collections of data for patients with a specific condition (like a cancer registry) or those who have received a specific device (like a heart valve registry). These are often highly curated and offer high-quality data for specific therapeutic areas.
- Pharmacy Data: Tracking medication dispensing and refills. This is the gold standard for measuring “adherence” — whether a patient actually picks up their medication.
- Patient-Generated Data: Information from home-use medical devices, wearable tech (like smartwatches), or mobile health apps. This provides a continuous stream of data between doctor visits, capturing “invisible” symptoms like sleep quality or daily activity levels.
For more specific use cases, see our collection of real-world data examples.
The Role of HMA-EMA Catalogues in a Real-World Study
In Europe, transparency and trust are being built through the HMA-EMA catalogues of real-world data sources and studies. These catalogues have replaced older databases (like ENCePP and the EU PAS Register) with a more modern system based on FAIR principles — meaning data must be Findable, Accessible, Interoperable, and Reusable.
These catalogues help researchers identify which data sources are “fit-for-purpose” for a specific research question. For instance, if a researcher needs to study a rare pediatric condition, the catalogue can point them to specific registries across Europe that have the necessary data volume. By standardizing metadata (the data about the data), regulatory bodies ensure that everyone is speaking the same language, which is essential for cross-border research in the EU. This initiative is part of the broader Data Analysis and Real-World Interrogation Network (DARWIN EU), which aims to provide the EMA with timely and reliable RWE.
Regulatory Support from FDA and EMA
Regulatory agencies are no longer just “open” to real-world evidence; they are actively demanding it. In the US, the 21st Century Cures Act of 2016 was a turning point, mandating that the FDA establish a framework to evaluate the use of RWE. This led to the FDA’s 2018 RWE Framework, which outlines how RWE can support the approval of new indications for already-approved drugs or satisfy post-approval study requirements.
The FDA has a long history of using RWD for post-market safety surveillance (through programs like Sentinel), but they are now increasingly using it to evaluate effectiveness. This can lead to “label expansions” — allowing a drug to be used for a new group of patients or a different stage of a disease without requiring a brand-new, years-long RCT. For example, RWE was used to expand the indication of the drug Ibrance (palbociclib) to include men with breast cancer, a population too small to power a traditional clinical trial. Check out our summary of US regulatory guidance on using real-world data for more details.
Proving Clinical Impact: Lessons from Semaglutide and Heart Failure
To see a real-world study in action, we only need to look at some of the most popular treatments for Type 2 Diabetes (T2D) and heart failure. These studies provide the “missing link” between clinical trial results and actual patient outcomes in diverse healthcare systems.
Case Study: Oral Semaglutide in Switzerland
In the PIONEER REAL Switzerland study, researchers looked at how oral semaglutide performed in routine Swiss clinical practice. While Phase III trials gave us the baseline, this real-world study provided crucial local insights that helped Swiss physicians tailor their prescribing habits:
- HbA1c Reduction: Oral semaglutide reduced HbA1c (a measure of long-term blood sugar) by an estimated mean of −0.91%-points. This confirmed that the drug’s potency remained high even outside of trial conditions.
- Weight Loss: Patients saw an absolute body weight reduction of 4.72 kg (about 4.85%) over 34–44 weeks. This is particularly important as weight management is a primary goal for T2D patients.
- Success Rates: 64.2% of participants achieved an HbA1c of less than 7% by the end of the study, demonstrating high effectiveness in a real-world setting.
Case Study: Heart Failure Management in Belgium
Similarly, a large-scale study in Belgium involving over 5,400 patients examined sacubitril/valsartan for heart failure with reduced ejection fraction (HFrEF). The results were staggering: the drug reduced the risk of hospitalization for cardiovascular reasons by more than 26%.
Crucially, these benefits were seen in patients aged 75 and older and those with severe (NYHA Class IV) symptoms — groups that are often underrepresented or entirely excluded from traditional trials. This study gave cardiologists the confidence to prescribe the medication to their most vulnerable patients, knowing that the benefits observed in trials translated to the most complex cases.
What a Real-World Study Reveals About Treatment Persistence
Effectiveness isn’t just about how well a drug works in a cell; it’s about whether the patient actually takes it. Real-world studies often reveal “persistence” issues that trials don’t, because trial participants are often highly motivated and receive frequent reminders from study staff.
For example, an Italian study compared oral semaglutide to empagliflozin. It found that while semaglutide was more effective at lowering blood sugar (a mean difference of -0.35%), it had lower persistence. Patients were 1.47 times more likely to discontinue semaglutide, often due to gastrointestinal adverse events like nausea, which are common with GLP-1 receptor agonists.
The study also showed that in the real world, dose escalation is slower than in trials. Only 28.6% of oral semaglutide users reached the maximum 14 mg dose, and only 31.4% of empagliflozin users reached the 25 mg dose. This “real-world lag” is vital for doctors to understand when managing patient expectations; if a patient isn’t seeing the trial-level results, it might be because they haven’t yet reached the optimal dose. You can find more insights from real-world data in clinical research here.
Overcoming Regional Challenges and the Future of Global Data
Conducting a real-world study isn’t without its hurdles, and these challenges vary significantly by region. The primary obstacles are data quality, data privacy, and the lack of interoperability between different health systems.
In India, for instance, the lack of standardized data collection formats and poor record-keeping in some rural clinics can hinder research. However, the sheer diversity of the population and the rapid digitization of health records through the Ayushman Bharat Digital Mission offer a massive opportunity for studying rare diseases and comparative effectiveness if data quality can be improved.
In contrast, Hong Kong has a robust infrastructure. The Hospital Authority database covers nearly 30 years of data from 43 public hospitals, serving 11 million patients. Hong Kong is also pioneering cross-border data use within the Greater Bay Area (GBA), allowing HK-registered drugs to be used in mainland institutions to generate RWD. This creates a unique “living laboratory” for East Asian patient data.
Meanwhile, Nordic countries (Denmark, Sweden, Norway, Finland) continue to be a “powerhouse” for rare disease research due to their comprehensive national registries, universal healthcare systems, and high levels of digital integration. Their use of unique personal identification numbers allows researchers to link data across health, social, and economic registries with unparalleled accuracy.
Enhancing Data Quality for a Better Real-World Study
To move from “average” to “best-in-class,” organizations must solve the technical barriers that plague 91% of pharma RWE programs. The solution lies in federated governance and AI analytics. Traditional methods of data sharing involve moving massive datasets to a central server, which is a nightmare for security, compliance (like GDPR), and data sovereignty.
Instead of moving sensitive patient data to a central location, Lifebit uses a federated approach. Our platform brings the analysis to the data. This allows for:
- Data Harmonization: Making sure data from a hospital in London looks the same as data from a clinic in New York. We use the OMOP Common Data Model (CDM) to ensure that different coding systems (like ICD-10 vs SNOMED) are mapped to a single standard.
- Trusted Research Environments (TRE): Secure spaces where researchers can analyze data without ever seeing personal identifiers. The data never leaves the original provider’s firewall, satisfying even the strictest privacy regulations.
- Real-time Insights: Using our R.E.A.L. (Real-time Evidence & Analytics Layer) to spot safety trends as they happen. This is a massive leap forward from traditional pharmacovigilance, which often relies on retrospective reports that can be months or years old.
Following the real-world data 7 steps for success can help organizations navigate these complexities, from defining the research question to selecting the right technology partner.
Frequently Asked Questions about Real-World Studies
What is the difference between RWD and RWE?
RWD (Real-World Data) is the raw “stuff” — the records, claims, and notes. It is the collection of facts. RWE (Real-World Evidence) is the “so what” — the clinical conclusions you draw after you’ve applied rigorous statistical analysis to that data to answer a specific clinical question.
Can a real-world study be used for drug approval?
Yes. While RCTs are still required for most initial approvals (Phase I-III), regulatory bodies like the FDA and EMA increasingly use RWE to approve new uses for existing drugs (label expansion), to support approvals in rare diseases where trials are difficult, or to fulfill safety requirements after a drug is already on the market.
What are the main limitations of real-world evidence?
The biggest challenge is “bias” and “confounding.” Because patients aren’t randomized, there might be underlying reasons why one person got a drug and another didn’t (e.g., one patient was healthier to begin with). This is known as “selection bias.” Also, RWD can be “messy” or incomplete compared to the perfect records kept during a clinical trial. Researchers must use advanced techniques like propensity score matching to account for these differences.
How does AI improve real-world studies?
AI and machine learning are essential for processing the vast amounts of unstructured data in EHRs. NLP can read doctor’s notes to identify symptoms or side effects that aren’t captured in structured codes. Furthermore, AI can help identify patterns in patient data that might predict which individuals are most likely to respond to a specific treatment, moving us closer to truly personalized medicine.
Is patient privacy protected in these studies?
Absolutely. Modern real-world studies use techniques like de-identification, anonymization, and federated learning. In a federated model, researchers only see the aggregated results of an analysis, never the individual patient records, ensuring compliance with laws like GDPR and HIPAA.
Conclusion: Unlocking the Next Era of Evidence Generation
The era of relying solely on “perfect” lab patients is ending. To truly understand human health, we must embrace the complexity of the real world. A real-world study provides the missing link between a drug’s potential and its actual performance in the hands of a patient. It allows us to see the nuances of age, ethnicity, and lifestyle that traditional trials intentionally ignore.
As we look to the future, the integration of RWE with other emerging technologies like digital twins and multi-omics will further accelerate drug discovery. We are moving toward a world where every patient’s journey contributes to a global knowledge base, ensuring that the next person with the same condition receives the most effective treatment possible from day one.
At Lifebit, we are building the infrastructure to make this possible. Our federated AI platform and Trusted Research Environments allow biopharma companies and governments to access global, multi-omic data securely and in real-time. By connecting the world’s biomedical data, we aren’t just looking at what happened yesterday — we are predicting what will work tomorrow. We are turning the “messy” reality of healthcare into a clear roadmap for better patient outcomes.
Ready to lead the RWE revolution? Download the White Paper on Regulatory Compliance and RWE to learn how to build a data architecture that satisfies regulators and saves lives.

