How to Choose a Compliant Pharmacovigilance Platform Without Losing Your Mind

Cut Case Processing Costs by 70% With a Compliant Pharmacovigilance Platform
A compliant pharmacovigilance platform is a regulated software system that automates the detection, collection, assessment, and reporting of adverse drug reactions (ADRs) — while meeting global regulatory standards like ICH E2B(R3), 21 CFR Part 11, and EU GVP.
The Evolution of Drug Safety Compliance
Historically, pharmacovigilance (PV) was a reactive, paper-based discipline. Following the Thalidomide tragedy in the 1960s and the subsequent Kefauver-Harris Amendment, the industry realized that passive monitoring was insufficient. Today, the landscape has shifted from simple record-keeping to complex, real-time data science. Modern pharmaceutical companies are no longer just managing spreadsheets; they are managing massive streams of multi-omic, clinical, and real-world data (RWD).
If you’re comparing options right now, here’s what separates the top-tier solutions:
- Regulatory Agility: Automated updates for E2B(R3) and local jurisdiction changes, ensuring that as the FDA or EMA updates their submission gateways, your system remains compatible without manual patches.
- AI-Driven Efficiency: Modular AI for case intake, automated medical coding, and duplicate detection that reduces the manual burden on drug safety associates (DSAs).
- Scalable Infrastructure: High-volume processing capabilities for enterprise-level data, capable of handling millions of ICSRs (Individual Case Safety Reports) annually without performance degradation.
- Data Sovereignty: The ability to analyze data in-situ without moving sensitive patient records, a critical requirement for complying with GDPR and other regional privacy laws.
The “Waterfall” Problem in Modern PV
Here’s the problem most teams run into: adverse event reports arrive from everywhere — doctors, social media, clinical trials, EHRs, and patient support programs — all at once, in different formats, across different regulatory jurisdictions. Without the right infrastructure, your team is, as one industry observer put it, “trying to catch a waterfall with a teacup.”
The volume of data is growing at an estimated 15-20% annually. For a mid-sized pharma company, this means thousands of new cases every month. The stakes are real. Enterprise cloud pharmacovigilance platforms cost between $150K and $400K annually. A wrong choice means compliance fines, operational bottlenecks, and in the worst case, delayed safety signals that put patients at risk.
The good news: the right platform can cut processing costs by up to 70% and accelerate case handling by 90%. But only if it fits your organization’s size, data environment, and global regulatory footprint.
I’m Dr. Maria Chatzou Dunford, CEO and Co-founder of Lifebit, and I’ve spent over 15 years at the intersection of AI, genomics, and health data infrastructure — working with pharmaceutical organizations and public institutions to build secure, compliant environments for drug safety and evidence generation. My work building federated data platforms has shown me exactly where traditional compliant pharmacovigilance platform approaches fall short — and where the next generation of real-time, AI-powered surveillance is heading.
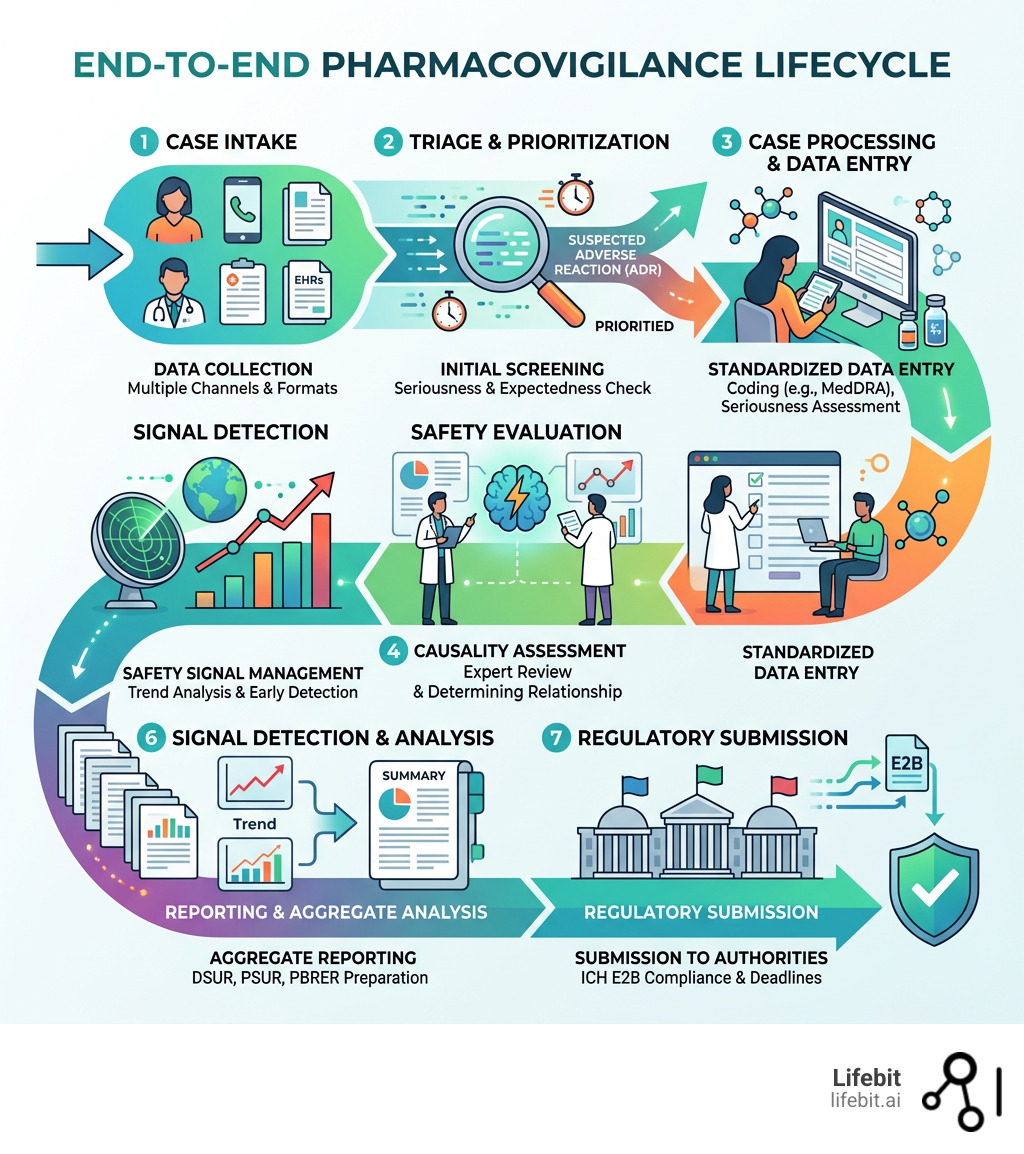
3 Regulatory Must-Haves for Your Compliant Pharmacovigilance Platform
When we talk about a compliant pharmacovigilance platform, we aren’t just talking about a fancy database. We are talking about a “safety command center” that must survive the scrutiny of global health authorities. If your system fails an audit, the financial and reputational damage can be catastrophic.
The Regulatory “Big Three”
To stay in the good graces of the FDA, EMA, and other bodies, your platform must adhere to:
- ICH E2B(R3): This is the international standard for transmitting Individual Case Safety Reports (ICSRs). Modern platforms use XML-based reporting to ensure that data flows seamlessly between sponsors and authorities. The R3 standard is significantly more complex than its predecessor (R2), requiring more granular data on dosage, timing, and patient history. A compliant platform must handle these complexities without losing data integrity.
- 21 CFR Part 11: If you are operating in the US, this is your bible for electronic records and signatures. It requires a permanent, unchangeable audit trail. If a user changes a single comma in a patient narrative, the system must record who did it, when, and why. This includes “versioning” of cases, where every iteration of a report is saved and retrievable for inspectors.
- EU GVP (Good Pharmacovigilance Practices): These modules (currently 16 in total) dictate how you manage signals, report adverse reactions, and maintain your Pharmacovigilance System Master File (PSMF). The PSMF is a living document that describes the PV system used by the marketing authorization holder. A compliant platform should automatically generate the data required to keep the PSMF current.
Data Validation and Medical Coding
A platform is only as good as the data inside it. High-performing systems use MedDRA (Medical Dictionary for Regulatory Activities) and WHO-DD (World Health Organization Drug Dictionary) for standardized coding.
MedDRA is organized into a five-level hierarchy:
- System Organ Class (SOC)
- High-Level Group Term (HLGT)
- High-Level Term (HLT)
- Preferred Term (PT)
- Lowest Level Term (LLT)
This ensures that a “pounding headache” reported in London and a “migraine” reported in New York are categorized correctly for signal detection. Without automated coding, medical reviewers spend hours manually mapping terms, which introduces human error and delays reporting timelines.
Audit Trails and Quality Control
We often tell our partners that transparency is the best defense against risk. A compliant pharmacovigilance platform should offer built-in cross-field validation. This prevents “garbage in, garbage out” by ensuring required fields (like the four minimum criteria for a valid ICSR: an identifiable reporter, an identifiable patient, a suspect product, and an adverse event) are completed before a case can be submitted.
Events like those discussed at Pharmacovigilance World 2025 | Drug Safety | Risk Management highlight that as regulations tighten, the ability to demonstrate a clear chain of custody for safety data is what keeps safety leads sleeping soundly at night. Quality control (QC) workflows should be embedded into the platform, allowing for second-person review and automated flagging of inconsistencies.
For a deeper dive into the technical requirements of these systems, check out our guide on pharmacovigilance compliance solutions.

Achieving Global Scalability with a Compliant Pharmacovigilance Platform
Whether you are a small biotech with one candidate in Phase I or a global giant managing a portfolio of 50 marketed drugs, your platform must scale. Scaling isn’t just about handling more data; it’s about handling more complexity, such as localized reporting rules in emerging markets.
Multi-tenant Architecture For organizations managing safety data for multiple clients or partners, a multi-tenant architecture is essential. This allows you to keep data strictly segregated while using a single, unified interface to monitor performance metrics and SLAs across the board. This is particularly useful for Contract Research Organizations (CROs) who manage PV for multiple sponsors.
Aggregate Reporting (PSUR/DSUR/PBRER) While individual cases (ICSRs) are the “daily grind” of PV, aggregate reports are the “big picture.” Your platform should automate the compilation of:
- DSUR (Development Safety Update Report): For drugs still in clinical trials, focusing on the safety profile of the investigational drug.
- PSUR/PBRER (Periodic Safety Update Report): For post-market surveillance, providing an evaluation of the risk-benefit balance of a medicinal product.
Manually pulling these reports can take weeks of data cleaning and formatting. A truly compliant pharmacovigilance platform reduces this to hours by pulling data directly from the safety database into pre-configured regulatory templates, ensuring that the data in the report perfectly matches the data in the database.
Global Regulatory Standards List:
- FDA (USA): FAERS reporting, MedWatch forms, and the requirement for Risk Evaluation and Mitigation Strategies (REMS).
- EMA (Europe): EudraVigilance integration, XEVMPD (Extended EudraVigilance Medicinal Product Dictionary), and strict adherence to GVP modules.
- PMDA (Japan): Specific J-E2B requirements and the need for Japanese-language narratives and local medical coding.
- NMPA (China): Rapidly evolving requirements for local adverse event reporting and periodic safety reports.
- Health Canada: Localized reporting rules and specific timelines for serious unexpected adverse drug reactions.
Why SaaS Is Failing Drug Safety—And Why Federated AI Is Replacing It
One of the biggest decisions you’ll face is where your data lives. Historically, companies had to choose between clunky on-premise systems or centralized cloud (SaaS) models. But there is a third way that is rapidly becoming the gold standard for high-security environments: Federated AI.
The Deployment Breakdown
| Feature | On-Premise | Standard SaaS/Cloud | Federated AI (Lifebit) |
|---|---|---|---|
| Speed of Setup | Slow (6-12 months) | Fast (3-6 months) | Rapid (Weeks) |
| Data Control | High | Low (Data is moved) | Absolute (Data stays in situ) |
| Compliance | Manual updates | Automatic updates | Built-in Governance |
| Scalability | Hard/Expensive | Easy | Infinite (Cross-border) |
The Problem with Centralized SaaS
In a traditional SaaS model, you must move your sensitive patient data to the vendor’s cloud. This often triggers “data residency” nightmares. For example, if you are a German pharmaceutical company, your data may be legally required to stay within German borders. If your SaaS provider hosts data in a US-based data center, you are immediately in violation of GDPR. Furthermore, moving massive datasets incurs “egress fees” and creates a security vulnerability during the transfer process.
Why Federated AI is Winning
With a federated approach, the AI comes to the data. At Lifebit, we use a Trusted Research Environment (TRE) and a Trusted Data Lakehouse (TDL). This means your safety data stays behind your firewall, in your region, while our AI tools perform the analysis.
This architecture provides several key benefits:
- Zero Data Movement: Sensitive patient identifiers never leave your secure environment, eliminating the risk of interception.
- Real-Time Analysis: Because the AI is local to the data, there is no latency caused by uploading large files. This is particularly vital for real-time pharmacovigilance, where waiting for data transfers can mean missing a critical safety signal.
- Unified Governance: You can grant and revoke access to specific datasets instantly, ensuring that only authorized personnel can view sensitive safety information.
By keeping data in-situ, you eliminate the risk of data breaches during transit and ensure you are always in compliance with local privacy laws. This proactive stance is the future of the industry, as explored in our piece on AI’s role in proactive drug safety.
Process Cases 90% Faster: The AI Strategy for Modern Drug Safety
If you are still using spreadsheets or manual entry for case intake, you are essentially trying to win a Formula 1 race on a bicycle. The volume of safety data is exploding. Between social media mentions, electronic health records (EHRs), and clinical trial data, the human-only model of case processing is officially broken.
The “Touchless” Case Workflow
A modern compliant pharmacovigilance platform uses AI to achieve what we call “near-touchless” processing. This doesn’t mean humans are removed from the loop; rather, it means humans are elevated to the role of “reviewers” rather than “data entry clerks.”
- Intelligent Intake (NLP): Natural Language Processing (NLP) extracts key entities (Patient, Drug, Event, Reporter) from unstructured text. This includes scanning medical journals, emails, and even phone call transcripts from patient hotlines.
- Auto-Triage: The system automatically prioritizes cases based on seriousness. A report of “death” or “hospitalization” is instantly moved to the top of the pile, while a “mild rash” is queued for standard processing. This ensures that the most critical safety issues are addressed within the strict 7-day or 15-day regulatory windows.
- Duplicate Detection: AI algorithms scan for similar reports across the entire database. It looks for matching patient initials, dates of birth, and event dates to ensure that the same event reported by both a doctor and a patient isn’t counted twice, which would skew your safety signals and lead to false alarms.
- Narrative Generation: Large Language Models (LLMs) can now draft initial case narratives. These models are trained on medical terminology to ensure the narrative is clinical, objective, and concise. Medical reviewers then simply verify and sign off on the draft, reducing narrative writing time from hours to minutes.
Signal Detection: Moving from Reactive to Proactive
Traditional PV is reactive — you wait for a report, then you analyze it. AI-driven platforms enable proactive surveillance. By integrating with systems like EudraVigilance, our platform can perform disproportionality analysis.
This involves calculating scores like:
- PRR (Proportional Reporting Ratio): Comparing the frequency of a specific adverse event for a drug against the frequency of that event for all other drugs in the database.
- ROR (Reporting Odds Ratio): A similar statistical measure used to identify if a drug-event combination is occurring more frequently than expected.
These tools allow safety teams to find hidden patterns in global datasets before they become “surprises.” For a comprehensive look at how these technologies work under the hood, read our complete guide to AI for pharmacovigilance.
3 Critical Questions About Your Compliant Pharmacovigilance Platform
What is the main purpose of a compliant pharmacovigilance platform?
Think of it as your drug’s “safety bodyguard.” Its primary job is to automate the detection and reporting of adverse reactions to ensure patient safety and regulatory adherence. It replaces manual, error-prone processes with a validated, audit-ready workflow that acts as a single source of truth for every safety event associated with your product. Beyond compliance, it serves as a strategic tool to protect the product’s market authorization and the company’s reputation.
How does AI reduce pharmacovigilance costs by 70%?
The majority of PV costs come from “human-in-the-loop” time — specifically manual data entry, coding, and narrative writing. By automating these repetitive tasks (touchless processing), organizations can handle 10x the case volume without increasing headcount. It also eliminates the “re-work” costs associated with human errors that lead to regulatory queries. When a system automatically codes a term correctly the first time, it removes the need for expensive medical directors to spend time correcting basic data entry mistakes.
Can these platforms handle unstructured data from social media?
Yes! Modern platforms use advanced NLP and OCR (Optical Character Recognition) to scan unstructured sources like social media posts, literature, and scanned PDFs. This is critical because a significant percentage of real-world evidence (RWE) is buried in these “messy” formats. A compliant pharmacovigilance platform can extract these mentions, validate them against safety dictionaries, and flag them for medical review. This is increasingly important as regulators like the EMA now require companies to monitor social media for potential safety signals.
How does the platform handle legacy data migration?
One of the biggest fears in switching platforms is losing historical data. A robust platform should include automated ETL (Extract, Transform, Load) tools that can map data from legacy systems (like old versions of Argus or ArisGlobal) into the new E2B(R3) format. This ensures that your historical safety profile remains intact and searchable for signal detection purposes.
What is the role of the QPPV in a digital-first environment?
The Qualified Person Responsible for Pharmacovigilance (QPPV) remains legally responsible for the safety system. In a digital-first environment, the platform empowers the QPPV by providing real-time dashboards and automated alerts. Instead of waiting for monthly reports, the QPPV can have a live view of the system’s performance, compliance rates, and emerging signals, allowing for much faster decision-making.
Future-Proof Your Safety Strategy With Federated AI
Choosing a compliant pharmacovigilance platform is one of the most significant infrastructure decisions your organization will make. The goal isn’t just to “check the box” for compliance today, but to build a system that can handle the multi-omic and real-world data challenges of 2030 and beyond. As personalized medicine and cell therapies become more common, the complexity of safety monitoring will only increase.
At Lifebit, we believe the answer lies in Federated AI. By allowing data to stay secure and localized while enabling real-time, global insights, we help biopharma and regulators move from “catching up” to “staying ahead.” This approach transforms safety data from a regulatory burden into a strategic asset. By analyzing safety signals in the context of genomic and clinical data, companies can better understand which patient populations are at risk and why, leading to safer drugs and better patient outcomes.
Whether you are looking for a Trusted Research Environment to analyze complex safety signals or a Real-time Evidence & Analytics Layer (R.E.A.L.) to monitor post-market data, the right technology should empower your team, not overwhelm them. Don’t let your safety strategy be a bottleneck to innovation. Transform your PV operations with Lifebit and start turning safety data into a strategic asset.

