How to Not Fail Your Audit with Post-Market Surveillance Software

When an Audit Hits, Your Drug Safety Monitoring Software Either Saves You or Sinks You
Drug safety monitoring software is the technology that pharma companies, regulators, and public health agencies use to collect, process, and act on adverse drug reaction (ADR) data — before a regulator finds a gap you missed.
Here is what you need to know at a glance:
| What it does | Why it matters |
|---|---|
| Automates ICSR intake and processing | Cuts case handling time by 50-60% |
| Detects safety signals from multiple data sources | Catches ADRs missed by manual review |
| Ensures compliant submissions (E2B(R3), MedDRA) | Keeps you audit-ready at all times |
| Integrates RWD, EHRs, and social media | Expands surveillance beyond spontaneous reports |
| Provides audit trails and traceability | Protects you when regulators come knocking |
The stakes are not abstract. ADRs cause at least 7,000 deaths annually in the US alone, and cost the healthcare system tens of billions of dollars each year. Globally, WHO’s VigiBase now holds over 40 million Individual Case Safety Reports (ICSRs) from more than 180 countries — a volume no manual process can keep pace with.
Yet many organizations are still running pharmacovigilance on legacy systems, spreadsheets, and fragmented workflows. When an audit arrives, those gaps become findings. Findings become warning letters. Warning letters become crises.
This guide walks you through what modern drug safety monitoring software actually does, how it automates the workflows that auditors scrutinize, and what to look for when your current setup is no longer enough.
I’m Dr. Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where I’ve spent over 15 years working at the intersection of AI, federated data platforms, and real-world evidence — including building systems that power real-time drug safety monitoring software for regulatory agencies and life sciences organizations worldwide. In the sections ahead, I’ll break down exactly what separates a compliant, audit-ready PV system from one that fails under scrutiny.
Drug safety monitoring software word roundup:
Why Manual Pharmacovigilance is an Audit Trap in 2025

If you are still relying on manual entry and siloed spreadsheets to manage your safety data, you are essentially walking into an audit with a blindfold on. In 2025, the sheer volume of data makes manual oversight impossible. We are seeing a massive “data explosion” where safety information isn’t just coming from doctors’ reports—it’s flowing in from Electronic Health Records (EHRs), insurance claims, and even social media.
The Regulatory Landscape: Beyond Basic Compliance
Regulators have noticed this shift and have responded by tightening the screws. Agencies like the FDA and EMA have moved beyond simple reporting requirements to mandating strict adherence to the ICH E2B(R3) standard for electronic submissions. This standard is significantly more complex than its predecessor, requiring more granular data and specific XML structures. If your drug safety monitoring software cannot handle these complex XML data exchanges or fails to map terms correctly to the latest MedDRA (Medical Dictionary for Regulatory Activities) coding, you face immediate compliance risks.
Furthermore, the introduction of ISO IDMP (Identification of Medicinal Products) standards means that every detail about a drug—from its ingredients to its packaging—must be uniquely and globally identifiable. This is not just a clerical task; it requires a system that can maintain a “single source of truth” across different global markets. Traditional systems often suffer from “signal detection lag,” where a dangerous pattern in patient reactions isn’t spotted until months after it begins. By implementing real-time pharmacovigilance, organizations can break down these data silos and identify risks as they happen, rather than during a post-mortem audit.
The Seven Pillars of Good Pharmacovigilance Practices (GVP)
To survive an audit, your software must align with the EMA’s GVP modules, which serve as the global gold standard. These include:
- Pharmacovigilance System Master File (PSMF): Your software must automatically generate and update this document, which describes the PV system.
- Quality Systems: Ensuring that every process is documented, from data collection to submission.
- Pharmacovigilance Inspections: The system must provide a clear, immutable audit trail that shows who accessed what data and when.
- Signal Management: Moving from reactive to proactive detection.
- Risk Management Systems: Tracking the effectiveness of risk-minimization measures.
- Management and Reporting of ADRs: Ensuring timely submission to EudraVigilance or the FDA FAERS.
- Periodic Safety Update Reports (PSURs): Automating the aggregation of data for these massive, recurring reports.
Critical Data Standards for Interoperability
Interoperability is the “secret sauce” of a successful audit. If your systems can’t talk to each other, data gets lost, and auditors find gaps. Key standards include:
- ICH E2B(R3): The international standard for transmitting Individual Case Safety Reports (ICSRs).
- HL7 eMDR: Used specifically for electronic Medical Device Reporting.
- WHO Drug Dictionary: Ensuring every medication is coded consistently across the globe.
- CDISC SDTM: Standardizing clinical trial data for regulatory review.
Using a pharmacovigilance compliance solution that natively supports these standards ensures that your data is “clean” from the moment it is captured. This standardized terminology allows for seamless XML data exchange between pharmaceutical companies and health authorities, reducing the risk of “lost in translation” errors that trigger regulatory red flags.
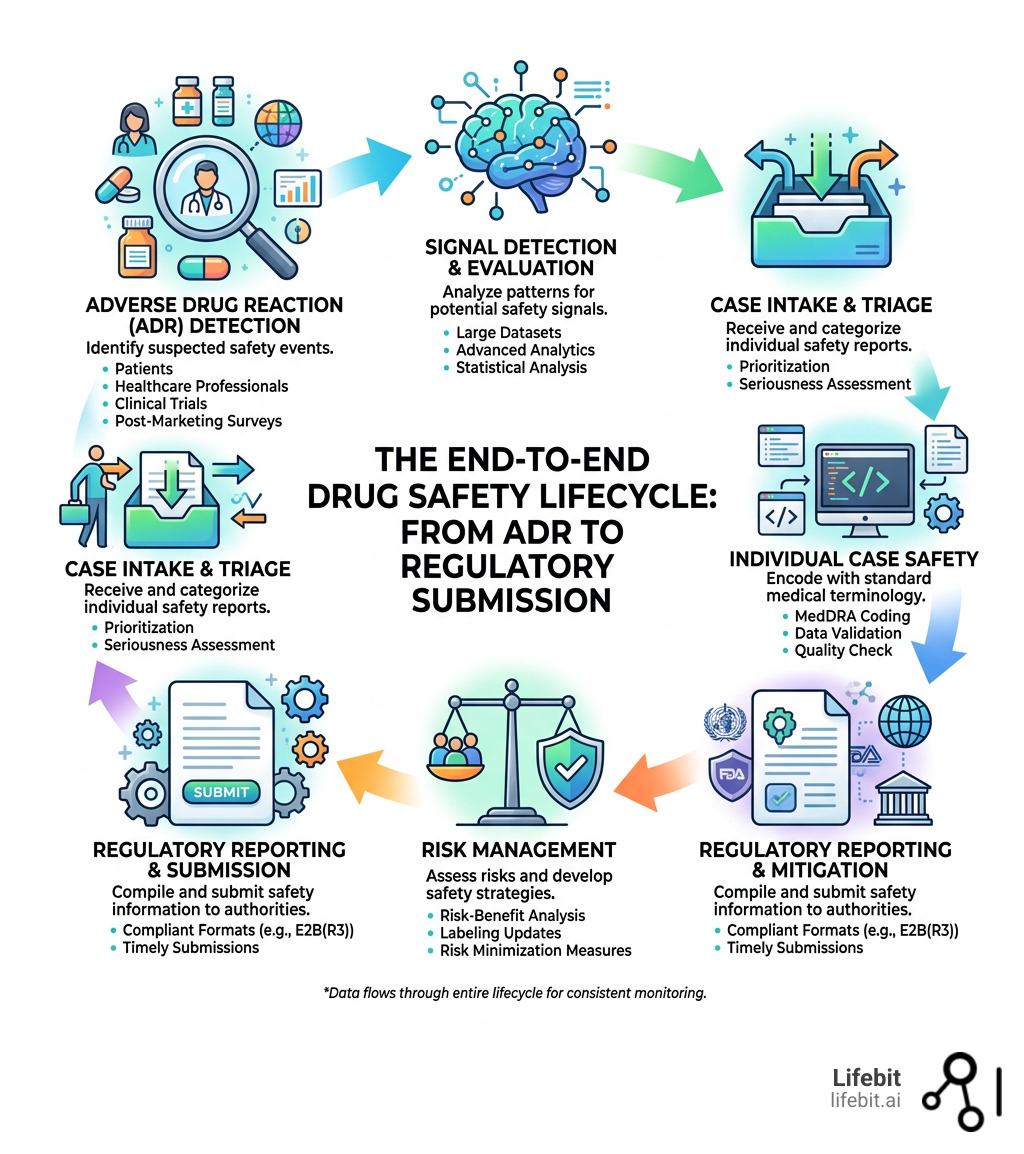
Automating the ICSR Lifecycle to Slash Processing Time
The Individual Case Safety Report (ICSR) is the heart of pharmacovigilance, but processing them manually is a recipe for burnout and error. A single case can involve dozens of pages of medical records, handwritten notes, and lab results.
| Feature | Manual Processing | AI-Driven Automation |
|---|---|---|
| Case Intake | Manual typing from PDFs/Emails | Automated OCR/NLP extraction |
| Data Entry Errors | High (Human fatigue) | Reduced by ~50% |
| Processing Time | Days/Weeks | Reduced by ~40-60% |
| Regulatory Compliance | Risk of missed deadlines | Automated alerts and tracking |
| Scalability | Requires hiring more staff | Scales instantly with data volume |
The Anatomy of an Automated ICSR Workflow
Modern drug safety monitoring software breaks down the ICSR lifecycle into several automated stages:
- Intake & Triage: Using Robotic Process Automation (RPA) and Optical Character Recognition (OCR), the software “reads” handwritten forms and unstructured emails. It identifies the four pillars of a valid case: an identifiable patient, an identifiable reporter, a suspect product, and an adverse event.
- Data Extraction & Coding: Natural Language Processing (NLP) extracts clinical entities and maps them to MedDRA terms. For example, if a reporter writes “bad headache,” the AI automatically codes it to the MedDRA Preferred Term (PT) “Headache.”
- Duplicate Detection: The system scans the database to ensure the same event hasn’t been reported twice through different channels, a common issue that skews safety data.
- Medical Review: The software flags serious or unexpected cases for immediate human review, while auto-populating the narrative section of the report.
- Submission: Once validated, the system generates the E2B(R3) XML file and transmits it directly to regulatory gateways.
This isn’t just about speed; it’s about quality. Automation ensures that every case is validated against regulatory rules before it ever reaches a human reviewer. For more on the science behind these shifts, you can explore the WHO’s research on pharmacovigilance.
Enhancing Signal Detection with AI-Powered Drug Safety Monitoring Software
Signal detection is the process of finding “needles in the haystack”—identifying a new side effect within millions of data points. Traditional methods were reactive, relying on disproportionality analysis (like ROR or PRR) which only works after a significant number of reports have accumulated. AI for pharmacovigilance has turned this into a proactive science.
Using Bayesian networks and machine learning, modern software can achieve a 30-40% boost in detection accuracy compared to manual methods. These algorithms look for patterns that humans might miss, such as a specific combination of a drug and a pre-existing condition causing a rare reaction. By prioritizing signals based on severity and frequency, AI ensures that the most critical risks are addressed first, providing real-time alerts that can save lives.
Streamlining Literature Review with Intelligent Triage
Pharmaceutical companies are legally required to monitor thousands of scientific journals for mentions of their drugs. Doing this manually is an impossible task, often leading to “backlogs” that are a primary target for auditors. Modern AI-driven pharmacovigilance solutions use NLP to “triage” these articles.
These tools can identify reportable adverse events 5x faster than a human researcher and reduce the overall literature review workload by up to 70%. By ranking articles based on “relevancy,” the software ensures that your team only spends time on the papers that actually matter for safety reporting, automatically formatting references in the required Vancouver style for aggregate reports.
Integrating Real-World Data Beyond Spontaneous Reports
Spontaneous reports (where a doctor or patient voluntarily reports an ADR) only capture about 5% of actual adverse events. This is known as the “under-reporting phenomenon.” To get the full picture, we must look at Real-World Data (RWD).
Modern drug safety monitoring software now integrates:
- Electronic Health Records (EHRs): Providing a deep dive into patient history, concomitant medications, and underlying comorbidities that might influence drug reactions.
- Insurance Claims: Tracking long-term outcomes across millions of patients, allowing for longitudinal studies that clinical trials cannot replicate.
- Social Media Monitoring: NLP studies have shown an 88% success rate in identifying ADRs from online forums and patient posts. Patients often discuss side effects on Reddit or Facebook long before they tell their doctor.
The Role of Real-World Evidence (RWE) in Safety
This shift toward post-marketing drug surveillance allows us to see how a drug performs in the “wild,” outside the controlled environment of a clinical trial. For instance, the FDA’s Sentinel Initiative uses RWD to monitor the safety of approved drugs in real-time across a massive network of healthcare partners.
Mobile safety apps and wearables are also becoming vital, providing real-time, patient-generated health data. A wearable device can flag a cardiac arrhythmia or a drop in blood oxygen levels that a patient might not even feel, providing a “digital biomarker” for safety that was previously unavailable. This allows for a “proactive” safety model where interventions can happen before a serious event occurs.
Overcoming Implementation Hurdles in Drug Safety Monitoring Software
Transitioning to AI-powered systems isn’t without challenges. Data privacy is paramount, especially under GDPR and HIPAA regulations. Organizations often struggle with “Black Box” AI—if a computer flags a safety risk, an auditor will want to know why. If the software cannot explain its reasoning, the finding is useless for regulatory purposes.
This is where explainable AI comes in. Modern systems must provide “human-in-the-loop” workflows where AI suggests, but humans verify. To be audit-ready, your software must come with a GxP validation package, proving that the algorithms have been tested and are reproducible. Constant monitoring for “data drift” ensures that your AI models remain accurate as new types of data (like new medical terminology or changing reporting patterns) are introduced.
Future-Proofing Your Safety Strategy: Predictive Analytics and Beyond
The future of drug safety is moving from detection to prediction. We are entering an era where we can use predictive modeling to identify which patients are most at risk of an ADR before they even take the first pill. This is the cornerstone of Precision Medicine.
Key Trends Shaping the Next Decade
- Blockchain for Traceability: Creating an unalterable “chain of custody” for safety reports. This ensures that from the moment a patient reports a side effect to the moment it reaches a regulator, the data cannot be tampered with, ensuring 100% traceability for auditors.
- Digital Therapeutics (DTx): Software-as-a-drug requires its own unique safety monitoring protocols. As DTx becomes more common, drug safety monitoring software must evolve to track “digital side effects” like cognitive fatigue or behavioral changes.
- Federated AI & Learning: This is a core part of our mission at Lifebit. Instead of moving sensitive patient data to the AI (which creates massive security risks), we move the AI to the data. This allows for global collaboration across borders without ever compromising patient privacy or violating data residency laws.
Federated Learning: The Privacy-First Frontier
By using Beyond Vigilance: AI’s Role in Proactive Drug Safety strategies, organizations can utilize Trusted Research Environments (TREs) to analyze global datasets securely. In a federated model, the data stays behind the hospital or agency’s firewall. The AI model travels to the data, learns from it, and then returns with only the “insights” (e.g., “Drug X shows a 5% higher risk of reaction in patients with Gene Y”). This enables a level of safety surveillance that was previously impossible due to the legal and ethical complexities of moving patient data across international borders.
Frequently Asked Questions about PV Compliance
What are the core data standards for drug safety monitoring software?
The most critical standards are ICH E2B(R3) for case reporting, MedDRA for standardized medical coding, and ISO IDMP for medicinal product identification. Supporting these ensures your data is interoperable with global regulators like the FDA, EMA, and PMDA. Failure to support these standards is one of the most common reasons for audit findings.
How does AI reduce data entry errors in pharmacovigilance?
AI uses Natural Language Processing (NLP) and Optical Character Recognition (OCR) to extract data directly from source documents. This eliminates the “human factor” of fatigue and typos, which are common in manual data entry. By automating the mapping of verbatim terms to MedDRA codes, the software ensures consistency across thousands of reports, typically halving the error rate and significantly improving data quality for signal detection.
Can modern software handle ADRs from social media and wearables?
Yes. Advanced drug safety monitoring software uses NLP to scan unstructured text on social media and integrates API feeds from wearables. This provides a “real-time” view of patient safety that spontaneous reporting systems often miss. However, the challenge lies in the “signal-to-noise” ratio; modern systems use sophisticated filtering to separate genuine adverse events from general complaints or unrelated health discussions.
What is GxP validation in the context of safety software?
GxP (Good Practice) validation is a formal process of documenting that a software system meets its intended requirements and is reliable and consistent. For drug safety, this usually involves Computer System Validation (CSV). Auditors require proof that the software has been tested under various scenarios and that its automated decisions (like AI-driven coding) are reproducible and explainable.
Conclusion
The era of “good enough” pharmacovigilance is over. As data volumes grow and regulators become more tech-savvy, your drug safety monitoring software must be more than just a database—it must be an intelligent, proactive partner.
At Lifebit, we built the R.E.A.L. (Real-time Evidence & Analytics Layer) platform to solve these exact challenges. By utilizing a federated AI architecture, we allow you to detect ADRs in real-time across global datasets with zero data movement. This not only ensures 100% compliance and traceability but can also lead to up to 30% savings in surveillance costs.
Don’t wait for an audit to find the holes in your safety strategy. Transitioning to a modern pharmacovigilance platform ensures that you are not just compliant today, but future-proofed for the data challenges of tomorrow.

