How to Accelerate Clinical Research Timelines: Strategies for Success

Why Speed Matters in Clinical Research Today
Accelerated clinical research is changing how quickly life-saving treatments reach patients. When the COVID-19 pandemic hit, countries with streamlined clinical trial processes developed vaccines in record time—while others struggled. This gap revealed a critical truth: the speed of clinical research directly impacts patient outcomes and public health. The pandemic served as a global stress test, proving that the traditional 10-to-12-year drug development cycle could be compressed into months when regulatory barriers were lowered and technology was leveraged effectively. For instance, the UK’s RECOVERY trial and the WHO’s Solidarity trial demonstrated how large-scale, adaptive platform trials could provide definitive answers on treatment efficacy in a fraction of the usual time.
Key approaches to accelerate clinical research include:
- Regulatory pathways: FDA Accelerated Approval allows drug approval based on surrogate endpoints for serious conditions, providing earlier access to life-saving therapies.
- Infrastructure initiatives: ACT Canada ($39M investment) and ACT EU (targeting 500 more multinational trials by 2030) streamline trial conduct through centralized coordination.
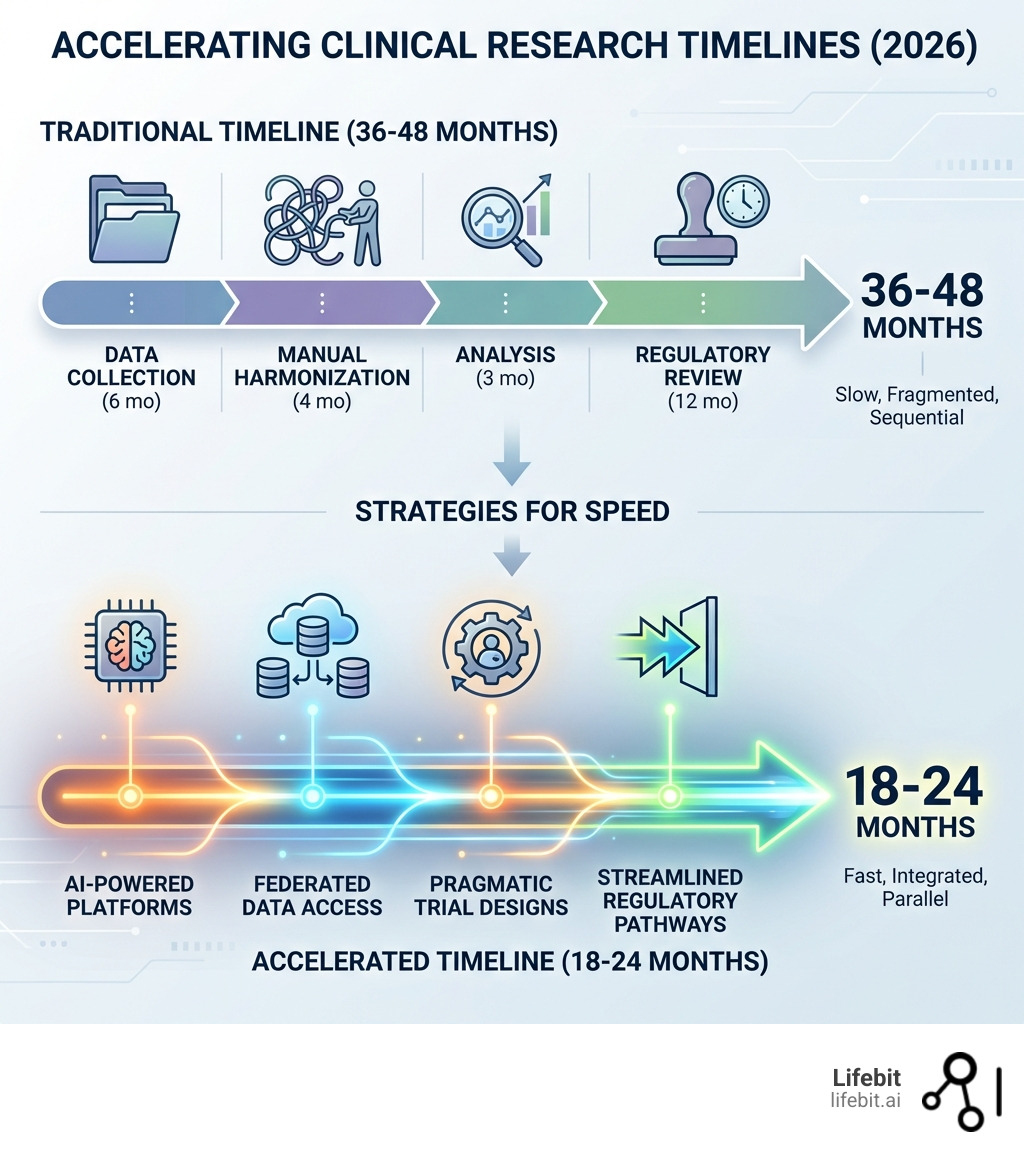
- Technology platforms: AI-powered systems reduce “time-to-insight” by automating data aggregation and harmonization across disparate global sources.
- Pragmatic trial designs: FDA’s C3TI STEP project integrates trials into routine clinical practice, reducing the burden on both patients and providers.
- Global collaboration: TransCelerate BioPharma’s 570+ tools and 19 member companies drive R&D change by standardizing data sharing and operational protocols.
The bottleneck isn’t just regulatory—it’s operational. Research teams spend months manually aggregating data from Electronic Medical Records (EMRs), vendor archives, and patient reports. Simple questions like “Which patients with KRAS mutations had exceptional drug responses?” can take months to answer because the data is trapped in silos, formatted differently, and governed by varying privacy laws. This delay throttles scientific progress and increases the cost of drug development, which is currently estimated at $2.6 billion per successful drug. Every day a trial is delayed represents not just a financial loss for the sponsor, but a day that patients go without potentially life-altering treatment.
Today’s successful research organizations are tackling these challenges head-on. Canada’s ACT consortium brings together 28 networks and 11 trial units with $55M in funding. The EU aims for 66% of trials to start recruitment within 200 days. The FDA’s QCARD initiative standardizes Real World Data submissions for faster review. These initiatives are not just about working harder; they are about working smarter by building a digital infrastructure that supports rapid evidence generation.
As Dr. Maria Chatzou Dunford, CEO of Lifebit, I’ve spent 15 years building platforms that accelerate clinical research through federated data analysis and AI-powered workflows. Throughout my career in computational biology and biomedical data integration, I’ve seen how the right technology can transform months of data wrangling into hours of insight generation. Let me show you the proven strategies that are cutting clinical research timelines across the globe.
Common Accelerated clinical research vocab:
- AI healthcare UK
- AI powered diagnostics
- What are the top biotech companies in London that focus on data analytics?
Leveraging Global Initiatives for Accelerated Clinical Research
To achieve Accelerated clinical research, we must look beyond individual trial sites and accept large-scale national and international consortia. These initiatives are designed to dismantle the silos that have historically slowed down drug development. In Canada and Europe, new frameworks are emerging to ensure that high-quality, high-impact randomized controlled trials (RCTs) are conducted more efficiently. These frameworks focus on “trial readiness,” ensuring that when a new molecule is ready for testing, the infrastructure to recruit, monitor, and analyze patients is already in place.
In Canada, the Canadian Institutes of Health Research (CIHR) committed $39 million over three years to the Accelerating Clinical Trials (ACT) consortium. This pan-Canadian initiative addresses a hard truth: while Canada has world-leading clinical trialists, the country has lacked the streamlined processes seen in other nations. The COVID-19 pandemic served as a wake-up call, prompting Canadian researchers to ask why other countries were breaking ground on vaccines and treatments at record speeds while domestic efforts faced bureaucratic problems. The ACT consortium aims to solve this by creating a national network that can launch a trial across multiple provinces simultaneously, bypassing the need for redundant ethics reviews and contract negotiations.
The ACT Canada consortium brings together hundreds of researchers from 28 networks and 11 trial units. By focusing on “trial readiness” and system-level solutions, ACT aims to make clinical research more responsive and resilient. We see a similar push in Europe with the Accelerating Clinical Trials in the EU (ACT EU) initiative. This program is a collaboration between the European Commission, the Heads of Medicines Agencies (HMA), and the European Medicines Agency (EMA). It represents a fundamental shift in how Europe manages clinical trials, moving away from a fragmented, country-by-country approach toward a unified, pan-European ecosystem.
The ACT EU Vision for Faster Recruitment
The primary goal of ACT EU is to transform the European Union into a global leader for clinical trial development. They aren’t just aiming for incremental improvements; they have set bold, measurable targets that are tracked through the Clinical Trials Information System (CTIS). The CTIS serves as a single entry point for submitting clinical trial applications in the EU and the European Economic Area (EEA), replacing the old EudraCT database and streamlining the authorization process.
Key targets for ACT EU include:
- 500 more multi-national trials authorized by 2030, specifically focusing on large-scale trials that can provide robust evidence for rare diseases and pediatric populations.
- 66% of trials to start recruitment in under 200 days from the initial application submission, a significant reduction from the current average which can often exceed a year.
- Implementation of the 10 Priority Actions, which include developing a multi-stakeholder platform for trial design, modernizing Good Clinical Practice (GCP) guidelines, and establishing a dashboard to monitor the clinical trial environment in real-time.
To reach these goals, ACT EU focuses on harmonisation. By aligning regulatory, technological, and process innovations across member states, the initiative reduces the friction of cross-border collaboration. For sponsors, this means a more predictable environment where “smarter” clinical trials can be launched without navigating 27 different sets of rules.
Canada’s National Strategy for Trial Readiness
In Canada, the RI-MUHC (Research Institute of the McGill University Health Centre) plays a leadership role within the national ACT Canada Consortium. In early 2023, RI-MUHC researchers were awarded a total of $55 million for ACT consortium projects. Their specific unit, the ACT-CTU, is a prime example of how local infrastructure supports national Accelerated clinical research goals. The focus here is on “Team Science,” where specialists in data management, ethics, and patient engagement work alongside clinicians to ensure trials are designed for success from day one.
The ACT-CTU initiatives include:
- Shared Staffing: Hiring methodologists and regulatory specialists to assist trialists in navigating complex design and approval phases, ensuring that protocols meet both scientific and regulatory standards.
- Knowledge Sharing: Hosting clinical trial rounds and protocol review sessions to improve trial quality before submission, which reduces the likelihood of regulatory “holds” or requests for clarification.
- Financial Support: Providing seed funding for internal pilot clinical trials that serve as “flagship studies,” allowing researchers to gather the preliminary data needed to secure larger international grants.
- Equity and Diversity: Actively fostering more diverse recruitment sites to ensure trial results are representative of the broader population, which is increasingly a requirement for FDA and EMA approval.
By leveraging these units, Canada is building a resilient research network that can pivot quickly to address emerging health threats while maintaining the highest standards of patient safety and data integrity.
Navigating FDA Pathways for Faster Market Entry
While infrastructure initiatives improve how trials are run, regulatory pathways determine how quickly they are approved. The FDA’s Accelerated Approval program is perhaps the most well-known mechanism for speeding up access to treatments for serious or life-threatening conditions. Established in 1992 in response to the HIV/AIDS crisis, this pathway was designed to allow drugs for serious conditions that filled an unmet medical need to be approved based on a surrogate endpoint.
This program allows the FDA to grant approval based on a surrogate endpoint—a marker like a blood test, an imaging scan, or a specific protein level—that is “reasonably likely” to predict a clinical benefit. For example, in oncology, a drug might be approved because it shrinks a tumor (the surrogate endpoint), even if it hasn’t yet been proven to extend the patient’s overall life (the clinical benefit). This is crucial because waiting for overall survival data can take years, during which time patients may have no other treatment options.
However, Accelerated clinical research under this pathway comes with strings attached. Sponsors must conduct confirmatory trials post-approval to verify the predicted benefit. If these trials fail to show a clinical benefit, or if the sponsor fails to conduct them with “due diligence,” the FDA has the authority to withdraw the drug from the market using expedited procedures. The Food and Drug Omnibus Reform Act (FDORA), part of the Consolidated Appropriations Act of 2023, recently strengthened these requirements. The FDA can now require that confirmatory trials be underway at the time of the accelerated approval, preventing the “dangling approval” phenomenon where drugs remain on the market for years without proven benefit.
Streamlining Trials with the C3TI STEP Project
The FDA isn’t just focused on approval pathways; they are also modernizing trial design through the Center for Clinical Trial Innovation (C3TI). A key initiative here is the Streamlined Trials Embedded in clinical Practice (STEP) Demonstration Project. This project aims to bridge the gap between clinical research and clinical care, making trials more accessible to a broader range of patients.
The STEP project encourages “pragmatic” trial designs. Instead of rigid, highly controlled research environments that often exclude patients with comorbidities, these trials are integrated into routine clinical practice. This approach allows for the collection of data in a real-world setting, which can be more representative of how the drug will perform once it is widely available.
Benefits for sponsors participating in STEP include:
- Improved CDER Engagement: Direct access to FDA leaders and subject matter experts to resolve design issues early in the process.
- Quality by Design: A fit-for-purpose inspection process that focuses on the most critical data elements rather than administrative minutiae.
- Efficiency: These trials often attract broader study populations and can be completed more rapidly because they use real-world data and decentralized procedures, such as remote monitoring and local lab testing.
Modernizing Data Collection for Accelerated Clinical Research
Data is the lifeblood of Accelerated clinical research, but poor data quality is its greatest enemy. The FDA’s Oncology QCARD (Quality, Characterization, and Assessment of Real-world Data) initiative aims to fix this. By establishing essential data elements for Real World Data (RWD) and Real World Evidence (RWE), QCARD facilitates more effective communication between sponsors and protocol reviewers. This is particularly important as more sponsors look to use RWD from electronic health records (EHRs) to support supplemental indications for already approved drugs.
When we use RWD—such as insurance claims or pharmacy records—to support a drug submission, the FDA needs to know that the data is reliable, consistent, and free from bias. Initiatives like QCARD provide a standardized approach, ensuring that early RWD studies in oncology are high-quality and review-ready. This reduces the “back-and-forth” between regulators and sponsors, shaving months off the approval timeline and allowing for more agile drug development cycles. Furthermore, the FDA’s Project Optimus is another example of modernization, focusing on dose optimization in oncology to ensure that the “maximum tolerated dose” is not automatically assumed to be the best dose, thereby reducing toxicity and improving trial completion rates.
Solving the Time-to-Insight Bottleneck with AI and Data
The most significant delay in clinical research today is what we call the “time-to-insight” bottleneck. This is the period between collecting raw data and actually understanding what that data means. Currently, this process is labor-intensive and manual. Research teams spend countless hours aggregating, harmonizing, and curating multi-modal data from disparate sources like Electronic Medical Records (EMRs), molecular archives, and patient-reported outcomes. In many cases, data scientists spend 80% of their time cleaning data and only 20% analyzing it. To accelerate clinical research, this ratio must be flipped.
This is where organizations like TransCelerate BioPharma come in. TransCelerate is a non-profit collaboration of 19 leading biopharma companies dedicated to simplifying and accelerating R&D. They have developed over 570 solutions and tools to address critical drug development issues. Their approach focuses on ecosystem-wide collaboration, ensuring that data standards are consistent across the industry. For example, their Common Protocol Template (CPT) helps sponsors create more consistent and machine-readable protocols, which in turn speeds up site activation and regulatory review.
At Lifebit, we take this a step further by using AI-powered platforms to automate the data wrangling process. Our federated AI platform allows researchers to access global biomedical and multi-omic data in real-time without moving the data from its secure location. This “code-to-data” approach is essential for maintaining data privacy and complying with strict regulations like GDPR and HIPAA, as it eliminates the need to transfer sensitive patient information across borders.
Reducing Manual Data Wrangling in Accelerated Clinical Research
To truly achieve Accelerated clinical research, we must move away from manual data processing and embrace automated, scalable solutions. Our platform’s capabilities include:
- Automated Harmonization: AI algorithms that map diverse data types to common standards like the OMOP (Observational Medical Outcomes Partnership) Common Data Model. This turns messy, unstructured EMR data into research-ready datasets that can be queried across different hospital systems.
- Trusted Research Environments (TREs): Secure, cloud-based workspaces where researchers can collaborate on sensitive data while maintaining strict federated governance. TREs provide the tools and computing power needed for complex analysis without compromising data security.
- Multi-omic Integration: Combining genomic, proteomic, and clinical data to provide a holistic view of patient health. This allows researchers to identify specific biomarkers that predict drug response, enabling the design of smaller, more targeted trials that reach their endpoints faster.
By reducing the manual steps in data curation, we can cut the time-to-insight from months to days. This allows research teams to be more agile, pivoting their strategies based on real-time evidence rather than waiting for quarterly reports. For instance, if an AI model identifies a safety signal in a specific patient subgroup, the trial can be adjusted immediately, protecting patient safety and saving resources.
Collaborative Frameworks for R&D Change
Collaboration is the secret sauce of Accelerated clinical research. TransCelerate’s initiatives have shown that when biopharma companies, health authorities, and technology providers work together, they can solve problems that are too big for any one organization. Their work on the “Digital Data Flow” initiative aims to create an end-to-end automated data pipeline from the clinical trial site to the regulatory submission, potentially saving thousands of hours of manual data entry.
For instance, TransCelerate’s research on improving clinical trial data collection—published in DIA’s Therapeutic Innovation & Regulatory Science—highlights how standardized tools can make patient participation more accessible and meaningful. By adopting these industry-wide standards, we ensure that the tools developed today will work for the trials of tomorrow, regardless of the sponsor or the site. This interoperability is the foundation of a modern, accelerated research ecosystem.
Frequently Asked Questions about Accelerated Clinical Research
What is the primary difference between ACT Canada, ACT EU, and FDA Accelerated Approval?
While all three aim to speed up the delivery of treatments, they operate at different levels of the research ecosystem:
- ACT Canada and ACT EU are infrastructure and policy initiatives. They focus on improving the “plumbing” of clinical trials—how they are funded, how sites are coordinated, and how recruitment is managed across borders. They aim to make the entire system more efficient.
- FDA Accelerated Approval is a regulatory pathway. It provides a specific legal mechanism to approve a drug earlier based on preliminary data (surrogate endpoints), provided the sponsor confirms the benefit later. It is a tool used at the end of the research process to get the drug to market faster.
How does the FDA’s C3TI STEP project benefit trial sponsors?
The STEP project is a game-changer for sponsors who want to use innovative trial designs. It provides “concierge-level” support from the FDA’s Center for Drug Evaluation and Research (CDER). Sponsors get help resolving issues with pragmatic trial designs, access to decentralized trial experts, and a streamlined inspection process that rewards “quality by design.” This means that if a sponsor builds high-quality data collection into their trial from the start, the FDA will focus its oversight on the most critical areas, reducing the administrative burden on the sponsor.
What role does AI play in shortening clinical trial timelines?
AI is the primary engine for reducing “time-to-insight.” In Accelerated clinical research, AI is used to:
- Automate Recruitment: Scanning EMRs to find eligible patients in seconds, rather than having site staff manually review charts.
- Harmonize Data: Using machine learning to clean and standardize multi-modal data, ensuring that data from different sources can be analyzed together.
- Predict Outcomes: Using AI models to identify which patients are most likely to respond to a treatment, allowing for smaller, more targeted (and faster) trials.
- Real-time Surveillance: Monitoring safety data as it comes in, allowing for immediate intervention if a safety issue is detected.
What are Decentralized Clinical Trials (DCTs) and how do they help?
Decentralized Clinical Trials (DCTs) use technology to bring the trial to the patient, rather than requiring the patient to travel to a central research site. This can include remote monitoring via wearable devices, tele-health visits, and local pharmacy drop-offs for medication. DCTs accelerate research by increasing patient recruitment and retention, as they reduce the travel burden on participants. They also allow for more diverse populations to participate, as patients who live far from major academic medical centers can still be included.
How is data privacy maintained in accelerated research?
Data privacy is maintained through technologies like Federated Analysis and Trusted Research Environments (TREs). Instead of moving sensitive patient data to a central server, researchers send their analysis code to where the data resides. Only the aggregated, non-identifiable results are sent back. This ensures that the data remains behind the firewalls of the hospital or research center, complying with regulations like GDPR and HIPAA while still allowing for large-scale, multi-national research.
Conclusion: Building a Resilient Research Ecosystem
The future of Accelerated clinical research depends on our ability to build a resilient, connected, and data-driven ecosystem. The days of siloed research and manual data entry are numbered. As we have seen through initiatives like ACT Canada, ACT EU, and the FDA’s C3TI, the trend is toward integration—integrating trials into clinical practice, integrating data across borders, and integrating AI into every step of the research lifecycle. This shift is not just a technical necessity; it is a moral imperative to ensure that medical innovation keeps pace with human need.
At Lifebit, we are proud to be at the forefront of this change. Our next-generation federated AI platform provides the secure, real-time access to global data that modern research demands. By utilizing our Trusted Research Environment (TRE) and Trusted Data Lakehouse (TDL), biopharma companies and public health agencies can conduct large-scale, compliant research that was previously impossible. We are moving toward a “Learning Health System,” where every patient encounter can contribute to our collective understanding of disease and treatment, creating a continuous loop of evidence generation and clinical improvement.
Whether you are a researcher looking to launch a multi-national trial or a sponsor navigating the complexities of FDA Accelerated Approval, the strategies are clear: accept collaboration, leverage regulatory innovations, and invest in the technology that turns data into life-saving insights. The tools to accelerate clinical research are already here; the challenge now is to adopt them at scale to transform the future of medicine.
Accelerate your clinical research with Lifebit and explore our federated data solutions to see how we can help you cut your research timelines today. Together, we can ensure that the next medical breakthrough reaches the patients who need it most—faster than ever before.

