An Essential Guide to Anonymized Patient Data Providers

Stop Waiting Months and Burning Budget: Where to Get Compliant Anonymized Patient Data Fast
I’m looking for services that provide access to anonymized patient data for research purposes. If that’s you, you’re facing a critical challenge: the healthcare data you need exists, but it’s locked behind privacy regulations, data silos, and technical barriers. This guide cuts through the complexity.
While a handful of commercial platforms, open-science initiatives, and government bodies provide access, choosing the right one is crucial. Here’s what you need to consider:
Key considerations when choosing a provider:
- Data type needed: EHR, claims, genomics, imaging, or multi-modal datasets
- Access model: Federated (data stays at source) vs. centralized (aggregated datasets)
- Geographic coverage: US-only, European, or global patient populations
- Compliance: HIPAA, GDPR, and regulatory-grade data handling
- Cost structure: Subscription, per-project, or subsidized academic access
The reality is stark. A significant portion of daily medical decisions are not backed by high-quality evidence, creating a dangerous “Evidence Gap.” Meanwhile, healthcare data is plentiful but siloed, making it nearly impossible to generate the real-world evidence needed for drug development, pharmacovigilance, and precision medicine. Traditional data procurement takes months, costs millions, and often results in datasets that are unsuitable for the research question.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where I’ve spent over 15 years building federated genomics and biomedical data platforms. We enable researchers like you to access services that provide access to anonymized patient data for research purposes without moving sensitive data from its source. This guide will walk you through the landscape of data providers, help you understand which access model fits your needs, and show you how to ensure data quality and compliance.
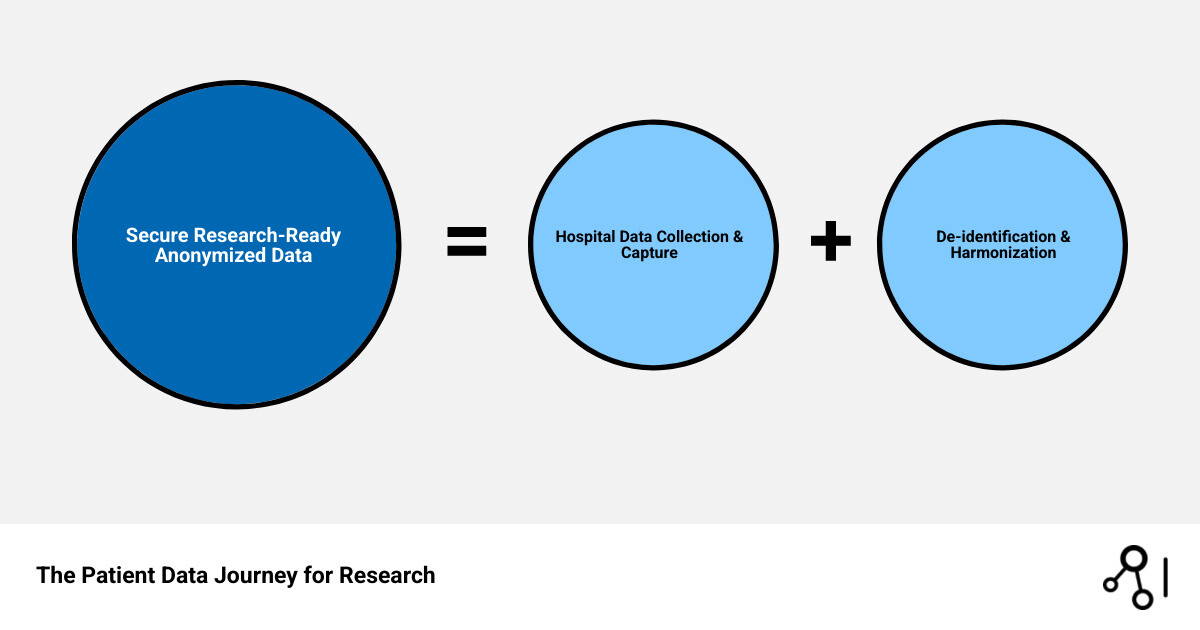
Easy I’m looking for services that provide access to anonymized patient data for research purposes. glossary:
Your RWD Is Costly and Slow—Fix These 5 Data Gaps and Access Clean, Compliant Patient Records
Modern medical research faces a paradox: we’re drowning in data but starving for insights. If you’re looking for services that provide access to anonymized patient data for research purposes, you’re facing a challenge that’s costing lives.
Somewhere in the world, a patient’s medical journey holds the key to your research question. The data exists—billions of patient records captured daily. But it’s locked away, scattered across incompatible systems, and wrapped in layers of regulatory protection.
This is the landscape of real-world data (RWD)—the messy, complex, incredibly valuable information generated during routine clinical care. Unlike the pristine conditions of clinical trials, RWD reflects actual patient experiences across diverse populations and treatment settings.
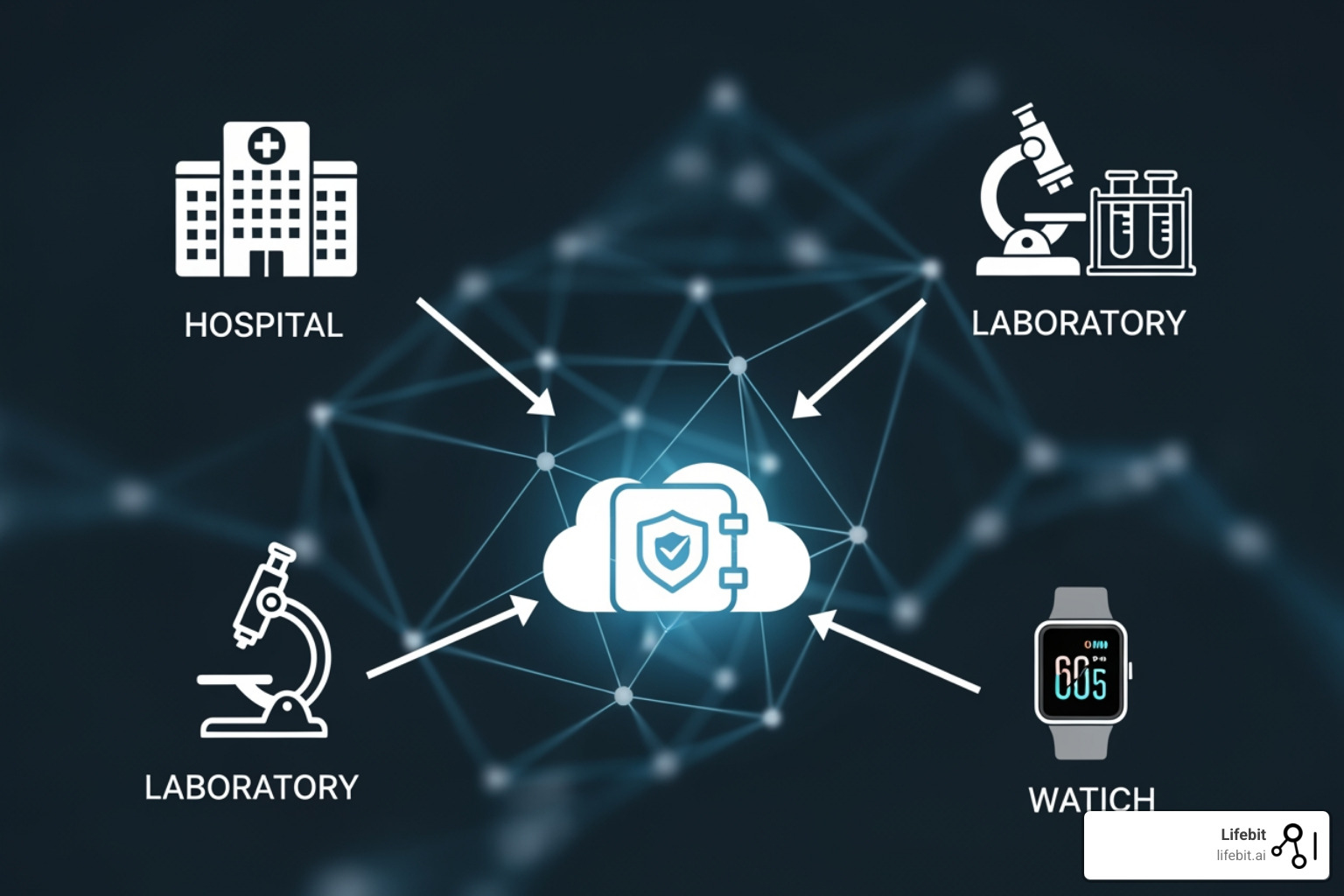
Electronic health records (EHRs) capture the clinical story—diagnoses, medications, lab results. Claims data tracks the financial footprint of healthcare. Genomic data opens up the molecular basis of disease, while medical imaging provides visual evidence. Together, they paint a comprehensive picture.
But data fragmentation means a patient’s information is scattered across dozens of systems. Data quality issues like missing values and errors are rampant. Regulatory barriers like HIPAA and GDPR create complex compliance problems. And the prohibitive expense of traditional data access can consume budgets before research even begins.
Yet, the promise of RWD has never been greater. It enables faster drug findy, makes precision medicine a reality, and improves drug safety surveillance. At Lifebit, our federated platform was built to address these challenges, enabling researchers to analyze sensitive patient data where it lives, without the risks and costs of traditional approaches.
What Types of Anonymized Patient Data Can You Access?
When you’re looking for services that provide access to anonymized patient data for research purposes, understanding the available data types is your first step.
- Electronic Health Records (EHR): The foundation of clinical data, containing diagnoses (coded with ICD-10), procedures (CPT), lab results (LOINC), and medications (RxNorm). While rich in clinical detail, EHR data is notoriously variable. The scale of some platforms, however, offers the statistical power needed for robust analysis, even with this inherent messiness.
- Insurance Claims Data: Complements EHRs by capturing the complete patient journey across different healthcare providers over long periods. It includes diagnoses, procedures, and prescription data, making it invaluable for health economics and outcomes research (HEOR). Its primary limitation is the lack of granular clinical data, such as lab values or vital signs, which are not required for billing.
- Genomics and Multi-omic Data: The cutting edge of precision medicine, including whole-genome sequencing (WGS), whole-exome sequencing (WES), transcriptomic, and proteomic data. The main challenge and opportunity lie in linking this molecular data to longitudinal clinical outcomes from EHRs and claims to discover novel biomarkers and therapeutic targets.
- Medical Imaging Data: X-rays, CT scans, and MRIs, typically stored in DICOM format, provide critical visual evidence. The field of “radiomics” uses AI to extract thousands of quantitative features from these images, turning them into high-dimensional data that can be analyzed alongside clinical and genomic information for more accurate diagnostic and prognostic models.
- Unstructured Clinical Notes: Physician’s notes, discharge summaries, and pathology reports contain rich, nuanced information missed by structured fields. Natural language processing (NLP) is essential to unlock this data, extracting concepts like specific symptoms, disease severity, and even social determinants of health to create a much richer patient phenotype.
Key Problems in Accessing Anonymized Patient Data for Research
Accessing data remains frustratingly difficult, delaying or halting critical research.
- Data Privacy Regulations: HIPAA and GDPR are essential protections, but compliance is complex, requiring expertise in de-identification, data use agreements, and secure environments. The patient and public involvement in research movement also rightly demands that ethical use goes beyond legal compliance, ensuring patient consent and benefit are central to any project.
- Data Silos: Data is often locked in separate databases across different departments and institutions. A single patient’s journey with a chronic condition might be split across a primary care physician’s EHR, a specialist’s system at a different hospital, and a third-party lab’s database. Each silo has its own access policies and technical infrastructure, making a comprehensive view of the patient nearly impossible to assemble.
- Lack of Standardization: Data from different sources uses different codes, formats, and terminologies. One hospital might use a local coding system for a lab test, while another uses the international LOINC standard. Without a common data model (CDM) to map these disparate sources into a unified structure, large-scale analysis is impossible. Extensive data harmonization is required before any meaningful research can begin.
- Prohibitive Costs: Commercial data access licenses can cost hundreds of thousands to millions of dollars. Beyond that, the costs of cloud infrastructure, data storage, and the specialized data science expertise needed to process and analyze the data can be prohibitively expensive for both academic and commercial researchers.
- Ethical Considerations: Beyond regulations, researchers must consider fairness, transparency, and social benefit. Is the cohort representative of the broader population, or does it over-represent certain demographics, leading to biased findings? Is the research for the public good, or does it primarily serve a narrow commercial interest? These questions are paramount for responsible research.
Modern federated architectures are designed to solve these challenges, enabling compliant, cost-effective access without compromising privacy.
Federated vs. Centralized: Choose the Fastest, Safest Way to Anonymized Patient Data—Before You Waste Another Quarter
When you’re looking for services that provide access to anonymized patient data for research purposes, you’ll find two primary approaches: federated networks and centralized platforms. Understanding the difference is key to choosing the right path for your research.
Federated networks are like sending a question to a library network where each library searches its own shelves and sends back only the answer. Centralized platforms are like having all the books collected in one enormous reading room. Both have their place.
| Feature | Federated Data Networks | Centralized Data Platforms |
|---|---|---|
| Data Location | Data remains at source (e.g., hospital, biobank) | Data is aggregated into a single repository |
| Privacy | Maximized; sensitive data never leaves the local environment | De-identified data is shared, but aggregation carries some risk |
| Scale | Can connect billions of patient records globally | Typically large-scale, from contributing partners |
| Compliance | Easier to comply with local regulations (GDPR, HIPAA) | Requires robust de-identification and data governance |
| Access Model | Queries sent to data; results returned | Direct access to pre-processed, analysis-ready datasets |
| Data Types | Highly diverse (EHR, claims, genomics, imaging, etc.) | Highly diverse, but standardization occurs post-aggregation |

Federated Data Networks
Federated networks represent a paradigm shift. Instead of collecting data, data remains at the source—safely within the institution that collected it. This prioritizes privacy and data sovereignty.
Your queries are sent to the data, not the other way around. This “code-to-data” paradigm is the core of federation. Each institution processes your query locally within its secure firewall and sends back only aggregated, anonymized results. The raw patient-level data never travels. This approach maximizes privacy, simplifies compliance with cross-border data transfer laws like GDPR, and respects the data governance policies of each participating institution. Some advanced federated systems also employ Privacy-Enhancing Technologies (PETs) like differential privacy to add statistical noise to results, making it mathematically impossible to re-identify individuals even from multiple queries.
The scale achievable through federation is staggering, enabling large-scale, global collaboration without the bottlenecks of centralized aggregation. Networks like OHDSI (Observational Health Data Sciences and Informatics) and EHDEN (European Health Data & Evidence Network) connect hundreds of healthcare organizations globally. Some commercial networks provide access to billions of patient records, making it possible to study rare diseases and diverse populations. Open-science approaches that use common data models make data interoperable across wildly diverse sources, from national health registries to academic medical centers.
For researchers who need massive, globally diverse datasets while maintaining the highest standards of privacy, federated networks offer an neat solution.
Centralized Data Platforms
Centralized platforms collect data from multiple sources, then clean, normalize, and standardize it into a unified format. The result is aggregated, analysis-ready data.
This model offers speed and convenience. You can skip the months typically spent on data preparation and move directly from question to analysis. Commercial providers like Flatiron Health (specializing in oncology data) or Truveta (a consortium of US health systems) provide access to deeply curated, research-grade datasets. These platforms often provide access to large-scale patient records from their partners, already de-identified and harmonized.
The trade-off is that data has been moved and aggregated. While robust de-identification is applied, this model requires absolute trust in the platform’s security, governance, and compliance. Researchers must rely on the provider’s processes for data protection and quality control. Reputable platforms invest heavily in this trust, undergoing rigorous third-party security audits (e.g., SOC 2, ISO 27001) and maintaining transparent governance policies.
For researchers who prioritize rapid insights and are comfortable with the centralized model, this approach can dramatically accelerate research timelines.
Public Health and Academic Data Initiatives
Beyond commercial platforms, public health agencies and academic partnerships offer invaluable data access. These initiatives focus on national-level data and public health research.
In the United States, the Centers for Medicare & Medicaid Services (CMS) provides research-grade data through a structured application process, and the NIH’s All of Us Research Program is building one of the most diverse health databases in history. In the United Kingdom, the National Institute for Health and Care Research (NIHR) supports collaboration with hospital partners, and the UK Biobank provides unprecedented data on half a million volunteers. UK patients can express their data-sharing preferences via the NHS Your Data Matters website, reflecting a growing emphasis on patient autonomy. Across Europe, initiatives like the 1+ Million Genomes (1+MG) initiative and national biobanks like FinnGen in Finland are creating powerful resources for genomic and clinical research.
Many academic medical centers have built their own data warehouses and often connect to broader academic partnerships. These public and academic initiatives are critical for democratizing data access, particularly for studies focused on underserved populations.
Avoid IRB Delays and GDPR/HIPAA Risk: The Checklist to Prove Data Quality and Compliance
Access to anonymized patient data is a huge step, but it doesn’t guarantee success. You need the right data, handled the right way. Your responsibility is to ensure the data meets rigorous quality and compliance standards. This is about protecting patients, upholding ethics, and producing credible research.
When I’m looking for services that provide access to anonymized patient data for research purposes, I know that compliance and quality are the foundation of any breakthrough.

How Leading Services Ensure Compliance with HIPAA and GDPR: Your Data, Protected.
Data privacy is a legal and ethical mandate, dominated by HIPAA in the US and GDPR in Europe. Understanding how providers steer these regulations is critical.
- De-identification Standards: Under HIPAA, there are two paths. The Safe Harbor method removes 18 specific identifiers (name, address, etc.). The Expert Determination method involves a qualified statistician assessing the data and concluding that the risk of re-identifying an individual is very small. GDPR sets a higher bar, requiring that an individual is no longer identifiable by any means (anonymization). Because this is a very high threshold, many European projects use pseudonymization, where direct identifiers are replaced with artificial codes and the key linking them is stored separately and securely.
- Data Use Agreements (DUAs): These legal contracts govern how you can use the data. They specify permissible uses, security requirements, and prohibitions on re-identification. Before signing, scrutinize clauses related to publication rights, intellectual property for any discoveries made using the data, and requirements for data destruction upon project completion. Violating a DUA has serious legal and reputational consequences.
- Federated Architecture: This is a game-changer for compliance. When queries travel to the data rather than the data traveling to you, you avoid triggering complex cross-border data transfer restrictions under regulations like GDPR. This is the core principle of modern, privacy-first platforms that enables global research without moving sensitive information.
- Trusted Research Environments (TREs): The gold standard for secure analysis. A TRE is a controlled digital workspace where authorized researchers can analyze sensitive data without being able to export it. Beyond security, TREs provide immense value by offering high-performance computing resources and pre-installed analytical software (like R, Python, and specialized bioinformatics tools), saving researchers significant time and effort. Public bodies like the NHS in the UK are increasingly mandating the use of TREs for research on their data. At Lifebit, our platform includes a robust TRE, ensuring data remains secure, auditable, and fully governed.
Reputable providers will also have technical security like end-to-end encryption and independent certifications like ISO 27001 or SOC 2. Ask for their compliance documentation.
How to Verify the Quality of Anonymized Patient Data for Your Research: Don’t Guess, Know.
Accessible data isn’t always good data. Verifying quality before you commit resources is critical to avoid a project-killing findy later.
- Common Data Models: The use of a Common Data Model (CDM), such as OMOP or PCORnet, is a strong signal of quality. It transforms disparate data into a standardized format with consistent vocabularies. For example, a patient’s diagnosis of “Type 2 Diabetes” might be recorded as ICD-10 code E11.9 in one hospital and an internal proprietary code in another. A CDM maps both to a single, standard concept ID, making it possible to run the same analysis across both datasets and get comparable results. This demonstrates a commitment to reproducibility and quality.
- Data Dictionaries and Metadata: Demand comprehensive documentation that defines every variable—what it measures, its units, its origin, and the time period it covers. Without a clear data dictionary, you’re flying blind and risk misinterpreting the data.
- Data Curation Process: Ask how raw data is ingested, cleaned, and quality-checked. Reputable providers are transparent about their data curation and normalization processes, including how they handle missing data, resolve inconsistencies, and validate information.
- Feasibility Counts: Many services allow preliminary queries to see how many patients meet your inclusion/exclusion criteria (e.g., “females, aged 40-60, with a diagnosis of rheumatoid arthritis, prescribed methotrexate”). This is invaluable for assessing if your study is viable before you invest in a full data request.
- Data Provenance: Know where the data comes from—which hospitals, regions, and patient populations are represented. This helps you assess relevance and potential biases. Data from a single, specialized urban cancer center may not be generalizable to the broader population, so understanding the source is crucial for interpreting your results correctly.
Finally, leverage external resources. Organizations like the National Institute for Health and Care Research (NIHR) in the UK offer guidance and support for primary care research. Your research is only as good as its data. Do the due diligence.
Don’t Get Left Behind: 5 Trends That Will Change Patient Data Access
The landscape of patient data access is constantly evolving. For those looking for services that provide access to anonymized patient data for research purposes, staying ahead of these trends is crucial for long-term success.
-
AI and Machine Learning: AI is revolutionizing how we extract insights from complex datasets. AI-powered analytics can identify subtle patterns, predict outcomes, and automate data harmonization. Lifebit’s platform is designed with built-in capabilities for advanced AI/ML analytics to extract maximum value from global biomedical data.
-
Federated Learning: This extension of the federated model allows AI models to be trained on decentralized data without the data ever leaving its source. The model travels to the data, preserving privacy while enabling powerful AI development.
-
Synthetic Data Generation: Artificially generated data that statistically mimics real patient data but contains no actual patient information. This emerging technology allows for broader testing of algorithms without privacy concerns. Initiatives like Digital NHS in the UK are already exploring its potential.
-
European Health Data Space (EHDS): A landmark EU initiative to create a single market for health data, facilitating cross-border research while reinforcing patient control. Leading platforms with a strong presence in Europe are actively working to support EHDS readiness.
-
Multi-omics Data Integration: The future of precision medicine lies in integrating genomics, proteomics, and other “omics” data with clinical records. Lifebit’s federated AI platform is specifically designed to handle and integrate this multi-modal data, providing a holistic view for advanced research.
These trends point towards more secure, intelligent, and integrated approaches to patient data access, all with an unwavering commitment to privacy.
Costs, Compliance, Risk—Answered Fast: Your Anonymized Patient Data FAQ
What are the typical costs and access models?
When you’re looking for services that provide access to anonymized patient data for research purposes, budgeting is key. Costs vary widely, but common models include:
- Subscription Fees: Annual fees for continuous platform and data access, ideal for ongoing research.
- Per-Project Costs: Fees for data extraction and curation for a specific study, aligning costs with usage.
- FTE Support Models: Engaging a dedicated data expert for a set period on complex, long-term projects.
- Subsidized Academic Access: Reduced-cost or free access for internal academic researchers or public health projects.
- Fixed Government Processing Fees: Standardized fees for accessing public datasets, such as those from CMS in the USA.
Always consider the total cost of ownership, including computation, storage, and expertise, not just the initial access fee.
How does using anonymized data advance medical research?
The impact is transformative. Access to real-world patient information at scale:
- Accelerates Real-World Evidence (RWE) Studies: Provides insights into how treatments perform in diverse, everyday patient populations, closing the “Evidence Gap” between trials and reality.
- Improves Drug Safety Surveillance: Allows researchers to monitor the long-term safety of medications and identify rare side effects by tracking millions of patient records over years.
- Optimizes Clinical Trial Design: Helps identify suitable patient cohorts and refine inclusion/exclusion criteria, saving time and money in recruitment.
- Enables Precision Medicine: Integrating clinical and genomic data helps researchers understand how individual characteristics influence disease, paving the way for personalized therapies.
- Supports Public Health Research: Critical for understanding disease epidemiology, tracking outbreaks, and evaluating interventions, as seen with the N3C Data Enclave for COVID-19 research.
What’s the difference between anonymized and de-identified data?
This distinction is crucial when you’re looking for services that provide access to anonymized patient data for research purposes. Though often used interchangeably, they have distinct legal meanings.
De-identified data, a term defined by HIPAA in the USA, has 18 specific personal identifiers removed (name, address, etc.). While the risk is low, a theoretical possibility of re-identification can remain, which is why strict Data Use Agreements are essential.
Anonymized data, under Europe’s GDPR, sets a much higher, irreversible standard. The data must be processed so that an individual cannot be re-identified by any means. Because this is technically challenging, many European projects use pseudonymized data, where direct identifiers are replaced with artificial codes and the key is stored separately under high security.
In practice, most research data is de-identified or pseudonymized and handled under strict controls to protect patient privacy.
Act Now: Move From Data Hunting to Insights With Privacy‑First Access
You started this journey looking for services that provide access to anonymized patient data for research purposes. You’ve seen the landscape: from the privacy-first architecture of federated data networks to the analysis-ready datasets of centralized platforms.
The challenges of fragmentation, privacy, and quality are real. But the opportunities are transformative: accelerated drug findy, precision medicine, and real-world evidence that closes the gap between clinical trials and patient care.
The future belongs to privacy-preserving technologies and federated analysis. The old model of moving massive datasets is giving way to a smarter approach: bringing computation to the data. This shift open ups global data while respecting sovereignty, privacy, and local regulations, turning siloed information into research-ready insights without compromising security.
At Lifebit, this is exactly what we’ve built. Our next-generation federated AI platform provides secure, real-time access to global biomedical and multi-omic data. With built-in capabilities for harmonization, advanced AI/ML analytics, and federated governance, we power large-scale research across biopharma and public health. Our platform includes the Trusted Research Environment (TRE), Trusted Data Lakehouse (TDL), and R.E.A.L. (Real-time Evidence & Analytics Layer)—delivering real-time insights and secure collaboration across hybrid data ecosystems.
We believe every researcher deserves access to the data they need to make breakthroughs. Our federated platform is designed to turn you from a data seeker into an insight generator.
The data exists. The technology is ready. What will you find?

