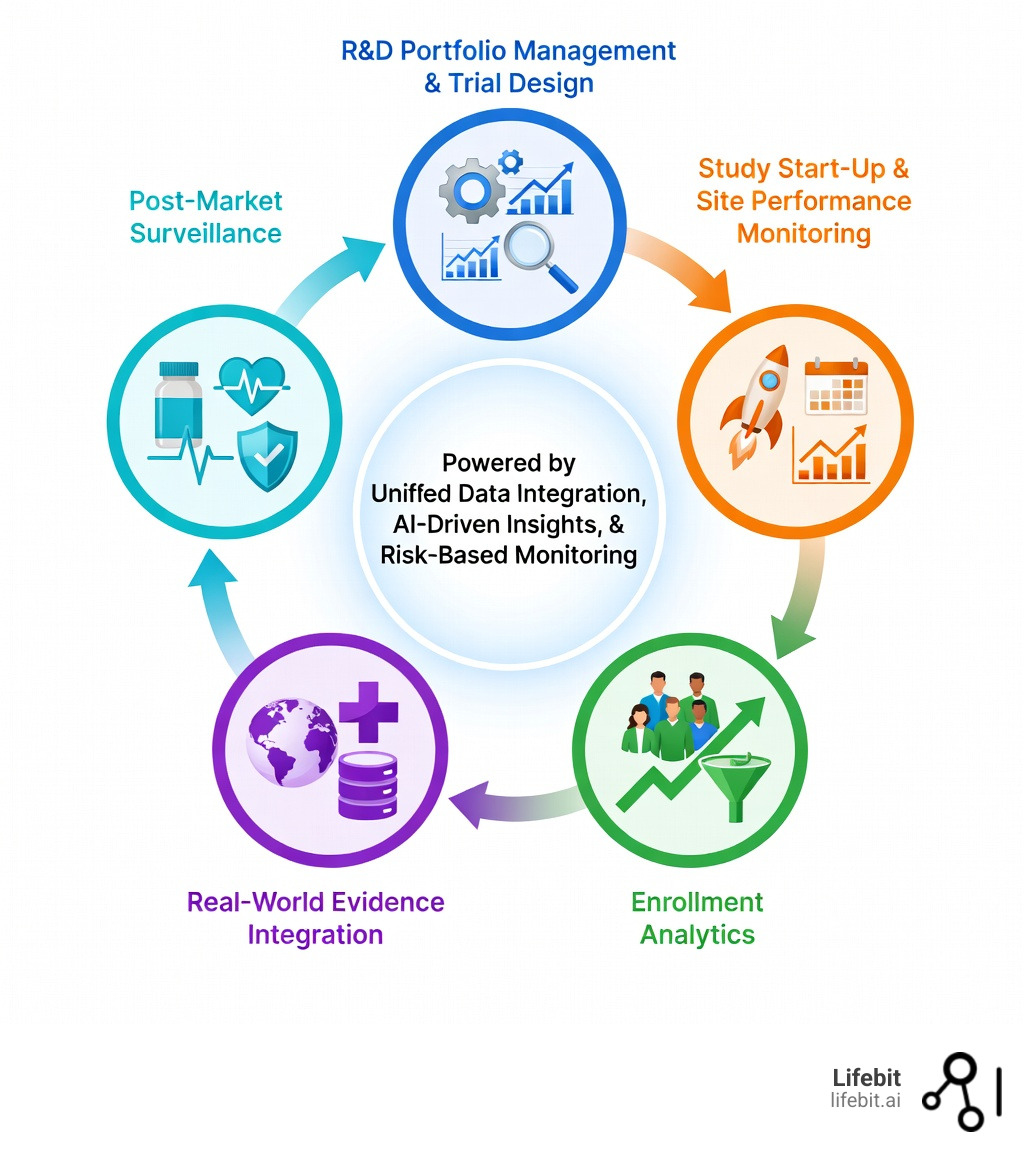
The data journey from clinical trials to post-market surveillance

Why Clinical Development Analytics Is Critical for Modern Drug Development
Clinical development analytics is the practice of integrating data from multiple sources—electronic data capture (EDC), clinical trial management systems (CTMS), electronic patient-reported outcomes (ePRO), and real-world data—to provide real-time operational and clinical insights across the entire drug development lifecycle.
Key capabilities include:
- Cross-enterprise visibility: Merge data from disparate systems into a single warehouse for unified reporting
- Risk-based monitoring (RBM): Track key risk indicators (KRIs) and site performance metrics to identify issues before they impact timelines
- Predictive analytics: Use AI and machine learning to model success rates, optimize enrollment, and simulate trial scenarios
- Real-world evidence integration: Leverage RWD for external control arms, natural history studies, and post-market surveillance
- Regulatory compliance: Align with ICH E6 R3, FDA diversity plans, and TransCelerate guidelines
The stakes are high. New molecular entity (NME) development success rates have fallen to just 6.1%, while R&D cycle times now exceed 15 years from discovery to approval. Phase III trial durations have increased by 47% over the last two decades, and every month a program can accelerate to market adds an estimated $5-6M in net present value per billion dollars of peak year sales.
These trends make clinical development analytics essential for sponsors, CROs, and clinical teams trying to extract efficiency from increasingly complex trials. The right analytics approach can reduce trial duration by one-third, improve site performance, and enable faster, more informed decisions across the R&D portfolio.
I’m Dr. Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where I’ve spent over 15 years building platforms that bring clinical development analytics to life through federated AI and secure genomic data integration. My work centers on empowering pharma and public sector teams to analyze diverse, siloed datasets in situ—without moving data—so they can generate decision-grade evidence faster and more compliantly.
What is Clinical Development Analytics?
At its core, clinical development analytics is about breaking down the walls between different types of data. In a traditional trial, your patient data lives in the EDC, your operational timelines are in the CTMS, and your safety signals might be tucked away in a separate pharmacovigilance database. This fragmentation is a recipe for delays, as teams spend more time reconciling spreadsheets than making strategic decisions.
By integrating these streams, we create a “single source of truth.” This isn’t just a buzzword; it’s the difference between seeing a problem three months too late and seeing it in real-time. Modern solutions provide cross-enterprise visibility, allowing executives to see performance across therapeutic areas and study managers to drill down into specific site queries.
The Data Ecosystem in Clinical Development
To understand the scope of clinical development analytics, one must look at the diverse data types it harmonizes:
- Operational Data: Metrics from CTMS regarding site activation, investigator performance, and monitoring visit reports.
- Clinical Data: Patient-level data from EDC systems, including lab results, vital signs, and adverse events.
- Patient-Reported Data: Insights from ePRO and eCOA (Electronic Clinical Outcome Assessment) that provide the patient’s perspective on treatment efficacy and quality of life.
- Biomarker and Genomic Data: High-dimensional data that helps in patient stratification and understanding the molecular mechanism of drug response.
- Real-World Data (RWD): Information from electronic health records (EHRs), insurance claims, and pharmacy records that provide context outside the controlled trial environment.
For a deeper dive into how this data is structured, you can explore our Clinical Research Data Analytics Guide and our specific insights on Clinical Trial Data Analytics.
The goal is to gain operational and clinical insights that evaluate the performance of clinical studies. This involves merging data into a single warehouse to provide a cross-enterprise view of operational data. When we talk about “decision-grade evidence,” we mean data that is clean, standardized (often using CDISC standards like SDTM and ADaM), and robust enough to satisfy both internal stakeholders and external regulators.
Maximizing ROI: Key Benefits and KPIs of clinical development analytics
In the current landscape, efficiency isn’t just a goal—it’s a survival mechanism. With a 6.1% NME success rate (2019-2023), the margin for error is razor-thin. When we look at the financial impact, every month we can shave off a development timeline adds roughly $5-6 million in net present value (NPV) for every billion dollars of peak year sales. For blockbuster drugs, this acceleration can represent hundreds of millions in additional revenue over the product’s lifecycle.
To capture this value, we must move beyond basic reporting. High-performing teams use a Clinical Analytics Software Ultimate Guide to establish sophisticated Key Performance Indicators (KPIs) that track both speed and quality.
Tracking clinical development analytics at the Portfolio Level
At the highest level, we are looking at the health of the entire pipeline. This requires:
- Success Rate Modeling: Using logistic regression or deep learning to predict which programs are likely to reach approval based on historical benchmarks and current trial signals.
- Productivity Metrics: Measuring the spend per approval to ensure resources are allocated to the highest-value assets. This includes analyzing the “cost per patient” across different therapeutic areas to identify outliers.
- Value Creation: Tracking the sales of products approved in the last five years relative to the R&D investment, providing a clear picture of the return on innovation.
- Cycle Time Analysis: Breaking down the time spent in each phase (Phase I, II, III) and identifying the “white space”—those gaps between clinical phases that represent a significant portion of development time.
By analyzing these gaps, we can often identify “at-risk” activities that can be started earlier to shorten transitions. For example, starting CMC (Chemistry, Manufacturing, and Controls) scale-up activities during Phase II rather than waiting for Phase III results can save 6-12 months. For more on this, see our guide on Biopharma Data Analytics.
Site Performance and Study Start-up Metrics
The most granular level of clinical development analytics happens at the site. If a site isn’t enrolling patients or is drowning in data queries, the whole study slows down.
| KPI Category | Metric to Track | Why It Matters |
|---|---|---|
| Study Start-up | Site initiation cadence | Faster initiation is often a better predictor of duration than enrollment rate. |
| Enrollment | Screen failure trends | High screen failures can signal overly restrictive eligibility criteria or poor site selection. |
| Data Quality | Query resolution time | Slow resolution delays statistical analysis and reporting, pushing back database lock. |
| Site Performance | Activation to first patient | Identifies bottlenecks in site contracting, IRB approval, and staff training. |
| Patient Retention | Drop-out rate by site | High attrition can bias results and require additional recruitment, increasing costs. |
By proactively monitoring these, we can manage non-performing sites and prioritize monitor visits based on risk rather than a fixed schedule. This is the heart of our Clinical Research Analytics Complete Guide.
How AI and Machine Learning Solve the 15-Year R&D Bottleneck
If it takes 15 years to get a drug to market, the system is broken. AI and machine learning (ML) are the “force multipliers” we use to fix it. We aren’t just talking about chatbots; we’re talking about discrete event simulation, predictive modeling, and natural language processing (NLP) applied to massive datasets.
Predictive Modeling for Site Selection and Enrollment
One of the most powerful applications is enrollment simulation. By using historical data from thousands of previous trials and current site performance, we can develop strategic enrollment plans that minimize costs and provide realistic timelines. Instead of guessing when a trial will finish, we can simulate thousands of scenarios to find the most efficient path.
ML models can also predict which sites are likely to be “super-enrollers” and which will fail to recruit a single patient. By analyzing factors such as local disease prevalence, competing trials in the area, and historical site performance, sponsors can optimize their site footprint before the first patient is even screened.
Generative AI and Data Literacy
Generative AI is also changing how study teams interact with data. Imagine a clinical manager asking an AI assistant, “Which sites in Western Europe have the highest query rates and how is that impacting our database lock date?” The AI can instantly query the integrated data warehouse, generate a visualization, and provide a summary of the impact. This real-time interaction improves data literacy across the organization and allows for immediate course correction.
Furthermore, GenAI is being used to automate the drafting of Clinical Study Reports (CSRs) and Trial Master File (TMF) indexing. By reducing the manual burden of documentation, clinical teams can focus on high-level strategy and patient safety. For those interested in the technical side, our resources on Advanced Analytics Healthcare and the Advanced Analytics Ultimate Guide explain how these models are built and validated.
Reducing Trial Complexity to Slash Duration by One-Third
We often see “complexity creep” in trial design. More endpoints, more eligibility criteria, and more procedures are added “just in case.” However, research published in Scientific Reports shows that a 10 percentage point increase in a Trial Complexity Score correlates with a one-third increase in overall trial duration.
Phase III trial duration has already increased by 47% over the last 20 years. To fight this, we use clinical development analytics to perform protocol optimization. By analyzing which endpoints are actually necessary for regulatory approval and market access, we can reduce patient and investigator burden. This is particularly vital in Oncology Analytics, where trials are notoriously complex, often involving multiple biopsies, frequent imaging, and intricate dosing schedules.
The Impact of Protocol Amendments
Analytics also helps in reducing the need for protocol amendments, which are a major source of delay. A typical Phase III protocol amendment can cost upwards of $500,000 and add months to the timeline. By using predictive analytics to “stress-test” a protocol against real-world patient data before it is finalized, sponsors can identify if their eligibility criteria are too restrictive to allow for successful recruitment.
Strategic Trade-offs: Phase Skipping vs. Aggregation
When trying to accelerate time to market, sponsors often face a choice: skip a phase or aggregate phases (like a Phase 1/2 trial).
- Phase Skipping: Our research indicates that skipping a dedicated phase (like a Phase 1 in a specific tumor type when you already have dosing data) can improve the historical success rate (4.5% vs 1.2%) and significantly reduce time to market. This is often seen in rare diseases or oncology where the unmet need is high.
- Phase Aggregation: Interestingly, aggregating phases can sometimes be slower overall. While it seems efficient on paper, it often leads to increased FDA scrutiny, more complex statistical considerations, and longer “internal” pauses as teams analyze data between the Phase 1 and Phase 2 portions of the same study.
Accelerated approvals have historically reduced time to market for oncology drugs by 28%, though this has recently narrowed to 18% as the FDA increases scrutiny on confirmatory trials. Navigating these trade-offs requires a robust Health Data Analytics Complete Guide.
Modernizing Strategies with RWD and Digital Clinical Measures
The future of clinical development analytics isn’t just in the clinic—it’s in the real world. Real-world data (RWD) and novel digital clinical measures are revolutionizing how we evaluate health and treatment efficacy.
The V3+ Framework for Digital Measures
We use the V3+ Framework to ensure that data from wearables and sensors is fit-for-purpose. This involves:
- Verification: Ensuring the hardware accurately measures the physical phenomenon (e.g., steps, heart rate).
- Analytical Validation: Confirming the algorithm correctly processes the raw data into a digital biomarker.
- Clinical Validation: Proving the digital biomarker relates to a clinical endpoint or patient state.
- Usability: Ensuring patients can and will use the device as intended.
These digital clinical measures capture symptoms that traditional clinic visits miss, such as sleep quality or continuous mobility, providing a more holistic view of patient health. This is a key part of our focus on Real-Time Healthcare Analytics.
Integrating RWD into clinical development analytics
RWD is no longer just for post-market surveillance. We are now using it for:
- External Control Arms (ECA): Using healthcare data to supplement or replace a placebo arm. This is transformative for rare diseases where recruiting a control group is ethically or practically impossible. By matching trial patients with “digital twins” from RWD, we can generate comparative evidence.
- Target Trial Emulation: Designing non-interventional studies that “emulate” a randomized trial. This allows researchers to evaluate causal effects using observational data, providing insights into how a drug might perform in a broader, more diverse population than the typical trial cohort.
- Inclusion/Exclusion Optimization: Testing how different criteria would impact the available patient pool. For instance, if we relax a creatinine clearance requirement by 5%, how many more patients become eligible? RWD provides the answer in seconds.
By following the PRINCIPLED framework (Prototypical, Relevant, Independent, Consistent, Integrated, Precise, Legitimate, Ethical, and Diverse) developed with the FDA, we can ensure these studies are robust and avoid the biases often associated with observational data.
Overcoming Data Quality and Regulatory Challenges
The biggest hurdle to effective clinical development analytics isn’t the technology—it’s the data quality and the evolving regulatory landscape. Standardization of site names, investigator details, and disease definitions is a constant battle. Without clean data, even the most advanced AI models will produce “garbage in, garbage out” results.
Navigating ICH E6 R3 and Diversity Mandates
Regulatory changes are shifting the goalposts. The ICH E6 R3 guidelines emphasize a “proportional” approach to quality management, encouraging sponsors to focus on the data that truly matters to patient safety and trial integrity. This aligns perfectly with Risk-Based Monitoring (RBM), where analytics identify high-risk sites or unusual data patterns (such as identical blood pressure readings across multiple patients) that require intervention.
Furthermore, the FDA’s diversity action plans now require sponsors to set enrollment goals for underrepresented populations. Clinical development analytics is essential here to track demographic data in real-time and identify if a trial is failing to meet its diversity targets. If a gap is identified, teams can pivot their recruitment strategy to sites in more diverse geographic areas.
Federated Analytics: A Solution for Data Privacy
As trials become more global, data residency laws (like GDPR in Europe or HIPAA in the US) make it difficult to centralize data for analysis. This is where federated analytics comes in. Instead of moving sensitive patient data to a central server, the analytics models are sent to where the data resides. The models are trained locally, and only the aggregated, de-identified insights are sent back. This approach ensures compliance with the strictest privacy laws while still allowing for large-scale, multi-national research.
To manage these complexities, we implement these proactive approaches directly into our Clinical Development Platform. For those conducting inferential studies, we highly recommend reviewing the scientific research on causal effects in RWD to ensure your methodology stands up to regulatory scrutiny.
Frequently Asked Questions about Clinical Development Analytics
How does clinical development analytics improve trial success rates?
By identifying “at-risk” programs early through success rate modeling and identifying operational bottlenecks (like slow site initiation or high screen failures), teams can intervene before a study fails. It allows for “failing fast” on low-potential assets and doubling down on winners.
What are the most critical KPIs for clinical study managers?
The most critical KPIs include site initiation cadence, enrollment rates vs. plan, screen failure trends, and query resolution time. These provide the most immediate signal of whether a trial will meet its database lock deadline.
How is Generative AI currently used in clinical development?
Currently, GenAI is used to streamline data visualization, improve data literacy for non-technical study teams, and help in the drafting of clinical study reports. It’s also being explored for synthetic data generation and protocol drafting to reduce manual effort.
Conclusion
The journey from a clinical trial to post-market surveillance is fraught with data silos and rising costs. At Lifebit, we believe the solution lies in a next-generation federated AI platform. Our technology enables secure, real-time access to global biomedical and multi-omic data without the need to move sensitive information.
With our built-in capabilities for harmonization, advanced AI/ML analytics, and federated governance, we power large-scale research across biopharma, governments, and public health agencies. Whether you are using our Trusted Research Environment (TRE) for secure collaboration or our Real-time Evidence & Analytics Layer (R.E.A.L.) for safety surveillance, our goal is to deliver the insights you need to bring therapies to patients faster.
Secure your clinical data journey with Lifebit and turn your clinical development analytics into a strategic advantage.

