Beyond the Regulations: Finding Your Ideal Pharmacovigilance Compliance Solution

Stop 50% Data Mismatches: How an AI Pharmacovigilance Compliance Solution Keeps You Audit-Ready
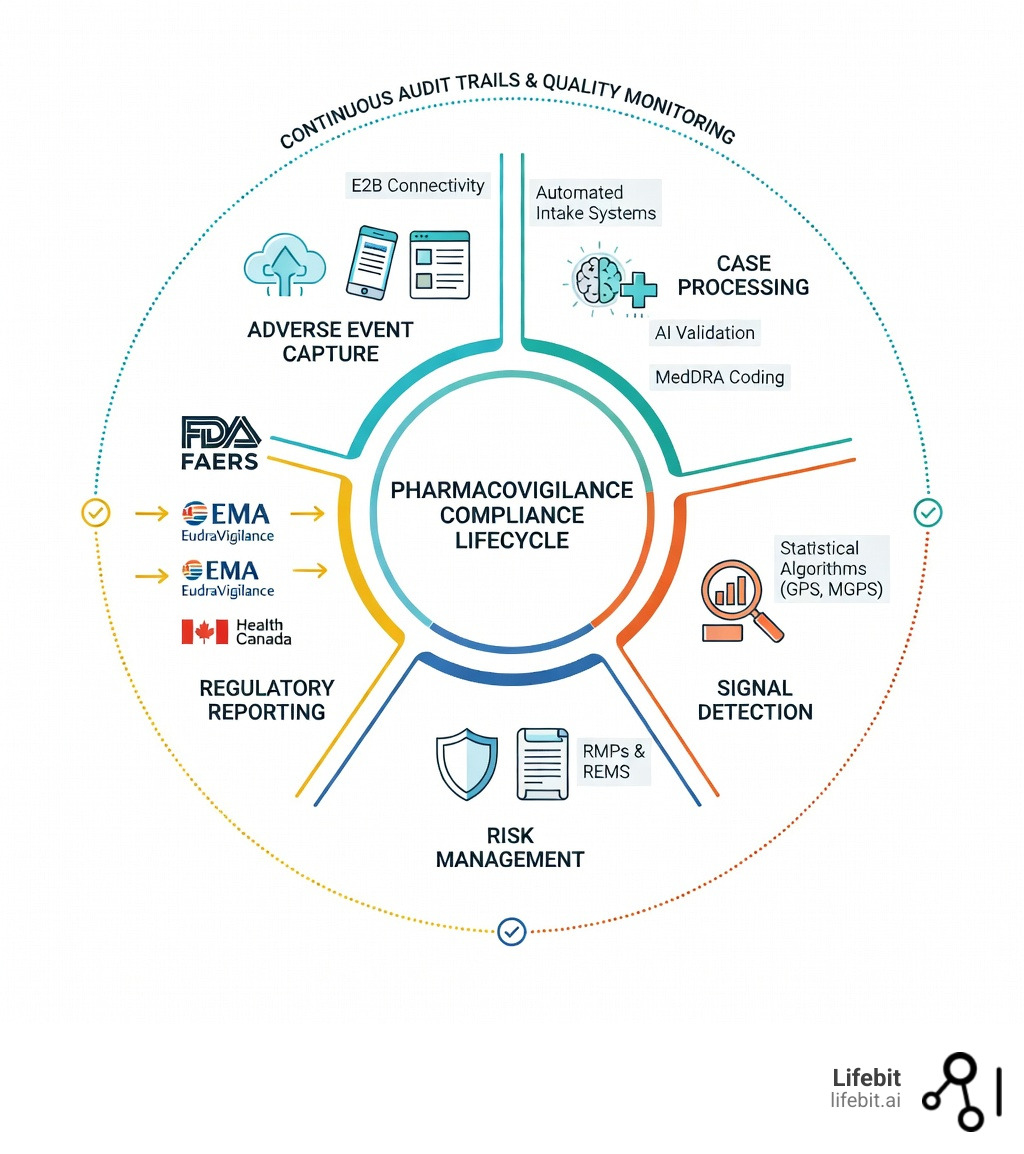
A pharmacovigilance compliance solution helps pharmaceutical and biotech companies monitor drug safety, detect adverse events, and meet global regulatory requirements like FDA, EMA, and Health Canada standards. The right solution combines automated case processing, real-time signal detection, and secure data management to protect patients while reducing compliance risk. In the modern era of “Pharmacovigilance 4.0,” the focus has shifted from reactive reporting to proactive risk management, where data integrity is the primary currency of trust between manufacturers and health authorities.
Key features to look for in a high-tier solution:
- Automated case intake and processing with E2B(R3) connectivity and MedDRA/WHODrug coding
- AI-powered signal detection using advanced statistical algorithms like GPS, MGPS, and PRR (Proportional Reporting Ratio)
- Global regulatory compliance across FDA FAERS, EMA EudraVigilance, and Health Canada
- Centralized data management with comprehensive audit trails, PSMF (Pharmacovigilance System Master File) repository, and quality monitoring
- Integration capabilities for literature review, medical information support, and REMS/RMP (Risk Evaluation and Mitigation Strategies) management
The stakes have never been higher. More than 10 million safety cases are processed annually across global pharmacovigilance systems, with over 70% of drug adverse event reports in FDA FAERS flowing through just a handful of proven platforms. Yet recent analysis of 5,000 new cases revealed a troubling reality: 50% contained data that didn’t match their unstructured narratives, and 10-20% of those cases were fundamentally incorrect. Manual processes, fragmented vendor relationships, and outdated technology are creating compliance gaps that put both patients and companies at risk. When a narrative describes a “severe myocardial infarction” but the structured data field only lists “chest pain,” the resulting signal detection is compromised, potentially delaying life-saving regulatory actions.
Today’s regulatory environment demands more than basic reporting. Health authorities like the FDA and EMA expect real-time surveillance, proactive risk management, and data integrity throughout the product lifecycle. Companies face growing adverse event volumes—often increasing by 15-20% year-over-year—stricter audit requirements, and evolving standards like GVP (Good Pharmacovigilance Practices) modules. These modules, particularly Module V on Risk Management and Module IX on Signal Management, require a level of data granularity that legacy systems simply cannot provide.
The solution isn’t just better software. It’s a complete change in how safety data is captured, validated, and analyzed. Modern pharmacovigilance compliance solutions use AI and machine learning to achieve 95% time savings in case review, 99% transcription accuracy across 130,000+ files, and near-touchless processing of adverse events from diverse sources including social media, patient support programs, and audio channels. They connect local expertise with centralized oversight, automate literature monitoring across regions, and integrate seamlessly with regulatory submissions platforms. By automating the mundane tasks of data entry and coding, safety professionals can focus on what truly matters: medical causality assessment and benefit-risk evaluation.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve built federated data platforms that power secure, compliant pharmacovigilance workflows across global health systems. Our work with public sector institutions and pharmaceutical organizations has shown me how the right pharmacovigilance compliance solution can turn regulatory burden into competitive advantage—enabling faster signal detection, stronger audit readiness, and ultimately, safer medicines for patients worldwide. In this guide, we will explore the technical and strategic components that define a world-class safety system.
Key pharmacovigilance compliance solution vocabulary:
Core Components of a Modern Pharmacovigilance Compliance Solution
To stay ahead of the curve, we must look beyond basic data entry. A robust pharmacovigilance compliance solution acts as the central nervous system of drug safety. It isn’t just about collecting Individual Case Safety Reports (ICSRs); it’s about ensuring that every piece of data—from a patient’s tweet to a clinical trial narrative—is captured, validated, and coded with surgical precision. The architecture of these systems must be designed to handle the “Three Vs” of modern data: Volume, Velocity, and Variety.

The Four Pillars of Safety Infrastructure
The “gold standard” for these solutions includes several non-negotiable pillars that ensure both regulatory adherence and operational efficiency:
-
Automated Case Intake and Triage: Manual entry is the enemy of accuracy and the primary driver of high operational costs. By using automated case intake, companies can manage multiple reporting formats—including XML, PDF, and even handwritten forms—and reduce the manual errors that lead to that 50% data mismatch rate. Advanced triage systems can automatically prioritize “serious” cases (e.g., death, life-threatening events, or hospitalization) to ensure they are processed within the strict 7-day or 15-day regulatory windows.
-
Standardized Coding and Interoperability: Integration with MedDRA (Medical Dictionary for Regulatory Activities) and WHODrug is essential for global interoperability. A modern solution uses AI to suggest the most appropriate Lowest Level Term (LLT) based on the narrative description, ensuring consistency across different case processors. Without this, your data is essentially “speaking a different language” than the regulatory authorities, leading to rejected submissions and potential safety signals being missed.
-
Data Integrity and Audit Trails: Every change, review, and submission must be time-stamped and traceable. This is the bedrock of digital quality processes that prevent erroneous releases and high-risk deviations. In the event of an inspection, the system must be able to produce a “reconstruction of events” for any given case, showing exactly who touched the data and what changes were made at every step of the workflow.
-
E2B(R3) Connectivity: This is the current technical standard for the electronic transmission of ICSRs. A modern solution must support the R3 standard to ensure seamless communication with the FDA, EMA, and Health Canada databases. This includes the ability to handle “acknowledgments” (ACKs) from regulatory systems, which confirm that a report has been successfully received and validated.
Navigating Global Regulatory Landscapes
Regulatory requirements are moving targets. In the USA, the FDA FAERS (FDA Adverse Event Reporting System) processes millions of reports, while in Europe, the EMA’s EudraVigilance and Good Pharmacovigilance Practices (GVP) Modules set a high bar for compliance. For those of us operating in Canada, Health Canada’s GVP inspections are becoming increasingly rigorous, focusing heavily on the quality of the Pharmacovigilance System Master File (PSMF).
A comprehensive pharmacovigilance compliance solution must provide a PSMF repository and real-time compliance monitoring. The PSMF is a legal requirement in the EU and other regions, serving as a detailed description of the pharmacovigilance system used by the marketing authorization holder. We’ve seen that the most successful companies don’t just “prepare” for audits; they live in a state of constant audit readiness. This involves maintaining regulatory intelligence across all regions—ensuring that if a regulation changes in Singapore, Brazil, or the UK, your system updates its validation rules accordingly.
Enhancing Signal Detection and Post-Market Surveillance
Signal detection is where “safety” meets “science.” It’s the process of looking at the noise of millions of reports to find the signal of a new side effect. Advanced platforms use sophisticated statistical algorithms like the Gamma Poisson Shrinker (GPS) and Multi-Item Gamma Poisson Shrinker (MGPS) to identify these signals early. These algorithms are designed to filter out “background noise”—side effects that are already known or common in the general population—to highlight truly novel safety concerns.
Modern surveillance also requires a cloud-native approach to handle the sheer volume of data. By moving to trusted regulatory spaces, we can integrate diverse data sources—including global literature reviews, social media monitoring, and real-world evidence (RWE)—to proactively mitigate risks before they become public health crises. This is especially critical for high-profile drug classes, such as GLP-1s or novel gene therapies, where real-time monitoring is essential to maintain public trust and ensure the long-term viability of the product.
Why AI and Automation are Non-Negotiable for Drug Safety
If you are still relying on human eyes to read every single case narrative, you are fighting a losing battle. The volume of safety data is growing exponentially, but our ability to hire and train safety professionals is not. This is where AI and Natural Language Processing (NLP) become life-savers. In the past, a safety scientist might spend 60% of their time on data entry and only 40% on medical analysis. AI flips this ratio, allowing experts to focus on high-value cognitive tasks.
The data speaks for itself regarding the impact of automation:
- 95% time savings in bot-to-human interactions during case review, where the AI pre-populates fields for human verification.
- 38.5% time savings even in complex human-to-human reviews by providing automated summaries and highlighting key clinical data points.
- 99% transcription accuracy across over 130,000 files processed in a single year, far exceeding the accuracy of manual data entry which is prone to fatigue-related errors.
AI doesn’t just work faster; it works smarter. It can perform “narrative reconciliation,” identifying when a doctor’s handwritten note says one thing but the structured data form says another. Research shows that 10% to 20% of mismatched cases are fundamentally incorrect, meaning the wrong drug or the wrong reaction was coded. AI flags these discrepancies instantly, allowing human experts to focus on the 20% of cases that actually require deep medical judgment, rather than the 80% that are routine and predictable.
The Power of Federated Governance in Safety Data
Furthermore, global regulatory collaboration is now powered by federated governance. This is a critical advancement for companies operating in multiple jurisdictions with conflicting data privacy laws. Federated governance allows us to analyze data across different borders—complying with GDPR in Europe, HIPAA in the USA, and PIPL in China—without moving sensitive patient data out of its home jurisdiction.
In a federated model, the AI algorithm travels to the data, rather than the data traveling to a central server. This “privacy-first” approach allows for global safety insights and signal detection across a much larger dataset than any single company could manage alone, all while maintaining the highest standards of data security. This is particularly vital for rare disease medications, where the number of cases in any single country may be too small to generate a statistically significant safety signal.
Centralized vs. Fragmented: Choosing the Right Partner Model
Many companies suffer from “vendor fatigue”—using one provider for medical information, another for case processing, and a third for local literature reviews in different countries. This fragmented model is a recipe for disaster. It leads to data silos, inconsistent reporting, and a nightmare during audits when you must prove oversight of multiple third parties. Regulators, particularly the EMA, are increasingly focusing on “Vendor Oversight” as a key component of GVP compliance.
We advocate for a centralized pharmacovigilance outsourcing model supported by a unified technology platform. By working with a single-vendor partner that offers secure global collaboration, you gain several strategic advantages:
- Uniformity and Standardization: Every ICSR is processed using the same Standard Operating Procedures (SOPs) and validation rules, regardless of where it originated. This eliminates the “drift” in data quality that often occurs when multiple vendors are involved.
- Efficiency and Speed: A single point of contact for your Qualified Person for Pharmacovigilance (QPPV) streamlines communication. When a safety crisis emerges, you don’t want to be calling five different vendors to aggregate data; you need a single source of truth.
- Local Expertise with Global Reach: A global partner can provide local safety officers (LSOs) in 100+ countries who understand the cultural and linguistic nuances of local reporting. For example, an LSO in Japan will understand the specific PMDA reporting requirements that a centralized team in the US might miss.
- Scalability and Flexibility: As your product moves from Phase II trials to global launch, a centralized solution scales with you. You can add new modules—such as social media monitoring or advanced signal analytics—without needing to rebuild your entire infrastructure or integrate new vendors.
The Role of the QPPV in a Centralized System
The Qualified Person for Pharmacovigilance (QPPV) holds legal liability for the safety of a company’s products in many regions. A centralized pharmacovigilance compliance solution empowers the QPPV by providing a real-time dashboard of the entire safety system’s health. They can monitor submission timelines, track open signals, and review the PSMF at the click of a button. This level of transparency is not just a convenience; it is a vital safeguard against the personal and corporate liability that comes with safety oversight.
Frequently Asked Questions about Pharmacovigilance Compliance
How does a pharmacovigilance compliance solution handle data mismatches?
The 50% mismatch rate between narratives and structured data is a major compliance risk. A modern pharmacovigilance compliance solution uses automated validation rules and NLP. When a case is entered, the system cross-references the narrative text against the MedDRA codes and structured fields. If the narrative mentions “acute kidney injury” but the structured field says “nausea,” the system flags this for immediate “narrative reconciliation” before the report is ever submitted to an agency. This prevents the submission of “dirty data” that can trigger regulatory inquiries.
What role does medical information support play in compliance?
Medical Information (MI) is often the “front door” for adverse events. A robust solution integrates MI with PV. This includes a centralized, multilingual call center capable of handling inquiries in 180+ languages. When a patient calls with a question and mentions a side effect, the system must automatically triage that call as a potential adverse event. Integration ensures that the MI specialist doesn’t just answer the patient’s question but also captures the necessary data for a valid ICSR, ensuring no safety data falls through the cracks.
How can companies prepare for remote GVP audits?
Remote audits have become the new norm since 2020. To prepare, companies should conduct a gap analysis and use “audit readiness” surveys. Your pharmacovigilance compliance solution should provide full traceability and digital documentation. If an inspector asks for the history of a specific signal from three years ago, you should be able to pull up the complete “chain of evidence”—including the initial report, the signal evaluation report, and the final regulatory action—in seconds. Having a “clean” system with no overdue reports or unaddressed signals is the best way to ensure a successful audit.
What is the difference between E2B(R2) and E2B(R3)?
E2B(R3) is the latest international standard for transmitting safety reports. Compared to the older R2 version, R3 allows for much more granular data, including better support for biological products, improved identification of the reporter, and more detailed clinical information. Most major health authorities now require R3, and using a solution that natively supports this format is essential for avoiding technical rejection of your safety submissions.
Can AI replace the medical review process?
No. AI is a tool to assist, not replace, the medical reviewer. While AI can handle data entry, coding, and initial triage, the final “Causality Assessment”—determining if the drug actually caused the event—requires deep medical expertise and clinical judgment. The goal of an AI-powered pharmacovigilance compliance solution is to remove the administrative burden so that medical doctors can spend their time on these critical safety evaluations.
Conclusion: The Future is Federated and Real-Time
Finding the ideal pharmacovigilance compliance solution is no longer just a “check-the-box” exercise for the regulatory department. It is a strategic imperative that impacts your company’s reputation, its bottom line, and most importantly, the lives of the patients you serve. As we move toward a future of personalized medicine and complex biologics, the volume of safety data will only continue to grow. Companies that cling to manual processes will find themselves buried under a mountain of data, while those that embrace AI and automation will thrive.
At Lifebit, we believe the future of drug safety lies in federated AI. Our platform enables secure, real-time access to global biomedical and multi-omic data without compromising privacy. By using our Trusted Research Environment (TRE) and R.E.A.L. (Real-time Evidence & Analytics Layer), biopharma companies can move away from reactive reporting and toward proactive, AI-driven safety surveillance. This allows for the detection of safety signals in specific patient subpopulations that would be invisible in traditional, aggregated datasets.
Whether you are navigating Health Canada GVP inspections, preparing for an FDA audit, or scaling a global launch across five continents, the right technology partner makes all the difference. Don’t let your safety data sit in silos where it can’t be analyzed or protected. Secure your pharmacovigilance data with Lifebit and join the ranks of the world’s most innovative life sciences organizations. The transition to a modern, AI-powered compliance solution is the single most important step you can take to ensure the long-term safety and success of your therapeutic portfolio.

