The Ultimate Guide to Adverse Event Reporting Software

Why Every Drug Safety Team Needs to Understand the Adverse Event Reporting System
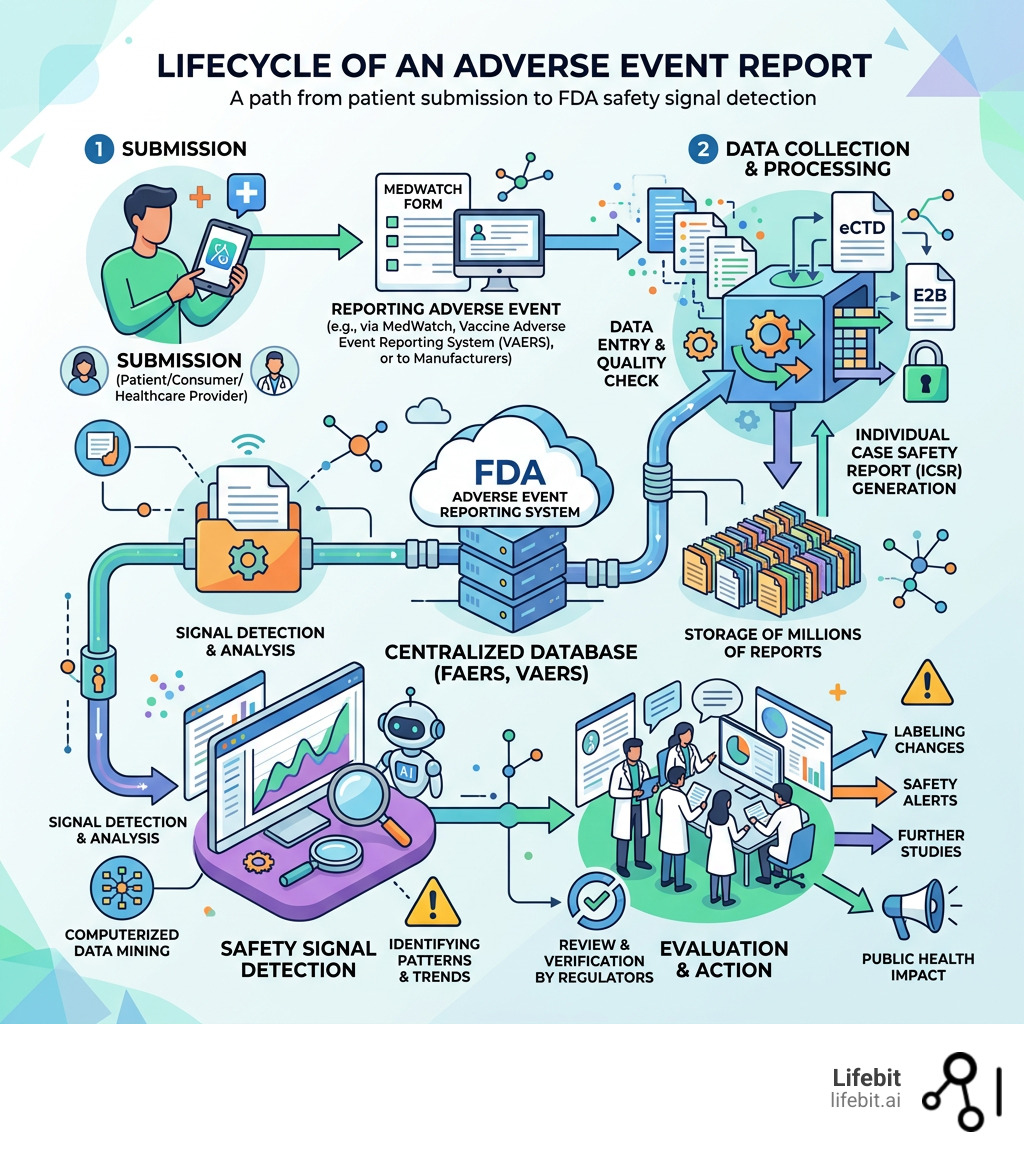
An adverse event reporting system is a structured database and process that collects, stores, and analyzes reports of unexpected health events linked to drugs, biologics, or vaccines — helping regulators detect safety signals before they become public health crises.
Here is a quick overview of the key systems in the U.S.:
| System | Managed By | Covers | Who Can Report |
|---|---|---|---|
| FAERS | FDA | Drugs & therapeutic biologics | Manufacturers, providers, consumers |
| VAERS | CDC & FDA | Vaccines | Anyone |
| MedWatch | FDA | Drugs, devices, supplements, cosmetics | Providers & consumers |
Post-marketing surveillance is where most real-world safety signals emerge. Pre-approval clinical trials are run on small, controlled groups — they simply cannot catch rare adverse events that only surface when millions of patients start using a product. That is exactly why systems like FAERS exist: to catch what trials miss.
The FDA’s Adverse Event Reporting System (FAERS) alone has collected data on over 4 million cases — and researchers have used that data to identify 115 sex-biased diseases with over 94% concordance with published literature. That is not just pharmacovigilance. That is population-level disease intelligence hiding in plain sight.
But accessing, interpreting, and acting on this data is harder than it sounds. Duplicate reports, missing fields, causation gaps, and evolving submission standards all stand between your team and reliable safety insights.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where I lead the development of federated AI platforms that enable compliant, real-time analysis of siloed biomedical data — including adverse event reporting system data — without ever moving sensitive records. My background in computational biology and AI-driven data integration gives me a close view of both the power and the friction points in large-scale pharmacovigilance workflows.
This guide walks you through everything: how FAERS works, its limitations, submission requirements, advanced analytics use cases, and the software solutions built to make adverse event reporting faster and more reliable.
Simple adverse event reporting system word guide:
Mastering the Adverse Event Reporting System for Faster Safety Insights
At its core, the FDA Adverse Event Reporting System (FAERS) database is the backbone of post-marketing safety surveillance in the United States. Its primary purpose is to support the FDA’s mission to monitor the safety of drug and therapeutic biologic products once they reach the general public. This system transitioned from the older AERS (Adverse Event Reporting System) to the current FAERS in 2012, significantly improving data structure and accessibility.
The database doesn’t just look for “side effects.” It aggregates a wide spectrum of safety data points:
- Adverse Event Reports: Unanticipated medical experiences that occur while a patient is taking a drug, regardless of whether the drug is suspected to be the cause.
- Medication Error Reports: Critical data regarding mistakes in prescribing, dispensing, or administering drugs, which often highlight issues with confusing packaging or similar-sounding drug names.
- Product Quality Complaints: Issues like contamination, stability problems, or packaging defects that lead to an adverse event, providing a direct link between manufacturing standards and patient safety.
By centralizing these reports, the FDA can identify “signals”—patterns of unexpected or high-frequency events—that might require regulatory action. These actions range from simple label updates and “Black Box Warnings” to the issuance of Risk Evaluation and Mitigation Strategies (REMS) or, in extreme cases, the complete removal of a product from the market.
Accessing Data via the Adverse Event Reporting System Public Dashboard
For years, FAERS data was locked behind complex data requests or difficult-to-parse raw files. Today, the Launch the FAERS Public Dashboard provides a highly interactive, web-based tool that allows the public to query data in a user-friendly fashion. This transparency is vital for building public trust in the pharmaceutical industry.
If you are a researcher or a safety professional, you have several options for data retrieval, depending on the depth of analysis required:
- The Public Dashboard: Best for quick searches on specific drugs or reactions. It visualizes trends and demographics instantly, allowing users to filter by year, age group, and seriousness of the event.
- Quarterly Data Files: For those who need to perform deep-dive analytics or build custom models, the FDA releases quarterly data files. These are available from 2012Q4 onwards via the National Bureau of Economic Research (NBER) and the FDA website. These files contain raw data tables including demographic information, drug therapy dates, and outcome codes.
- Legacy AERS Data: If your research requires a longer historical view, you can access data from 2004Q1 to 2012Q3. Many researchers use harmonized variable names to bridge the gap between legacy and modern FAERS records, though caution is advised as coding standards have evolved over the decades.
Information Included in Standard Safety Reports
What exactly is inside an Individual Case Safety Report (ICSR)? A valid report typically requires four key elements, often referred to as the “Four Pillars” of a valid case: an identifiable patient (initials, age, or sex), a suspect drug or biologic, a detailed adverse event (or death), and an identifiable reporter (contact information for the person submitting the report).
Specifically, the adverse event reporting system captures:
- Patient Demographics: Age, sex, and weight, which are essential for identifying populations at higher risk for specific reactions.
- Drug Information: Dosage, frequency, route of administration (e.g., oral, intravenous), and therapy start/end dates. It also distinguishes between “suspect” drugs and “concomitant” drugs (other medications the patient was taking).
- Reaction Information: The specific symptoms or diagnoses coded using MedDRA (Medical Dictionary for Regulatory Activities) terminology. This ensures that a “heart attack” is consistently recorded as “Myocardial infarction” across all global reports.
- Patient Outcomes: Whether the event resulted in hospitalization, disability, life-threatening illness, congenital anomaly, or death.
However, data quality remains a significant hurdle for the industry. For instance, approximately 36% of data in the FAERS “indication” field (the reason the drug was taken) is missing, particularly in older records. This makes it challenging to distinguish whether a symptom was caused by the drug or the underlying disease, a phenomenon known as “confounding by indication.”
Overcoming Data Limitations and Causation Challenges
While FAERS is a goldmine, it isn’t perfect. We often remind our partners that a report in an adverse event reporting system does not establish causation. Just because a patient had a heart attack while taking “Drug X” doesn’t mean “Drug X” caused it—it could be related to their diet, genetics, or another medication. The system is designed for signal detection, not signal validation.
Key limitations include:
- Duplicate Reports: A doctor, a manufacturer, and the patient might all report the same event, creating “noise” in the data. Advanced de-duplication logic is required to ensure a single event isn’t counted three times.
- Under-reporting: This is a classic challenge in pharmacovigilance. Many mild or even moderate events are never reported to the FDA, meaning the database primarily captures the most severe or unusual cases.
- Verification Gaps: Reports reflect the observations and opinions of the reporter; they are not always medically verified by a physician or supported by lab results.
- Reporting Bias (The Weber Effect): Media attention on a specific drug often triggers a surge in reports. Historically, reporting for a new drug tends to peak at the end of the second year after launch and then declines, regardless of the actual safety profile. This is known as the Weber Effect.
Statistical Methods for Signal Detection
To navigate these issues, modern pharmacovigilance platforms use advanced Disproportionality Analysis (DPA). This involves comparing the frequency of a specific drug-event combination against the frequency of that event for all other drugs in the database. Common metrics include:
- Proportional Reporting Ratio (PRR): A simple ratio that compares the proportion of a specific adverse event for a drug to the proportion of the same event for other drugs.
- Reporting Odds Ratio (ROR): Similar to an odds ratio in clinical trials, this helps identify if the odds of an event are significantly higher for a specific product.
- Information Component (IC): A Bayesian tool used by the World Health Organization (WHO) to find associations that stand out from the background noise.
| Feature | FAERS Public Dashboard | Raw Data Downloads (NBER/FDA) |
|---|---|---|
| Ease of Use | High (Interactive/Web-based) | Low (Requires SAS/Stata/Python) |
| Granularity | Summary level | Patient/Case level |
| Best For | Quick trend spotting | Academic research & signal detection |
| Update Frequency | Quarterly | Quarterly |
| Technical Skill | None required | High (Data Science/Bioinformatics) |
By applying these statistical thresholds, safety teams can separate true safety signals from background noise, allowing them to focus their limited resources on the most credible threats to patient safety.
Technical Standards and Submission Requirements
To ensure that safety data can be shared globally, the FDA adheres to strict international standards developed by the International Council for Harmonisation (ICH). The most critical of these is the ICH E2B Individual Case Safety Report (ICSR) specification. This standard defines the data elements and the XML (Extensible Markup Language) format required for electronic transmission.
Currently, the industry is in a major transition phase. The FDA began accepting electronic ICSRs using the E2B(R3) standard in January 2024. This new version allows for more granular data, including better tracking of drug components and improved mapping of clinical trial data to post-marketing reports. While companies can still use the older E2B(R2) standard for now, the final deadline for full implementation is April 1, 2026. Once a company makes the switch to R3, there is no going back to R2, making the transition a high-stakes technical project for IT and safety teams.
Standardized vocabularies are also mandatory to prevent linguistic ambiguity:
- MedDRA (Medical Dictionary for Regulatory Activities): The global standard for coding all adverse events, medical history, and medication errors. It is organized into a five-level hierarchy, from “Lowest Level Terms” to “System Organ Classes.”
- SNOMED CT: Often used for mapping clinical terms and diseases within Electronic Health Records (EHRs) that feed into safety systems.
- UMLS (Unified Medical Language System): A set of files and software that brings together many health and biomedical vocabularies to enable interoperability between different systems.
Compliance Requirements for an Adverse Event Reporting System
Compliance isn’t optional—it’s a regulatory mandate with significant legal and financial consequences. Manufacturers, distributors, and even retailers named on a product label must report serious adverse events to the FDA.
- Submission Methods: Most large pharmaceutical companies use the Electronic Submissions Gateway (ESG). This is a highly secure, database-to-database transmission system that provides an automated “Acknowledgement” (ACK) message once the FDA successfully receives the file. For smaller entities or low-volume reporters, the Safety Reporting Portal (SRP) allows for manual entry via a web form.
- Timelines: The “15-Day Alert” rule is the industry standard. Serious and unexpected events must be reported within 15 business days of the company becoming aware of the event. Non-serious events are typically bundled into Periodic Safety Update Reports (PSURs) or Periodic Adverse Drug Experience Reports (PADERs).
- The Impact of MoCRA: The Modernization of Cosmetics Regulation Act of 2022 (MoCRA) has expanded these requirements to the cosmetics industry. As of late 2023, cosmetic companies must also report serious adverse events within 15 days, using the same E2B standards previously reserved for drugs and devices.
- Inspections: The FDA’s Office of Surveillance and Epidemiology (OSE) actively inspects companies to ensure they aren’t “cherry-picking” reports or failing to meet submission deadlines. A failure in the adverse event reporting system workflow can lead to Warning Letters or consent decrees.
Advanced Analytics: Identifying Sex-Biased Diseases and Drug Interactions
One of the most exciting developments in recent years is the use of FAERS for things other than just catching side effects. Because the database is so vast, it can be used for “disease monitoring” and identifying complex drug-drug interactions (DDIs) that were never studied in clinical trials.
A landmark scientific research study on sex differences in autoimmune disease demonstrated this utility beautifully. Researchers analyzed over 4 million FAERS cases from 1997 to 2011. By filtering out sex-related terms and applying high statistical thresholds (p-value < 10⁻¹⁰), they identified 115 diseases with significant sex bias.
Key Findings from the Study:
- Of the 115 sex-biased diseases identified, 53 had existing literature confirming the bias, validating the use of FAERS as a reliable research tool.
- The concordance rate between FAERS data and published literature was a staggering 94.34%.
- The study successfully identified sex-biased prevalence for conditions like rheumatoid arthritis, lupus, and autoimmune hepatitis, which are known to affect women more frequently.
- It even caught “hidden” signals, such as optic ischemic neuropathy, which showed a sex bias in FAERS data years before it was officially reported in 2012 publications.
Beyond Sex Bias: Drug-Drug Interaction (DDI) Screening
FAERS is also being used to identify dangerous combinations of medications. In the real world, patients often take five or more drugs simultaneously (polypharmacy). Clinical trials rarely test these specific combinations. By using “Association Rule Mining” on FAERS data, researchers can identify pairs of drugs that, when taken together, result in a much higher rate of adverse events than when taken alone.
For example, researchers have used the adverse event reporting system to study the interaction between certain antibiotics and statins, which can lead to a higher risk of rhabdomyolysis (muscle breakdown). This type of “real-world evidence” (RWE) is becoming a cornerstone of modern precision medicine, allowing doctors to tailor prescriptions based on a patient’s entire medication profile.
This proves that an adverse event reporting system can serve as a richer resource for disease surveillance than many Electronic Medical Records (EMRs) because of its massive scale, standardized coding, and public availability. It allows us to see patterns that are invisible at the level of a single hospital or even a single country.
The Future of Integrated Disease Surveillance
The next frontier for the adverse event reporting system is integration. We are moving away from passive “wait and see” reporting toward real-time pharmacovigilance. This involves connecting FAERS data with other data streams, such as social media sentiment, wearable device data, and genomic databases.
The future of drug safety lies in Federated AI. Instead of moving massive, sensitive datasets into a single silo (which creates security risks, data lag, and regulatory hurdles regarding data residency), federated architectures allow AI models to travel to the data. This enables:
- Secure Collaboration: Governments and biopharma companies can analyze global safety data without compromising patient privacy or violating GDPR/HIPAA regulations. The raw data stays behind the organization’s firewall, while only the “insights” or model weights are shared.
- Real-Time Monitoring: Detecting a safety signal in hours rather than months. By using Natural Language Processing (NLP), AI can scan incoming reports and automatically flag those that meet certain “seriousness” criteria, bypassing the manual triage process.
- Data Harmonization: Automatically bridging the gap between MedDRA, SNOMED, and genomic data. This allows researchers to see if a specific genetic marker makes a patient more likely to experience a side effect reported in FAERS.
The Role of Real-World Evidence (RWE)
By integrating FAERS with other public health databases and real-world evidence (RWE), we can overcome current challenges like under-reporting and missing indication fields. For instance, if a FAERS report is missing the “indication” for a drug, an integrated system could potentially pull that information from a linked (and anonymized) electronic health record.
This creates a proactive “early warning system” that doesn’t just react to harm but predicts it. We are entering an era where the adverse event reporting system is no longer just a regulatory requirement, but a strategic asset for drug development and patient care.
For a deeper dive into how technology is changing the game, check out our AI for pharmacovigilance complete guide.
Frequently Asked Questions about Adverse Event Reporting
How do I submit a report to the adverse event reporting system?
Consumers and healthcare professionals should use the MedWatch online voluntary reporting form. Pharmaceutical companies must use the Electronic Submissions Gateway (ESG) or the Safety Reporting Portal (SRP) to meet regulatory requirements. For vaccine-related reactions, reports should be submitted to VAERS.
What are the main limitations of FAERS data?
The primary limitations are that reports do not prove causation, data may be incomplete (36% of indication fields are often missing), and the system is subject to reporting bias from media coverage. Additionally, FAERS cannot be used to calculate the actual incidence or “rate” of an event because the total number of people taking the drug is often unknown.
How often is the FAERS data updated?
The FDA typically releases FAERS data files on a quarterly basis. The Public Dashboard is also updated regularly to reflect these quarterly additions, allowing for timely safety surveillance.
Conclusion
The adverse event reporting system has evolved from a simple “complaint box” into a sophisticated engine for global health intelligence. From the 4 million cases in FAERS that help us understand sex-biased diseases to the strict new E2B(R3) standards ensuring data integrity, the system is more powerful than ever.
However, the “noise” in the data remains a challenge. To truly unlock the potential of these databases, safety teams need tools that can handle scale, complexity, and compliance simultaneously.
At Lifebit, we specialize in helping organizations navigate this landscape. Our federated AI platform, featuring the R.E.A.L. (Real-time Evidence & Analytics Layer), allows you to access and analyze global biomedical data securely and in real-time. Whether you are looking for a pharmacovigilance compliance solution or aiming to lead the way in drug safety AI, we are here to help you turn raw reports into life-saving insights.
Ready to modernize your safety surveillance? Explore Lifebit’s Real-time Adverse Drug Reaction Surveillance

