Data Harmonization Methods: Your Guide to Seamless Integration

Stop Wasting 67% of Tech Resources on Fragmented Data
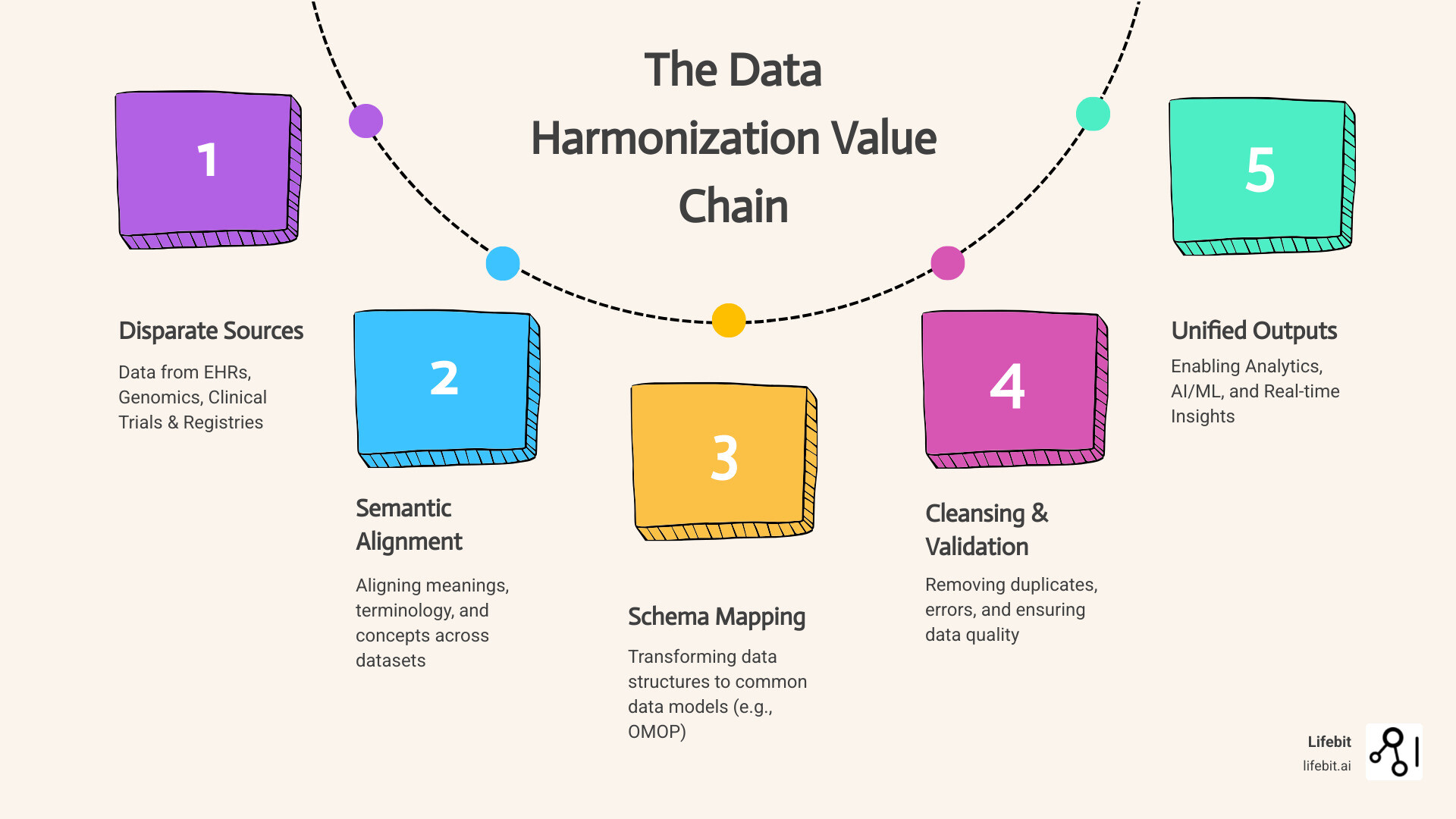
Data harmonization methods are systematic approaches to combining data from different sources, formats, and systems into a unified, consistent dataset that enables meaningful analysis and decision-making. Organizations today collect massive volumes of data from electronic health records, clinical trials, genomics platforms, and patient registries—but when this data sits in silos using different formats, terminologies, and standards, it becomes nearly impossible to extract actionable insights.
Quick Overview: Essential Data Harmonization Methods
- Semantic harmonization – Aligning meanings and terminology across datasets (e.g., mapping “myocardial infarction,” “heart attack,” and ICD-10 code I21 to one concept)
- Schema mapping – Changing data structures to match a common data model like OMOP or PCORnet
- Statistical harmonization – Correcting measurement differences and batch effects across platforms
- Data cleansing – Removing duplicates, errors, and inconsistencies before integration
- Record linkage – Merging patient or entity records across disparate datasets
Imagine researchers at an international conference presenting temperature data—one in Celsius, another in Fahrenheit, a third in Kelvin. They’re all measuring the same thing, but comparison is impossible. This is the daily reality for organizations trying to analyze fragmented data. The cost is staggering: data scientists spend more time cleaning than analyzing, tech resources waste 67% of their capacity on data quality tasks, and executives make decisions based on incomplete information.
Data harmonization changes this dynamic. Modern unification solutions free up over 67% of tech resources, slash analysis time, and create a single source of truth that enables real-time insights, early disruption detection, and confident decision-making. For pharma and public health leaders working with millions of patient records across federated environments, harmonization isn’t optional—it’s the foundation for AI-powered evidence generation, pharmacovigilance, and compliant multi-site research.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over 15 years developing data harmonization methods for genomics and biomedical data that enable secure, federated analysis across diverse healthcare datasets. My background in computational biology and AI has shown me how proper harmonization transforms fragmented data into powerful analytical assets that drive drug findy and precision medicine.
Data harmonization methods word guide:
Data Harmonization Methods: Turn Messy Data Into Actionable Insights
In the modern landscape of clinical research and biopharma, we are drowning in data but starving for information. Data harmonization refers to all efforts to combine data from different studies, ensuring that disparate fields, formats, and dimensions speak the same language.
Why is this essential? Because raw data is messy. A survey might record age as a specific date of birth, while a clinical registry might bucket patients into “young adults” (18–22) or “adults” (18–30). Without data harmonization methods, you can’t accurately measure disease incidence across these two groups. You’re essentially trying to build a puzzle where the pieces come from ten different boxes.
The primary benefits include:
- Increased Statistical Power: By pooling datasets (e.g., combining eight UK cohort studies to examine physical capability), we gain the sample size needed for robust findings.
- Business Agility: Harmonized data allows organizations to detect market disruptions or drug safety signals early.
- Confidence and Trust: When data is cleansed and reconciled, stakeholders trust the insights. Executives no longer have to ask, “Which version of this report is correct?”
- Operational Efficiency: Modern solutions free up tech resources by over 67%, allowing your team to focus on innovation rather than data wrangling.
Key Differences: Harmonization vs. Integration vs. Standardization
It is common to use these terms interchangeably, but in high-stakes research, the nuances matter.
- Data Integration is the physical or virtual “plumbing.” It’s the process of connecting sources (like EHRs and genomics platforms) so they can be accessed in one place.
- Data Standardization is about the “rules.” It applies uniform formats, such as ensuring all dates follow the YYYY-MM-DD format or that all measurements use the metric system.
- Data Harmonization is about the “meaning.” It goes a step further by ensuring semantic alignment. It reconciles different conceptual definitions so that a “heart attack” in one system is recognized as identical to “myocardial infarction” in another.
| Feature | Data Integration | Data Standardization | Data Harmonization |
|---|---|---|---|
| Primary Goal | Unified access to sources | Uniform data formats | Semantic alignment of meaning |
| Focus | Connectivity/Plumbing | Formatting/Syntax | Context/Interpretation |
| Typical Tool | ETL/Data Virtualization | Data Quality Rules | Common Data Models (CDMs) |
| Outcome | Data is in one place | Data looks the same | Data means the same thing |
To learn more about these distinctions, see our guides on health data standardization and clinical data integration.
The Role of Data Harmonization Methods in Master Data Management (MDM)
Master Data Management (MDM) is the discipline of managing an organization’s “core” data (patients, products, sites) to provide a single source of truth. Data harmonization methods are the engine inside the MDM vehicle.
Without harmonization, MDM is just a collection of links. Harmonization provides the metadata alignment necessary to ensure that when a clinician queries a patient’s history across five different hospitals, the system understands those records belong to the same individual. This is particularly critical when addressing big data challenges for scientific data infrastructure, where the sheer volume and variety of data can quickly lead to “data swamps” if governance and harmonization aren’t prioritized.
For biopharma, this relationship is vital for pharmacovigilance and target identification.
5 Steps to Harmonize Data and Slash Analysis Time
Successfully harmonizing data isn’t a “one-and-done” task; it’s a lifecycle. We recommend following a structured workflow to ensure accuracy and reproducibility.
Step 1: Data Findy and Selection
You can’t harmonize what you don’t know you have. This step involves a full inventory of your data sources—ERPs, CRMs, electronic health records (EHRs), and genomic platforms. We profile the data to understand its quality, granularity, and “quirks.”
Step 2: Metadata Alignment and CDM Definition
Here, we define the “Target Language.” For healthcare, this usually means selecting a Common Data Model (CDM) like OMOP, PCORnet, or i2b2. By using established standards, you save months of work and ensure your data is interoperable with global research networks.
Step 3: Schema Mapping
In this phase, we create “crosswalks.” We map source fields to the target schema. For example, mapping “PatientID” in Source A and “SubjectNum” in Source B to the “person_id” field in an OMOP CDM.
Step 4: Data Change and Processing
This is where the actual “heavy lifting” happens. Using ETL (Extract, Transform, Load) or data virtualization, we convert incompatible formats, handle missing values, and remove duplicates. This step often uses the Maelstrom research guidelines for rigorous retrospective data harmonization, which provide a roadmap for standardizing variables across large cohort studies.
Step 5: Multi-layered Validation
Finally, we test the results. Validation should include:
- Technical checks: Did all records migrate?
- Business logic: Are blood pressure readings within a realistic range?
- Semantic checks: Does “Stage 4 Cancer” in the source still mean “Stage 4 Cancer” in the harmonized dataset?
Scale AI Research With Advanced Data Harmonization Methods
AI is only as good as the data it eats. If you feed a machine learning model unharmonized data, the “garbage in, garbage out” rule applies. In fact, scientific research on big data systems meeting machine learning challenges highlights that data heterogeneity is one of the primary roadblocks to scalable AI in science.
Multi-omics and Pathogen Genomics
In fields like genomics, harmonization is even more complex. You aren’t just dealing with text; you’re dealing with “batch effects”—technical noise introduced because samples were processed on different days or using different sequencing machines. Advanced data harmonization methods, such as the ComBat algorithm or deep learning-based confound removal, are used to “unlearn” these biases.
Tools like The DataHarmonizer tool for pathogen genomics have emerged to provide faster validation and aggregation of contextual information, which was critical during the COVID-19 pandemic for tracking variants across borders.
Federated Learning: Harmonization Without Data Movement
At Lifebit, we specialize in federated data harmonization. Often, sensitive biomedical data cannot be moved due to GDPR or HIPAA regulations. Our platform allows organizations to harmonize data locally and then perform federated analytics. This means the AI model travels to the data, learns from the harmonized local dataset, and only shares the insights—never the patient-level data.
Prospective vs. Retrospective Data Harmonization Methods
When should you start harmonizing?
- Prospective Harmonization (Ex-ante): This is the “gold standard.” It happens before data collection. Researchers agree on identical protocols, questionnaires, and measurement tools. For example, guidelines for cleaning and harmonization of generation and gender survey data emphasize planning from the outset to ensure cross-national comparability.
- Retrospective Harmonization (Ex-post): This is the most common method. It involves taking existing (and often inconsistent) datasets and mapping them to a common format. While more challenging, it allows for the re-use of massive amounts of historical data for new findies.
Choosing the Right Data Harmonization Methods for Decision-Making
To transform data into an analytical asset, you must choose methods that support your specific goals. If your goal is real-time evidence generation, you might prioritize automated, AI-driven mapping. If your goal is regulatory submission, you might prioritize manual oversight and rigorous clinical data governance.
More info about secure data collaboration can help you steer the complexities of working with confidential data while ensuring your harmonization methods meet the highest security standards.
Avoid These 4 Mistakes Killing Your Data Harmonization Strategy
Harmonization is hard work. Anyone who says otherwise has never tried to merge three different legacy EHR systems. Common challenges include:
- Data Silos: Departments often guard their data, fearing a loss of control.
- Incompatible Formats: Mixing structured SQL data with unstructured free-text notes.
- Privacy Concerns: Harmonizing sensitive data across borders requires secure clinical data solutions.
- Semantic Interoperability: Ensuring that “Value A” always equals “Value A” across ten different languages and cultures.
Best Practices for Success
- Follow the FAIR Principles: Ensure your data is Findable, Accessible, Interoperable, and Reusable. The FAIR guiding principles for scientific data management are the foundation of modern data stewardship.
- Involve Domain Experts Early: Don’t let the IT team harmonize clinical data in a vacuum. You need clinicians and data scientists who understand the context of the measurements.
- Use a Trusted Research Environment (TRE): A secure TRE provides a safe space for researchers to access harmonized data without the risk of data breaches.
- Automate with Oversight: Use AI to handle repetitive mapping tasks, but always keep a human in the loop for complex semantic decisions.
- Start Small: Begin with a pilot project—harmonize two critical datasets first, prove the ROI (like that 67% resource saving), and then scale.
Data Harmonization Methods: Your Top Questions Answered
How do data harmonization methods improve decision-making?
By creating a “single source of truth,” these methods ensure that executives and researchers are looking at the same high-quality information. This eliminates the need to reconcile conflicting reports and allows for real-time insights into trends, anomalies, and opportunities.
What are the risks of not harmonizing business data?
The risks are significant: wasted technical resources (up to 67%), delayed research findies, incorrect clinical insights, and a lack of competitiveness for grants or market share. In biopharma, unharmonized data can lead to missed safety signals in pharmacovigilance.
What tools support large-scale data harmonization?
Large-scale efforts often use a combination of OMOP common data models, federated data platforms, and specialized R pipelines (like multiomics). Platforms like Lifebit provide built-in harmonization layers that automate much of the schema mapping and validation process.
Start Your Data-Driven Future With Lifebit Today
Data is the lifeblood of modern innovation, but only if it’s usable. Data harmonization methods are the bridge between fragmented chaos and actionable intelligence. By standardizing formats, aligning meanings, and leveraging advanced AI-driven techniques, organizations can finally open up the full value of their data ecosystems.
At Lifebit, we believe that the most important research shouldn’t be held back by data silos. Our federated AI platform is built to handle the heaviest harmonization lifts, providing secure, real-time access to global biomedical and multi-omic data. Whether you’re a government agency tracking public health trends or a biopharma company accelerating drug findy, we’re here to help you turn your data into your greatest asset.
Ready to transform your fragmented data into a powerful analytical asset? Find Lifebit’s Real-time Evidence & Analytics Layer and take the first step toward your data-driven future.

