Everything You Need to Know About Electronic Consent in Clinical Trials

How Electronic Consent in Clinical Trials Cuts Protocol Deviations by 50%
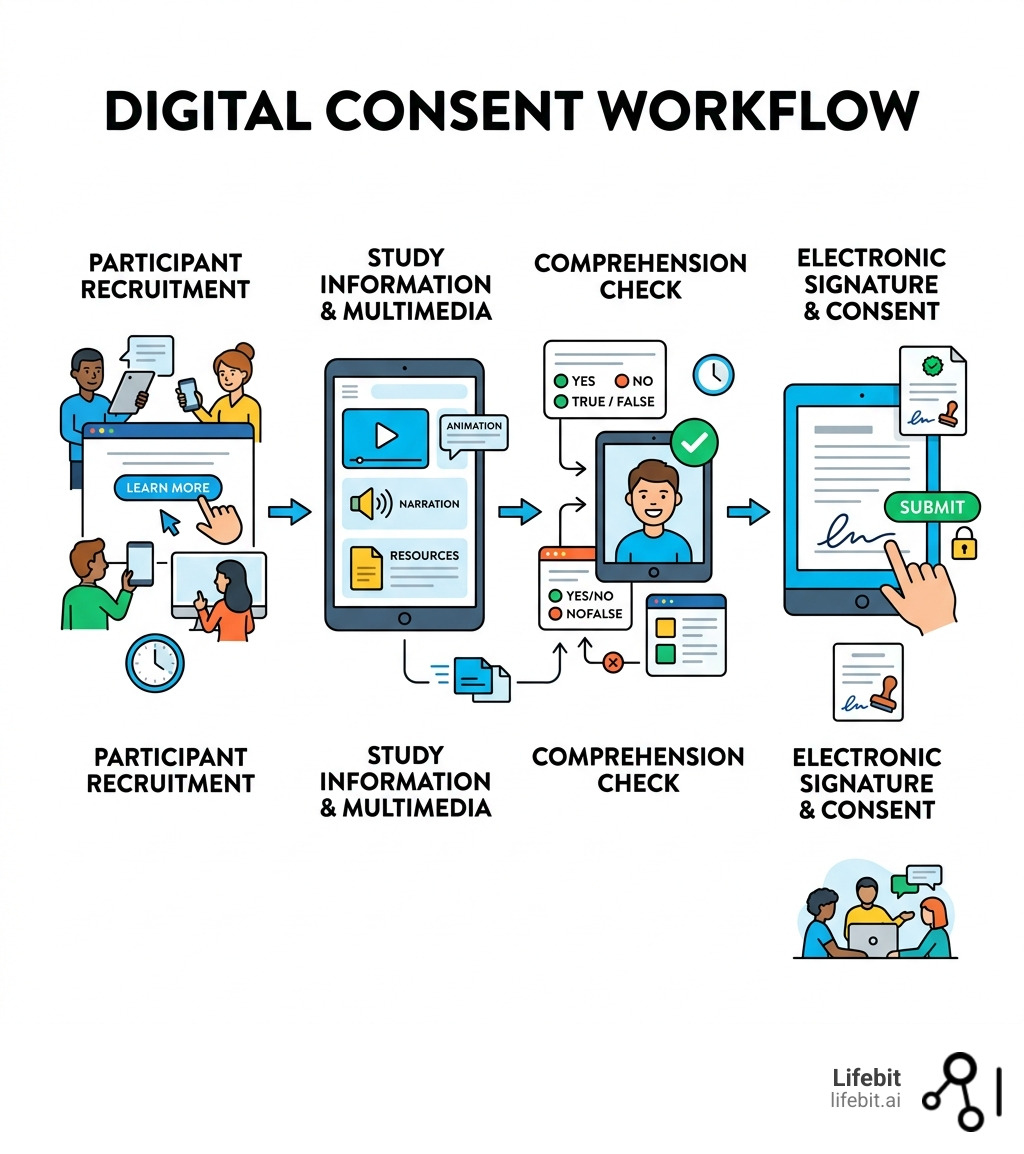
Electronic consent in clinical trials — known as eConsent — is the use of electronic systems to present study information, verify participant understanding, and capture a legally valid signature, replacing the traditional paper consent form.
Here is what you need to know at a glance:
| Question | Quick Answer |
|---|---|
| What is eConsent? | A digital process to inform, engage, and obtain consent from trial participants |
| Is it legally valid? | Yes, when compliant with FDA 21 CFR Part 11 and applicable regulations |
| Does it replace paper entirely? | Not always — hybrid models combining digital and paper are common |
| Who needs IRB approval? | All studies using eConsent, regardless of platform |
| Key benefit? | Fewer protocol deviations, better participant comprehension, faster enrollment |
Paper-based informed consent has been the default in clinical trials for decades. But it comes with real costs. Incomplete consent forms are one of the most common causes of protocol deviations. A review of nearly 100 trials found that switching from paper to eConsent cut consent-related major and critical protocol deviations from 14% to just 6% — less than half.
Beyond compliance, the problem is deeper. Lengthy paper forms written in dense clinical language leave many participants confused about what they are agreeing to. That is not just an ethical concern — it slows enrollment, increases dropout, and creates downstream data quality problems.
eConsent changes the equation. It brings multimedia, interactive comprehension checks, remote signing, and real-time tracking into a process that has barely changed in 50 years.
I am Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where I work at the intersection of federated data platforms, regulatory compliance, and scalable clinical research infrastructure — areas that directly shape how electronic consent in clinical trials must be designed and governed. In this guide, I will walk you through everything you need to implement eConsent correctly, from regulatory requirements to platform selection and IRB submission.
Quick look at electronic consent clinical trials:
- clinical trial patient recruitment
- digital clinical trial recruitment
- remote clinical trial recruitment
Why Electronic Consent in Clinical Trials is Replacing Paper
The shift toward digital isn’t just about following tech trends; it’s about fixing a broken administrative process. In traditional trials, site staff often struggle with “paper fatigue.” When you are managing hundreds of participants across multiple sites, a missing date or a forgotten checkbox on page 12 of a 20-page document can trigger a major protocol deviation.
Industry data highlights the impact of this transition. According to a large-scale review of nearly 100 trials, the use of electronic consent clinical trials reduced major and critical consent-related protocol deviations from an average of 14% down to only 6%. This reduction is largely due to the “forced logic” of digital platforms — the system simply won’t let a participant submit the form until every required field is completed and every signature is captured.
The Financial Impact of Paper Errors
Beyond the regulatory headache, paper-based errors have a massive financial footprint. Every time a Clinical Research Associate (CRA) identifies a missing signature during a monitoring visit, it triggers a query. Resolving a single query can cost hundreds of dollars in administrative time, site staff effort, and potential re-consenting travel costs for the participant. By eliminating these “sloppy” errors at the point of entry, eConsent platforms pay for themselves through reduced monitoring and reconciliation costs.
Beyond error reduction, eConsent offers:
- Real-time Tracking: Sponsors and sites can see exactly who has viewed the document, who has signed, and who is stuck on a specific section.
- Multimedia Engagement: Instead of a wall of text, participants can watch videos explaining the procedure or use a glossary for complex medical terms.
- Dynamic Re-consenting: When a protocol is amended, digital systems can automatically notify participants and track their new signatures, ensuring no one falls out of compliance.
Effective Patient Consent Management is the foundation of modern research. It bridges the gap between recruitment and data collection, ensuring that the transition into the trial is smooth and legally sound. For a deeper dive into the nuances of digital vs. paper, you can explore this guide on Understanding Remote and Electronic Consent (e-Consent).
Improving Participant Comprehension and Retention
One of the biggest hurdles in clinical research is the “informed” part of informed consent. Many participants sign paper forms without fully grasping the risks or the time commitment involved. Electronic consent clinical trials solve this by using interactive elements.
Imagine a participant being able to click a “What is a biopsy?” link to see a 30-second animation, rather than reading a paragraph of jargon. Interactive quizzes can also be embedded to verify that the participant actually understands the study before they are allowed to sign. This isn’t just helpful for the patient; it’s a protective measure for the investigator. If a participant fails a comprehension check, the system can flag the investigator to have a more detailed conversation with that individual before proceeding.
By presenting information in layman’s terms and allowing for longitudinal engagement — where the participant can revisit the information at any time — we see much higher retention rates. When people understand what they are doing, they are less likely to drop out mid-trial. This is a core component of any Clinical Trial Patient Recruitment Complete Guide, as the quality of the initial consent often dictates the success of the entire enrollment funnel.
Streamlining Site and Sponsor Workflows
For the people running the trial, eConsent is a massive productivity booster. Automated alerts notify site staff the moment a consent is signed, allowing them to schedule the next visit immediately. Version control becomes effortless; instead of printing hundreds of new paper copies when a protocol changes, the sponsor pushes a digital update to all sites simultaneously.
This level of centralized oversight is essential for any Decentralized Clinical Trials Complete Guide. It provides audit-ready traceability that makes regulatory inspections far less stressful. Every action — from the time spent reading a page to the exact moment of the signature — is time-stamped and logged, creating a bulletproof audit trail. This transparency allows sponsors to identify high-performing sites and those that may need additional training on the consent process.
Navigating Regulatory Requirements: FDA 21 CFR Part 11 and Beyond
To use electronic consent clinical trials in a regulated environment, you must adhere to specific standards. In the United States, the primary regulations come from the FDA (21 CFR Part 11) and the Department of Health and Human Services (HHS) under 45 CFR Part 46.
The FDA’s Use of Electronic Informed Consent in Clinical Investigations provides the roadmap. The most critical requirement is that the electronic system must be “validated.” This means the sponsor must prove the system does what it claims to do, maintains data integrity, and ensures that electronic signatures are as legally binding as “wet ink” signatures.
Global Regulatory Variations: GDPR and EMA
While the FDA provides clear guidance for the US, global trials must also navigate the European Medicines Agency (EMA) and the General Data Protection Regulation (GDPR). The EMA emphasizes that the eConsent process must not disadvantage participants who lack digital literacy. Furthermore, GDPR requires that the “legal basis” for processing data is clearly explained during the consent process. This often means that eConsent platforms must have localized versions that meet the specific legal language requirements of each member state, including specific clauses on data withdrawal and the “right to be forgotten.”
Key regulatory pillars include:
- Data Integrity: Ensuring the consent document cannot be altered after signing. This is often achieved through cryptographic hashing.
- Identity Verification: Confirming the person signing the digital form is actually the participant (often via biometrics, unique codes, or video verification).
- HIPAA Compliance: Protecting the privacy and security of the participant’s protected health information (PHI) through encryption at rest and in transit.
Essential Features of a Compliant Electronic Consent Clinical Trials Platform
Not all digital signature tools are created equal. While a standard e-signature might work for a business contract, clinical trials require higher levels of security and documentation.
| Feature | Standard E-Signature (Non-Compliant) | Part 11 Compliant eConsent |
|---|---|---|
| Audit Trail | Basic (who signed) | Comprehensive (every click, view, and edit) |
| Identity Proofing | Email verification only | Multi-factor, IDs, or biometrics |
| Version Control | Manual | Automated with re-consent triggers |
| Storage | Standard cloud | Secure, encrypted, GxP-validated |
| Verification | Simple timestamp | Cryptographically linked timestamp |
An ideal platform for electronic consent clinical trials must include secure document storage and the ability to provide the participant with a copy of their signed consent in a format they can keep (such as a downloadable PDF or a printed copy). It should also support “Short Forms” for participants with limited English proficiency, ensuring that the digital experience is equitable across all demographics.
IRB and Ethics Committee Review Processes
Before you can send your first digital consent form, you need approval from an Institutional Review Board (IRB) or Ethics Committee. They don’t just review the text of your consent; they review the process.
When submitting to an IRB, you should provide:
- A link or “walkthrough” of the eConsent interface.
- Any multimedia files (videos, audio) that will be shown to participants.
- The plan for identity verification (e.g., will the site staff witness the signature via Zoom?).
- Documentation that the system is Part 11 compliant (often a vendor’s validation certificate).
As noted by the VUMC Human Research Protections Program, the investigator remains responsible for the consent process, even if it is automated. You must ensure that the digital version is an exact match for the IRB-approved language and that participants still have a way to ask questions to a qualified member of the research team. IRBs are increasingly looking for “usability testing” data to ensure that the digital interface doesn’t confuse elderly or vulnerable populations.
Implementing eConsent in Decentralized and Hybrid Trials
The rise of decentralized clinical trials (DCTs) has made electronic consent clinical trials a necessity rather than a luxury. When participants are recruited from across the country — or across the globe — you cannot wait for paper forms to be mailed back and forth.
Remote consent (or teleconsent) allows a clinician to walk a participant through the form via a video call, answering questions in real-time while the participant views the document on their own device. This setup is a cornerstone of any Decentralized Clinical Trials Software Guide 2025. For those operating in specific regions, resources like the Clinical Trials Ontario eConsent Guide offer localized best practices for maintaining compliance in remote settings.
Technical Architecture and Integration
For eConsent to be truly effective, it cannot exist in a vacuum. Modern trial infrastructure requires that the eConsent platform integrates seamlessly with other systems:
- EDC (Electronic Data Capture): Once a participant signs, their demographic data should automatically flow into the EDC to reduce manual entry errors.
- IRT (Interactive Response Technology): The signature should trigger the randomization process and the shipment of the first dose of the study drug.
- CTMS (Clinical Trial Management System): Real-time consent status allows project managers to track enrollment milestones across hundreds of sites globally.
Overcoming Barriers to Widespread Adoption
Despite the benefits, adoption hasn’t been universal. A TransCelerate survey found that many pharmaceutical developers still have limited experience with eConsent. Why?
- Digital Literacy and Inclusivity: There is a concern that older or less tech-savvy populations might struggle with digital forms. To combat this, platforms must be designed with high contrast, adjustable font sizes, and screen-reader compatibility (ADA compliance).
- Site Hesitation: Research sites are often set in their ways and wary of learning new software for every different study. Providing a single, intuitive interface that works across multiple trials is key to site buy-in.
- Initial Costs: Setting up a validated system requires an upfront investment in technology and training. However, the long-term ROI in terms of reduced protocol deviations and faster enrollment usually outweighs these costs.
The Hybrid Consent Model
The solution is often a hybrid consent model. This gives participants the choice: sign digitally on a tablet at the clinic, sign remotely from home, or stick with traditional paper if they prefer. This flexibility ensures that technology remains an enabler, not a barrier. As we move toward more AI-driven clinical trials, the interoperability of these consent systems with larger data platforms will become the standard, allowing for automated screening based on the specific permissions granted by the participant.
Frequently Asked Questions about eConsent
What is the difference between eConsent and remote consent?
While the terms are often used interchangeably, they refer to different things. eConsent is the technology (the digital form and signature). Remote consent is the location (obtaining consent when the participant is not at the site). You can do eConsent in person (e.g., using an iPad at the doctor’s office) or remotely. You can also do remote consent using paper (mailing forms), though this is increasingly rare. For more on reaching participants where they are, see our Digital Patient Recruitment Complete Guide.
Is eConsent mandatory for FDA-regulated trials?
No, it is not mandatory, but it is highly encouraged. The FDA provides regulatory flexibility, allowing sponsors to choose the method that best fits their study’s risk profile. However, if you choose to go digital for an FDA-regulated study, you must comply with 21 CFR Part 11. To learn more about the broader regulatory landscape, visit our Clinical Trials Complete Guide.
How does eConsent handle protocol amendments?
This is where digital systems shine. When a protocol changes, the system can automatically flag which participants need to “re-consent.” The new version is pushed to their devices, and they can review only the changes or the entire document, with their new signature tracked on a real-time dashboard. No more chasing down participants to sign “Version 2.1” months after the change was made.
Can eConsent be used for pediatric trials or vulnerable populations?
Yes, eConsent platforms are designed to handle complex consent workflows, including parental permission and child assent. For pediatric trials, the system can present a simplified, age-appropriate version of the information to the child while providing the full legal document to the parent. For vulnerable populations, such as those with cognitive impairments, the system can facilitate the involvement of a Legally Authorized Representative (LAR) through remote multi-party signing.
What happens if a participant loses internet access mid-consent?
Top-tier eConsent platforms include “offline mode” capabilities. If a participant is filling out the form at a site with poor connectivity, the data is saved locally on the device and encrypted. Once a connection is re-established, the system automatically syncs the data to the central server, ensuring no progress is lost and the audit trail remains intact.
How is data privacy maintained in the cloud?
Data privacy is maintained through a combination of technical and organizational measures. This includes end-to-end encryption, where only authorized personnel with specific decryption keys can view the participant’s identity. Furthermore, the use of federated data models ensures that the sensitive consent data remains within secure, localized environments while only the necessary metadata is shared with the sponsor for tracking purposes.
Conclusion: The Future of Digital Consent
The era of the 30-page paper consent form is coming to an end. As clinical trials become more complex and more global, the need for standardized, secure, and participant-friendly systems is undeniable. Electronic consent in clinical trials is the key to unlocking faster enrollment and higher data quality.
At Lifebit, we believe that the future of research lies in federated data ecosystems. By keeping data secure and bringing the analysis to the data, we enable global collaboration without compromising privacy. eConsent is the “front door” to this ecosystem — it is the moment trust is established between the participant and the researcher.
When implemented correctly, eConsent does more than just capture a signature; it empowers participants to make truly informed choices, reduces the administrative burden on sites, and provides sponsors with the transparency they need to bring life-saving treatments to market faster.
Ready to modernize your trial operations? Learn more about how Lifebit’s federated platform can secure your research data and streamline your global studies.


