How AI is revolutionizing disease detection and prevention

AI Disease Prediction: Predict 1,000 Conditions 20 Years Early (2026 Guide)
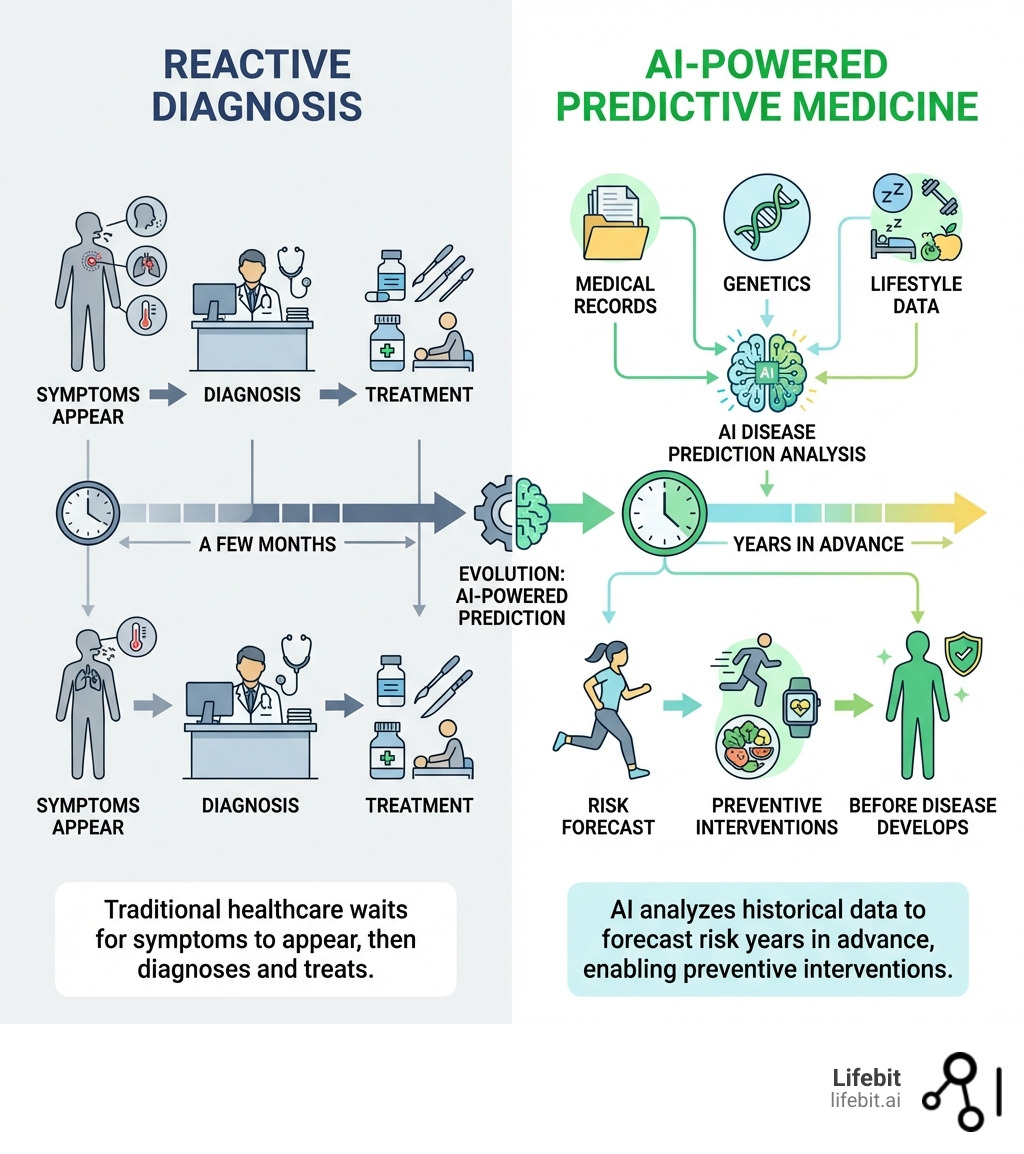
AI disease prediction uses machine learning and deep learning algorithms to analyze medical records, genetic data, sleep patterns, and lifestyle factors—forecasting health risks years or even decades before symptoms appear. Instead of waiting for disease to strike, AI models like Delphi and SleepFM can identify individuals at high risk for conditions ranging from heart attacks to Parkinson’s disease, enabling early intervention and personalized prevention strategies.
Key capabilities of AI disease prediction:
- Long-term forecasting: Models predict over 1,000 diseases up to 20 years in advance using medical history
- Multi-modal analysis: Combines sleep data, genetic variants, medical records, and lifestyle factors
- High accuracy: Achieves 95%+ accuracy for conditions like coronary heart disease and breast cancer
- Early detection: Identifies high-risk patients before symptoms appear, enabling preventive care
- Population-scale validation: Tested on millions of patients across UK Biobank and Danish registries
Until recently, healthcare operated reactively—diagnosing illness after it caused damage. Today, AI models trained on millions of patient records can spot patterns invisible to human clinicians. Delphi, built on GPT-2 architecture, analyzes 400,000 UK Biobank participants to predict disease trajectories with accuracy rivaling or exceeding traditional clinical tools like QRisk3. SleepFM examines overnight polysomnography from 35,000 patients to forecast 130 disease categories, achieving C-index scores above 0.8 for cancers, circulatory conditions, and mental disorders. PrimateAI-3D uses 4.5 million primate genetic variants to predict which human mutations cause disease, enabling ancestry-portable risk scores that work across diverse populations.
The Economic and Social Imperative of Predictive Medicine
The shift from reactive treatment to proactive prevention represents a fundamental change in medicine. AI doesn’t just diagnose faster—it predicts disease before it happens, giving clinicians and patients time to act. For conditions like heart disease and dementia that progress slowly, early intervention can delay or prevent onset entirely. For rare genetic disorders, AI identifies undiagnosed patients by recognizing subtle patterns in their medical history.
Beyond individual health, this shift addresses the “Silver Tsunami”—the global aging population that threatens to overwhelm healthcare budgets. By identifying high-risk individuals decades in advance, public health agencies can allocate resources more efficiently, focusing on high-impact preventive measures rather than expensive late-stage treatments. This transition is estimated to save trillions in global healthcare costs by reducing hospitalizations and long-term disability.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over 15 years building federated genomics and biomedical data platforms that enable secure, real-time AI disease prediction across siloed datasets—powering drug discovery, pharmacovigilance, and precision medicine for public health agencies and pharmaceutical organizations worldwide. Our work focuses on making these breakthrough AI models accessible and actionable in real-world clinical settings.
Common AI disease prediction vocab:
The “Grammar” of Disease: 3 Models Predicting Your Health Future
The landscape of modern medicine is being rewritten by a handful of “foundation models” specifically designed for human health. Just as ChatGPT learned the grammar of human language, these new models are learning the “grammar” of disease progression. By treating a patient’s medical history as a sequence of events—much like words in a sentence—these tools can predict the next “word” (or diagnosis) in a person’s life story.

Several notable models have emerged recently, including Delphi, SleepFM, and RiskPath. These aren’t just academic exercises; they are robust systems validated against millions of real-world data points. For example, AI-powered diagnostics are now capable of analyzing medical images with a level of precision that rivals human experts, but the real magic happens when we look decades into the future.
Delphi: Forecasting 1,000 Conditions Using AI Disease Prediction
Delphi represents a massive leap forward. Its architecture is based on GPT-2, the same technology that powered earlier versions of ChatGPT. However, instead of reading the internet, Delphi was trained on the medical histories of over 400,000 participants from the UK Biobank.
According to scientific research on Delphi AI, the model can predict the onset of more than 1,000 diseases up to 20 years in advance. It works by analyzing “disease tokens”—discrete medical events like a high blood pressure reading, a specific prescription, or a minor surgery—to understand how one condition often leads to another. For instance, it can group diabetes and vision loss together because it “sees” the statistical inevitability of their connection in the data.
What makes Delphi particularly impressive is its generalizability. When tested on a completely different population—1.9 million people in the Danish National Patient Registry—it maintained its predictive power. This suggests that the “language of disease” it learned in the UK applies across borders. This is exactly the kind of cross-border insight we facilitate at Lifebit through our federated platform, ensuring that models are not just accurate for one demographic but are globally applicable.
SleepFM: Using Sleep Data for Multi-Organ AI Disease Prediction
While Delphi looks at medical records, SleepFM looks at your body while it rests. Developed using data from the Stanford Sleep Medicine Center, this model analyzes polysomnography (sleep study) data from 35,000 patients aged 2 to 96.
Sleep is a window into our systemic health. SleepFM can predict 130 different disease categories by looking at brain waves (EEG), heart activity (ECG), and oxygen levels. The results are staggering:
- Parkinson’s disease: C-index of 0.89
- Dementia: C-index of 0.85
- Prostate cancer: C-index of 0.89
- Heart attack: C-index of 0.81
A C-index of 0.8 means the model is correct 80% of the time in ranking which patient is more likely to develop the condition. Interestingly, SleepFM often finds trouble when the “brain is asleep but the heart is awake,” identifying physiological “mismatches” that precede clinical symptoms. This highlights the importance of multi-modal data; by combining sleep signals with traditional EHR data, we can achieve a level of predictive granularity previously thought impossible.
RiskPath: Decoding the Neural Grammar of Mental Health
While physical ailments often have clear biomarkers, mental health has historically been harder to predict. RiskPath, a model developed by researchers including Nina de Lacy, MD, at the University of Utah, is changing this. RiskPath treats psychiatric history as a longitudinal sequence, identifying patterns that precede major depressive episodes or the onset of schizophrenia. By analyzing the “pathway” of symptoms and interventions, RiskPath helps clinicians intervene before a crisis occurs, moving mental healthcare from crisis management to long-term stability.
Multi-Omics: Decoding Genetic Risk Before It Becomes Deadly
The future of AI disease prediction isn’t just about looking at one data source; it’s about “multi-omics”—combining DNA, proteins, and clinical history into a single, unified view. This is where AI for precision medicine truly shines. By integrating these diverse data streams, we can move away from “one-size-fits-all” healthcare.
The Multi-Omic Stack: Beyond DNA
To truly understand disease, we must look at the entire biological stack:
- Genomics: Your blueprint (DNA).
- Transcriptomics: Which genes are actually “turned on” (RNA).
- Proteomics: The functional machines of the cell (Proteins).
- Metabolomics: The chemical fingerprints left behind by cellular processes.
AI models are now capable of “fusing” these layers. For example, a patient might have a genetic predisposition for heart disease (Genomics), but their Proteomics might show that their body is currently compensating for it. AI can weigh these conflicting signals to provide a real-time risk score that is far more accurate than a simple DNA test.
Decoding Genetic Risk with PrimateAI-3D
One of the biggest problems in genomics is knowing which genetic mutations (variants) actually cause disease. Most are “benign” (harmless), but “missense variants” can be deadly. Scientific research on PrimateAI-3D solved this by looking at our closest relatives.
By sequencing 233 primate species, researchers discovered that variants found in primates are 99% likely to be benign in humans. This provided a massive “clean” dataset to train a 3D convolutional neural network. PrimateAI-3D uses AlphaFold structures to see how a mutation physically breaks a protein. This model has enabled:
- 73% more gene-phenotype associations detected in rare variant tests.
- Ancestry portability: Unlike traditional genetic scores that only work for Europeans, PrimateAI-3D-based scores work across diverse ethnicities because they are based on evolutionary conservation.
This is a game-changer for newborn genomic sequencing early detection disease, allowing us to identify life-threatening risks the moment a child is born, regardless of their background.
Infectious Disease Surveillance and Outbreak Prediction
AI isn’t just for chronic illness; it’s a frontline defense against global pandemics. The BlueDot platform is a prime example. By scanning global news reports in 65 languages, airline ticketing data, and animal disease reports in real-time, BlueDot successfully flagged the COVID-19 outbreak in Wuhan days before official alerts were issued by the WHO.
This type of surveillance allows governments to allocate resources—like hospital beds and ventilators—long before a surge hits. It also enables “digital twinning” of cities to simulate how a virus might spread, allowing for targeted interventions rather than blunt, city-wide lockdowns. It’s the ultimate early warning system, turning “unforeseen” disasters into manageable public health events.
98% Accuracy: Why AI Outperforms Traditional Clinical Screenings
The question every doctor asks is: “Is this better than what I already use?” The data says yes. Traditional tools like QRisk3 (for heart disease) or the Framingham score often require blood tests, manual entry, and only look at a handful of variables. AI models like Delphi only need age, sex, and history to achieve similar or superior results.
| Model | Target Condition | Metric (AUC/C-index) | Traditional Baseline |
|---|---|---|---|
| Delphi | 1,000+ Diseases | 0.80+ (Avg) | QRisk3 / Framingham |
| SleepFM | Parkinson’s | 0.89 | Clinical Exam |
| RiskPath | Mental Health | High Precision | Standard Screening |
| LSSVM | Breast Cancer | 98.53% | Mammography |
| SVM | Coronary Heart Disease | 95.00% | Traditional Risk Factors |
Scientific research on RiskPath shows that these models can even be integrated into clinical decision support systems to help psychiatrists understand the neural underpinnings of mental illness. By providing a “probability score” for various outcomes, AI acts as a co-pilot for the physician, highlighting patients who might otherwise slip through the cracks.
The Role of Federated Learning in Global Validation
Accuracy is meaningless if it only applies to one group of people. We know that many AI models suffer from demographic bias because they are trained on mostly white, European populations. To fix this, we need to follow the machine learning precision medicine guide and validate models on external, diverse datasets.
At Lifebit, we solve this through Federated Learning. Instead of moving sensitive patient data across borders—which is a privacy nightmare and often illegal under GDPR—our platform allows the model to travel to the data. We can train an algorithm on a cohort in Singapore, then move it to a dataset in Brazil, and finally to the UK. The model learns from all these diverse populations without the data ever leaving its secure home environment. This ensures that the resulting AI is robust, fair, and accurate for everyone, regardless of their ethnicity or geographic location.
High-Accuracy Predictions for Chronic Conditions
The numbers coming out of recent studies are, frankly, astonishing:
- Breast Cancer: The LSSVM (Least Squares Support Vector Machine) algorithm achieved 98.53% accuracy on the Wisconsin Breast Cancer Dataset, significantly reducing false positives that lead to unnecessary biopsies.
- Alzheimer’s: The EfficientNetB0 model reached 92.98% accuracy in predicting the disease by analyzing subtle changes in brain MRI scans that are invisible to the naked eye.
- Parkinson’s: A combination of genetic algorithms and random forests achieved 95.58% accuracy for early diagnosis by analyzing motor patterns and vocal tremors.
These aren’t just incremental improvements; they are tectonic shifts in diagnostic capability that will redefine the standard of care in the coming decade.
Privacy vs. Prediction: Solving the Ethics of Predictive Health
With great power comes great responsibility—and a lot of paperwork. Moving AI disease prediction from the lab to the clinic requires solving three major problems: privacy, bias, and consent.
Addressing Data Bias and Regulatory Needs
If a model is trained only on 60-year-old men, it might fail to predict a heart attack in a 40-year-old woman. As Tomas Fitzgerald from the European Molecular Biology Laboratory notes, the most obvious application is an early warning system, but we must ensure it works for everyone. Underrepresented populations—including ethnic minorities and younger age groups—must be included in the training data to prevent “algorithmic bias.”
Furthermore, there is the “right not to know.” As we improve our ability to predict rare disease diagnosis genomics, we have to ask: do patients want to know they might get an incurable disease in 20 years? Regulatory frameworks like the EU AI Act and the FDA’s Digital Health Software Precertification Program are beginning to address these issues, requiring developers to prove their models are both safe and transparent.
The Insurance Dilemma and Genetic Privacy
One of the most significant ethical hurdles is the potential for “genetic discrimination.” If an AI predicts a high risk of a costly chronic condition, could an insurance company raise premiums or deny coverage? In the US, the Genetic Information Nondiscrimination Act (GINA) provides some protection, but it does not cover life, disability, or long-term care insurance. As predictive AI becomes more ubiquitous, we will need stronger legislative protections to ensure that a person’s future health forecast isn’t used against them.
The Future of Wearables and Real-Time Monitoring
The next frontier is integrating the data from your wrist. While Delphi uses hospital records, the next generation of models will use continuous data from wearables. Alejandro Frangi from the University of Manchester emphasizes that “it’s not about collecting more data, it’s about using models that extract the right knowledge.”
Imagine a world where your smartwatch detects a subtle change in your gait—invisible to you—and alerts your doctor that you have an 85% risk of developing Parkinson’s in the next five years. This allows for “resource allocation” at a national level, ensuring that specialists and treatments are ready before the patient even feels “sick.” This real-time monitoring, combined with federated AI, will create a “living” health record that evolves with the patient, providing a constant safety net of predictive care.
Frequently Asked Questions about AI Disease Prediction
How accurate is AI at predicting diseases 20 years in advance?
For many chronic conditions with clear progression patterns, like heart disease and diabetes, AI models like Delphi are remarkably accurate, often matching or exceeding the performance of current clinical tools. For example, Delphi achieves a C-index of over 0.80 for many conditions, meaning it correctly identifies the higher-risk individual in 80% of cases. However, they are less reliable for “random” events like viral infections or very rare inherited disorders that lack sufficient training data.
Can AI predict diseases from wearable device data?
Yes. Models like SleepFM already use physiological signals (heart rate, brain waves, oxygen saturation) that are increasingly being captured by high-end consumer wearables. While hospital-grade polysomnography is currently more accurate, the gap is closing rapidly as wearable sensors improve in sensitivity and AI algorithms become better at filtering out “noise” from daily activity.
What are the main ethical risks of AI in healthcare?
The primary risks include data privacy (who owns and has access to your future health forecast?), algorithmic bias (does the AI work for all ethnicities and genders?), and psychological impact (how does knowing a future diagnosis for an incurable disease affect a person’s mental health and life choices?). There is also the risk of “over-diagnosis,” where AI identifies a risk that might never have manifested as a clinical disease, leading to unnecessary anxiety and medical intervention.
Will AI replace doctors in diagnosing diseases?
No. AI is designed to be a “clinical decision support tool.” It processes vast amounts of data that no human could ever read, but the final diagnosis and treatment plan remain the responsibility of the clinician. AI acts as a high-powered microscope, allowing doctors to see patterns and risks that were previously invisible, but the human element of care—empathy, ethical judgment, and complex clinical reasoning—cannot be automated.
Conclusion: The End of Reactive Medicine
The era of reactive medicine is ending. Through AI disease prediction, we are gaining the ability to see through time, identifying health risks decades before they manifest. From the transformer-based “language” models of Delphi to the sleep-based insights of SleepFM and the genetic precision of PrimateAI-3D, the tools are already here. We are moving from a world where we treat the sick to a world where we maintain the healthy.
At Lifebit, we are proud to be the infrastructure that makes this possible. Our federated AI platform ensures that this revolution is built on a foundation of security, diversity, and real-time collaboration. By connecting the world’s biomedical data, we aren’t just predicting the future—we are helping clinicians and researchers change it.
The goal is simple: a world where “early detection” is replaced by “total prevention.” By identifying the “grammar” of disease early, we can rewrite the ending of the story. And with AI, that world is finally within reach.

