The Best Resources for Rare Disease Research and Registries

10,000 Diseases, Millions Without Answers: The State of Rare Disease Research
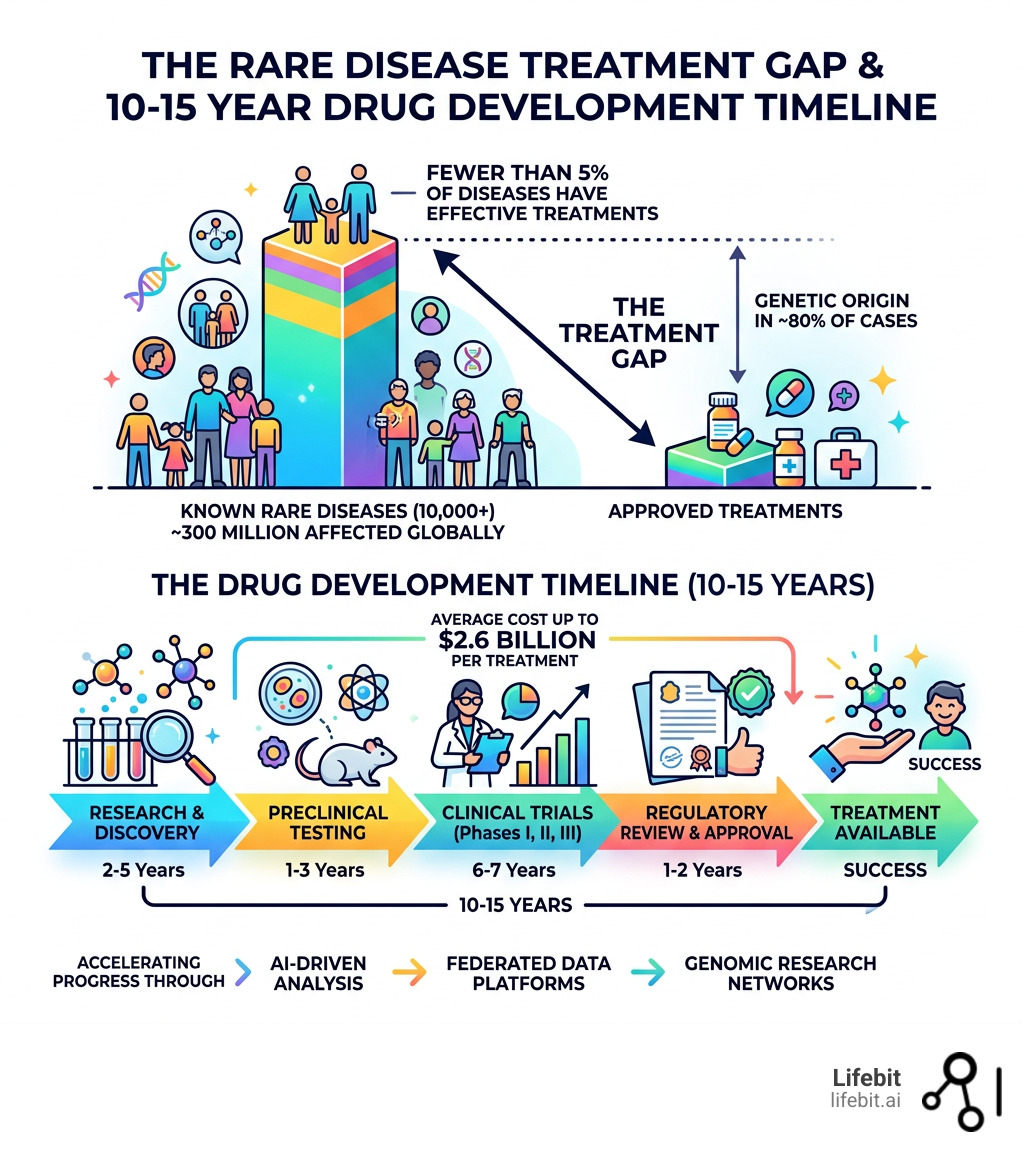
Rare disease research is one of the most urgent — and underserved — areas in modern medicine. While each individual condition affects a small number of people, the collective impact is staggering. Here’s a quick snapshot of where things stand:
| Fact | Figure |
|---|---|
| Known rare diseases | 10,000+ |
| People affected globally | ~300 million (8% of world population) |
| Americans affected | 25–30 million |
| Diseases with effective treatments | Fewer than 5% |
| Rare diseases with a genetic origin | ~80% |
| Average drug development timeline | 10–15 years |
| Cost to develop a single treatment | Up to $2.6 billion |
| Average time to diagnosis | 6–8 years |
| Misdiagnoses per patient | 2–3 on average |
Despite affecting hundreds of millions of people worldwide, the vast majority of rare diseases have no approved treatment. Most are genetic. Most begin in childhood, and tragically, 30% of children with a rare disease will not live to see their fifth birthday. For those who do survive, the journey is often defined by the “Diagnostic Odyssey”—a grueling period of years, sometimes decades, where patients bounce between specialists, undergoing invasive tests and receiving incorrect diagnoses before finally finding the molecular cause of their suffering.
The challenge isn’t just scientific; it’s structural and economic. The EveryLife Foundation for Rare Diseases recently estimated the total economic burden of rare diseases in the U.S. at nearly $1 trillion annually, including direct medical costs and indirect costs like lost productivity for caregivers. Because patient populations are tiny and scattered across the globe, data is often siloed in individual hospitals or countries. Funding is scarce because the traditional “blockbuster” drug model doesn’t easily apply to a disease that might only affect 50 people worldwide.
But that’s changing fast.
Advances in whole genome sequencing, AI-driven genomic analysis, federated data platforms, and international research networks are opening new doors. Researchers are finding diagnoses in cases that stumped specialists for years. New gene-disease associations are being confirmed through global data sharing. Drug repurposing is shrinking development timelines by using existing, safe medications for new indications. And patient advocates — including young voices shaping global policy — are accelerating progress from the ground up, demanding that research be patient-centric and data be accessible.
This guide pulls together the best resources available for rare disease research today: from NIH-funded networks and patient registries to genomic tools and clinical trial databases.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, with over 15 years of experience in computational biology, genomics, and federated data infrastructure — fields that sit at the heart of modern rare disease research. My work building scalable genomic analysis platforms has shown me both the enormous promise and the persistent data challenges that define this space. We are moving toward a future where a patient’s location no longer dictates their access to a cure.
Key terms for rare disease research:
Top Networks and Consortia for Rare Disease Research
Navigating the landscape of rare disease research can feel like searching for a needle in a global haystack. Because individual diseases affect so few people—defined in the U.S. as fewer than 200,000 individuals—collaboration isn’t just a “nice-to-have”; it is the only way to achieve statistical significance. Without pooling data from multiple sites, researchers cannot identify patterns or validate the efficacy of new treatments.
The Rare Diseases Clinical Research Network (RDCRN) stands as the gold standard for this collaborative model. Managed by the National Institutes of Health (NIH), the network recently entered its fifth funding cycle with approximately $26 million in grants awarded for 2025. This network currently comprises 21 research consortia involving over 300 clinical sites worldwide. Each consortium focuses on a cluster of related diseases, such as the Porphyrias Consortium, the Urea Cycle Disorders Consortium, or the Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR).
What makes the RDCRN so effective is its focus on “clinical trial readiness.” By conducting natural history studies—which track how a disease progresses over time without intervention—researchers create the baseline data needed to design efficient trials. These studies identify biomarkers and clinical endpoints that the FDA requires for drug approval. To date, this network has contributed to 11 FDA-approved treatments and supported hundreds of research projects. For those looking deeper into how genetic data is transforming these networks, our guide on Rare Disease Diagnosis Genomics explains the shift from analyzing symptoms to identifying the precise molecular cause of a condition.
The Undiagnosed Diseases Network (UDN) and Genomic Discovery
When a patient’s symptoms don’t match any known condition, they often turn to the Undiagnosed Diseases Network (UDN). This clinical research initiative uses advanced technologies like whole genome sequencing (WGS) and metabolomics to solve medical mysteries. The UDN brings together clinical and research experts from across the United States to solve the most challenging cases.
A recent scientific research paper on joint genomic analysis highlighted the power of analyzing large cohorts rather than isolated cases. By examining 4,236 individuals within the UDN, researchers used a statistical framework called RaMeDiES (Rare Mendelian Disease Evidence Support) to identify new gene-disease associations. This framework is revolutionary because it automates the process of looking for “de novo” mutations—genetic changes that appear in a child but are not present in either parent.
Key findings from these joint analyses include:
- New Diagnoses: Establishing 5 new diagnoses and 3 putative ones in previously “cold” cases that had remained unsolved for years.
- Gene Reclassification: Identifying H4C5 variants in patients with neurodevelopmental phenotypes and LRRC7 missense variants linked to hypotonia and ataxia, providing closure for families.
- Statistical Power: Using de novo mutation counts to confirm diagnoses that case-by-case analysis might miss due to the rarity of the variants.
Specialized Centers Advancing Rare Disease Research
Beyond national networks, several university-based centers are driving localized breakthroughs that have global implications:
- UCLA California Center for Rare Diseases (CCRD): This center integrates clinical care with research-only testing to solve undiagnosed genetic disorders. They focus heavily on drug repurposing—using existing, safe medications to treat rare conditions based on a patient’s genetic profile. This “precision medicine” approach ensures that patients receive the right drug for their specific mutation. If you are a patient or clinician seeking guidance, you can Contact UCLA CCRD directly.
- Notre Dame Center for Rare and Neglected Diseases: This center is unique for its focus on molecular mechanisms and its commitment to training the next generation of patient advocates. Their research has even uncovered surprising links, such as the rare disease drug nitisinone making human blood deadly to mosquitoes, which could have massive implications for malaria prevention.
- The Jackson Laboratory (JAX) Rare Disease Translational Center: JAX excels at creating “mouse models” that mimic human rare diseases. By using CRISPR/Cas9 technology, they can insert a specific human genetic mutation into a mouse, allowing scientists to test gene therapies in a living system before moving to human trials. Their work on the PGAP3-CDG project, a condition affecting only 65 people worldwide, identified a promising therapy candidate in less than four months.
- GREGoR Consortium: Short for Genomics Research to Elucidate the Genetics of Rare Diseases, this group focuses on accelerating genomics for rare diseases by applying new technologies like long-read sequencing and RNA sequencing to cases that remain unsolved even after standard whole-exome sequencing. Their goal is to uncover the “dark matter” of the genome that traditional tests miss.
Essential Patient Registries and Clinical Trial Databases
In rare disease research, data is the most valuable currency. For many conditions, a patient registry is the first step toward a cure. These databases collect health information, family histories, and genetic data from patients to help researchers understand disease prevalence, progression, and the impact of various treatments.
The National Organization for Rare Disorders (NORD) hosts the IAMRARE® platform, a powerful tool that allows patient advocacy groups to build their own registries. Unlike traditional clinical databases owned by hospitals, these registries often prioritize patient data ownership, ensuring that families have a say in how their information is used and who can access it. This model empowers patients to become active partners in the research process rather than just subjects.
For a deep dive into how these systems work, see our Rare Diseases Registry Complete Guide. Registries provide four primary benefits for rare disease research:
- Generating clinical-grade data: This data is essential for regulatory submissions to the FDA or EMA, providing the “real-world evidence” needed for drug approval.
- Attracting biopharma interest: By proving a trial-ready population exists and is organized, registries make it much more attractive for pharmaceutical companies to invest in a specific disease.
- Shortening the diagnostic odyssey: Registries help identify common symptoms that can be shared with primary care physicians to speed up diagnosis for future patients.
- Informing research design: Through patient-reported outcomes, researchers can learn which symptoms matter most to patients, ensuring that clinical trials measure meaningful improvements in quality of life.
The Role of FAIR Data Principles
To be truly effective, rare disease data must follow the FAIR principles: Findable, Accessible, Interoperable, and Reusable. Because data is so scarce, it cannot be trapped in a single institution. Initiatives like the Rare Disease Cures Accelerator-Data and Analytics Platform (RDCA-DAP) are working to aggregate data from various registries and clinical trials into a single, searchable platform. This allows researchers to run complex queries across multiple diseases to find common biological pathways.
How to Participate in Rare Disease Research Trials
Participation is the engine of progress. While ClinicalTrials.gov is the primary federal database for searching studies, it can be overwhelming for families. Many organizations now offer more accessible pathways. For instance, some independent research sites provide a simple Intake Form for Potential Participants to match patients with ongoing studies based on their specific diagnosis and location.
Advocacy is also evolving from local support groups to global policy influencers. Youth leaders like Liam McCarthy are proving that patients can influence global policy. McCarthy’s work with Rare Diseases International (RDI) contributed to the adoption of the World Health Assembly Resolution on Rare Diseases, which urges 194 member states to strengthen their rare disease frameworks and integrate rare disease care into universal health coverage. This highlights that participating in research isn’t just about taking a drug; it’s about lending your voice to a movement that strengthens rare disease research groups worldwide.
Breakthrough Technologies: AI, Genomics, and Drug Repurposing
We are entering an era where technology is finally catching up to the complexity of rare diseases. The convergence of high-throughput sequencing and massive computing power is transforming rare disease research from a game of chance into a data-driven science. Three areas are currently revolutionizing the field:
1. Whole Genome Sequencing (WGS) as the New Standard While older tests looked only at specific genes (exomes), WGS looks at the entire DNA sequence, including the non-coding regions that regulate gene expression. This is crucial because 72% of rare diseases have a genetic component, and many mutations lie outside the protein-coding regions. Institutions like UCLA Health are collaborating with industry leaders to make WGS the backbone of rare disease diagnosis. This allows for “molecular etiology”—finding the exact genetic “typo” causing a disease, which is the first step toward developing a targeted therapy.
2. AI and Statistical Frameworks Artificial Intelligence is the only tool capable of processing the petabytes of data generated by genomic sequencing. Tools like RaMeDiES use machine learning to analyze “de novo” (new) mutations across thousands of patients simultaneously. This allows researchers to spot patterns that are invisible when looking at just one family. Furthermore, Natural Language Processing (NLP) is being used to scan millions of electronic health records (EHRs) to identify undiagnosed patients who fit the profile of a specific rare disease, effectively “flagging” them for clinical review before their symptoms worsen.
3. Drug Repurposing and Virtual Screening Developing a new drug from scratch takes 15 years and billions of dollars. Drug repurposing—finding new uses for existing FDA-approved drugs—can cut that time in half and reduce costs by 80%. Events like the Findacure Conference on Drug Repurposing showcase how AI can predict which existing medications might “fix” the biological pathway broken by a rare genetic mutation. For example, a drug originally developed for arthritis might be found to block the specific inflammatory pathway active in a rare neurological condition.
4. Gene-Targeted Therapies and N-of-1 Trials The NeuroNEXT network and other specialized groups are currently accelerating “ultra-rare” gene therapies. These include antisense oligonucleotides (ASOs), which act like “molecular Band-Aids” to cover up genetic errors, and CRISPR-based editing, which can permanently correct a mutation. We are even seeing the rise of “N-of-1” trials, where a drug is custom-designed for a single individual. The goal is to move from treating symptoms to actually correcting the genetic code, providing a permanent cure rather than lifelong management.
Frequently Asked Questions about Rare Disease Research
How many rare diseases currently exist?
There are more than 10,000 known rare diseases. While each disease is individually rare—affecting fewer than 0.05% of the population—collectively they are quite common. About 1 in 10 Americans (25-30 million people) and 8% of the global population live with a rare condition. This means that while the disease is rare, the “rare disease patient” is not.
What is the success rate for rare disease treatments?
Currently, fewer than 5% of rare diseases have an FDA-approved treatment. The high cost (up to $2.6 billion per drug) and the 10-15 year development timeline are major hurdles. However, collaborative networks like the RDCRN and strategies like drug repurposing are beginning to increase this success rate. The Orphan Drug Act of 1983 has also helped by providing financial incentives for companies to develop treatments for small patient populations.
Where can families find support for unstudied diseases?
If a disease is so rare that it doesn’t have a dedicated research consortium, families should contact the NIH’s Genetic and Rare Diseases Information Center (GARD). They provide specialists who can be reached at 888-205-2311 to help navigate the complex landscape of rare disease prevalence and find relevant advocacy groups. They can also help patients apply to the Undiagnosed Diseases Network (UDN).
Why is international data sharing so important for rare diseases?
Because any single country may only have one or two patients with a specific rare mutation, researchers need to look globally to find enough cases to conduct a valid study. International data sharing allows scientists to confirm that a specific genetic variant is indeed the cause of a disease by finding other patients with the same variant and the same symptoms halfway across the world.
How does newborn screening impact rare disease research?
Newborn screening is a public health program that tests infants shortly after birth for a list of conditions that can cause health problems if not treated early. For rare disease research, this provides a critical window for early intervention. Research into new screening methods, such as using Whole Genome Sequencing at birth, could potentially identify thousands of rare conditions before symptoms even appear, allowing for preventative treatments that could save lives.
Conclusion: Scaling Global Rare Disease Research
The future of rare disease research lies in breaking down data silos and embracing a global, collaborative mindset. For too long, life-saving data has been trapped behind institutional firewalls or national borders. At Lifebit, we believe that a patient’s geography should never limit their access to a cure. The technology now exists to connect the world’s biomedical data without compromising patient privacy or data security.
Our next-generation federated AI platform enables researchers to securely access and analyze global biomedical and multi-omic data without the data ever leaving its home jurisdiction. This is a paradigm shift: instead of moving massive, sensitive datasets to the researcher, we move the analysis to the data. This ensures compliance with strict regulations like GDPR and HIPAA while still allowing for the large-scale analysis required to solve rare diseases.
By using our Trusted Research Environment (TRE) and R.E.A.L. (Real-time Evidence & Analytics Layer), biopharma companies, academic institutions, and government agencies can collaborate on large-scale, compliant research. This federated approach is essential for rare diseases, where the data needed to save a child’s life in New York might be sitting in a lab in Singapore, London, or Sydney. By connecting these dots, we can accelerate the path from diagnosis to treatment.
We are committed to providing the infrastructure that turns fragmented data into life-saving insights. The era of the “isolated researcher” is over; the era of global, federated discovery has begun. Explore how the Lifebit Platform is powering the next wave of genomic discovery and helping to bring answers to the millions of people still waiting for a cure.


