The Ultimate Guide to AI-Enabled Precision Medicine

Why Precision Medicine AI Is Changing How We Treat Every Patient
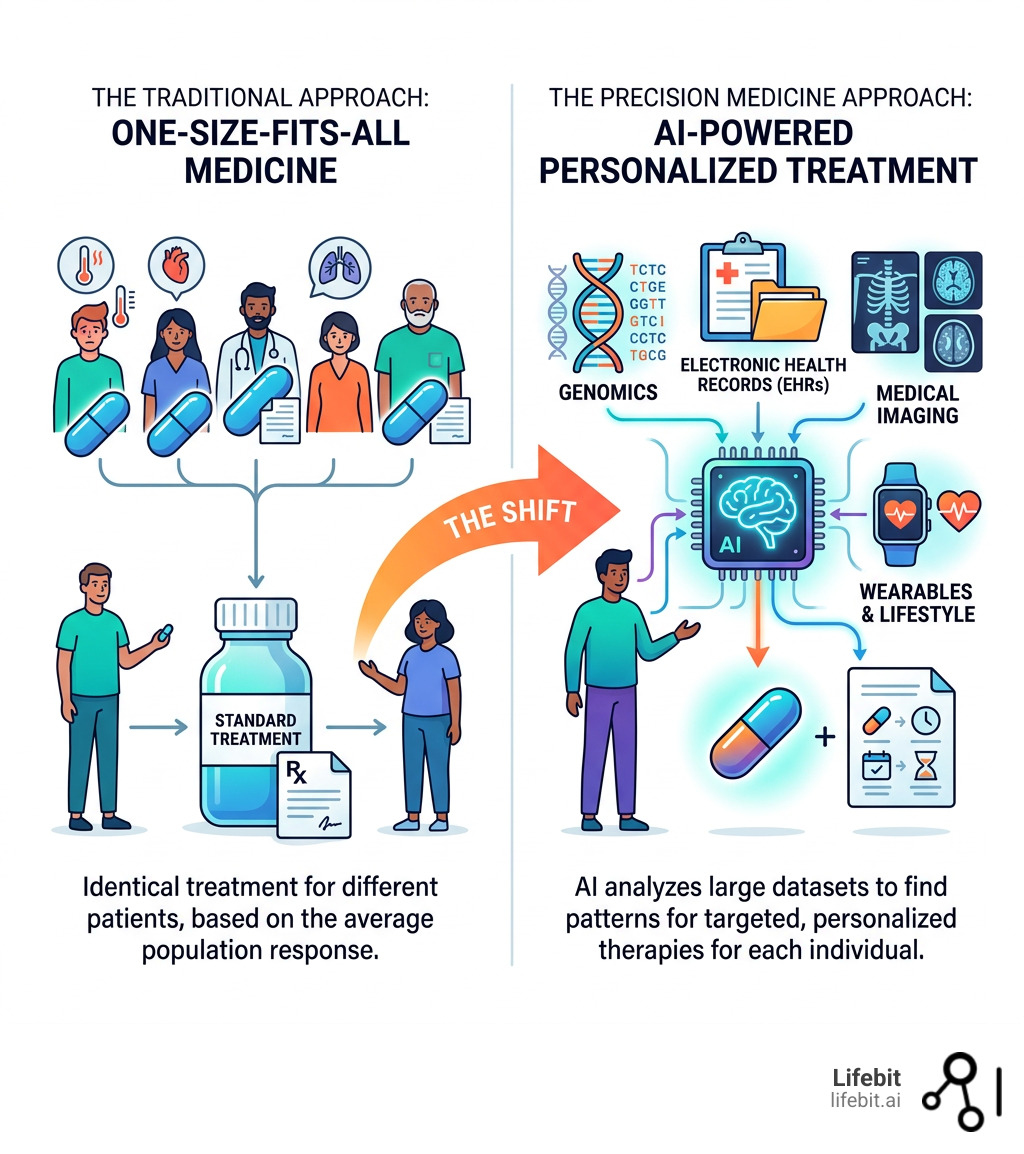
Precision medicine AI combines artificial intelligence with personalized healthcare to match the right treatment to the right patient — based on their unique biology, lifestyle, and environment.
Here’s what that means in practice:
- What it is: AI analyzes genomic, clinical, imaging, and lifestyle data to build a complete picture of each patient
- Why it matters: Traditional medicine treats the average patient — precision medicine treats your patient
- What AI adds: Speed, pattern recognition, and the ability to process data no human team could handle alone
- Who benefits: Patients get more accurate diagnoses and targeted treatments; clinicians get better decision support; researchers find drug targets faster
Nearly two-thirds of doctors already see AI’s potential to improve healthcare, according to an AMA survey of more than 1,000 physicians. The gap between potential and reality, however, remains wide — and understanding why is the first step to closing it.
This guide breaks down exactly how AI is reshaping precision medicine: what works, what’s still hard, and where the field is heading.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, with over 15 years of experience in computational biology, genomics, and federated AI platforms — fields that sit at the heart of precision medicine AI. I’ve spent my career building the infrastructure that makes secure, large-scale biomedical data analysis possible, and I’ll share what I’ve learned throughout this guide.
Explore more about Precision medicine AI:
What is Precision Medicine AI and Why Does Traditional Healthcare Need It?
For decades, the “gold standard” of medicine was built on the average. If a drug worked for 60% of people in a clinical trial, it was deemed a success and prescribed to everyone. But what about the other 40%? Traditional healthcare often relies on a “trial and error” approach that is slow, expensive, and sometimes dangerous for patients with unique genetic makeups. This inefficiency isn’t just a clinical problem; it’s an economic one. Adverse drug reactions (ADRs) are estimated to cost billions annually and are a leading cause of hospitalization.
Precision medicine AI represents a fundamental shift. Instead of treating the disease, we treat the individual. By integrating AI and Precision Health principles, we can move from reactive care to proactive, targeted interventions. AI acts as the “brain” capable of making sense of the five Vs of healthcare big data:
- Volume: The sheer amount of data generated per patient, from terabytes of genomic sequencing to years of clinical notes.
- Velocity: The speed at which data is generated, particularly from real-time monitoring devices and wearables.
- Variety: The diverse formats of data, including structured lab results, unstructured physician notes, and high-resolution medical imaging.
- Veracity: The challenge of ensuring data quality and accuracy in a messy clinical environment.
- Value: The ultimate goal of turning this raw information into actionable clinical insights.
The core difference lies in how we use information. Conventional methods rely on limited clinical observations and broad population stats. In contrast, AI-powered systems use machine learning to identify subtle patterns across millions of data points, offering real-time clinical decision support (CDS) that is tailored to a patient’s specific sensitivity to a drug or susceptibility to a disease. This allows for “stratified medicine,” where patients are grouped by biological similarities rather than just symptoms.
| Feature | Conventional Healthcare | AI-Powered Precision Medicine |
|---|---|---|
| Treatment Strategy | One-size-fits-all (The “Average” Patient) | Personalized (The Individual Patient) |
| Data Usage | Limited clinical notes and basic labs | Multimodal (Genomics, EHR, Imaging, Wearables) |
| Diagnosis | Reactive (after symptoms appear) | Predictive (early risk identification) |
| Drug Selection | Trial and error based on guidelines | Targeted based on pharmacogenomics |
| Monitoring | Periodic clinical visits | Real-time, continuous monitoring via IoT |
| Economic Impact | High waste due to ineffective treatments | Optimized spending on targeted therapies |
Core Components of AI-Powered Personalized Systems
To deliver truly personalized care, an AI system needs more than just a smart algorithm; it needs a massive, high-quality “diet” of data. These systems are built on four foundational pillars that work in concert to transform raw biological signals into clinical action:
Multimodal Data Integration: This is the process of weaving together different types of information. It’s not just about your DNA; it’s about your Electronic Health Records (EHRs), medical imaging (MRIs, CT scans), and even data from your smartwatch. The challenge here is “data harmonization”—ensuring that a blood pressure reading from a hospital in London can be compared accurately with one from a clinic in New York. AI models use “early fusion” or “late fusion” techniques to combine these disparate data streams into a single patient profile.
Machine Learning (ML) Algorithms: These are the engines that find the “needle in the haystack.”
- Deep Learning: Used for image recognition in radiology and pathology.
- Natural Language Processing (NLP): Used to extract meaning from millions of unstructured doctor’s notes.
- Transformers: The same tech behind ChatGPT is now being used to predict how proteins will fold or how a specific genetic mutation might lead to disease.
Clinical Decision Support (CDS) Tools: These tools act as a co-pilot for doctors. They pull from all available data to provide evidence-based, patient-specific recommendations at the point of care. A CDS might flag that a specific chemotherapy drug is likely to be toxic for a patient based on a rare genetic variant found in their profile, suggesting an alternative before the first dose is even administered.
Secure Infrastructure: Because health data is incredibly sensitive, these systems require secure environments like Trusted Research Environments (TREs). These are highly secure, cloud-based platforms where researchers can analyze data without ever actually “seeing” or moving the raw files, ensuring compliance with regulations like GDPR and HIPAA.
Scientific research on AI in healthcare highlights that the true power of precision medicine AI comes from “augmented intelligence”—AI that supports and enhances human expertise rather than replacing it. By automating the data-crunching, AI frees up clinicians to focus on the human element of care.

Clinical Applications: Improving Outcomes in Oncology and Beyond
The impact of precision medicine AI is most visible in complex fields where biological “noise” makes traditional treatment difficult. By using AI for precision medicine, we are seeing major breakthroughs in several key areas.
Cardiology: Predicting the Unpredictable
In Cardiology, AI models are now being used to identify low left ventricular ejection fraction from standard ECGs—something that previously required much more invasive or expensive testing like echocardiograms. AI can detect “silent” signals in a heartbeat that a human eye would miss, allowing for the early detection of heart failure or atrial fibrillation. Furthermore, AI-driven risk scores are replacing traditional calculators, incorporating lifestyle data and genetic predispositions to predict heart attacks years in advance.
Neurology: Early Detection of Neurodegeneration
In Neurology, automated analysis of speech and gait patterns is helping to predict the onset of conditions like Alzheimer’s and Parkinson’s years before clinical symptoms become obvious. For instance, subtle changes in the cadence of a person’s voice or the length of their stride can be captured by smartphone sensors and analyzed by AI to flag early neurodegenerative changes. This opens a window for early intervention that was previously impossible.
Rare Diseases: Ending the Diagnostic Odyssey
For patients with rare genetic diseases, the “diagnostic odyssey” can last an average of five to seven years. AI is shortening this to weeks. By using automated variant prioritization, AI can scan a patient’s entire genome and compare it against thousands of known disease-causing mutations, highlighting the most likely culprit for a clinician to review. This is life-changing for pediatric patients where early diagnosis can mean the difference between a normal life and permanent disability.
Research on AI for tumor diagnosis shows that AI can interpret complex imaging data from PET-CT and MRI scans with a level of precision that helps clinicians catch diseases earlier and monitor treatment response in real-time.
Using Precision Medicine AI for Targeted Oncology
Oncology is the “front line” of the precision revolution. Cancer isn’t one disease; it’s thousands of different genetic variations. AI helps by enabling:
- Disease Subtyping: Moving beyond “lung cancer” to identify the specific molecular subtype of a tumor. Research on disease subtyping explains how this leads to tailored treatment plans that are far more effective than broad-spectrum chemotherapy. For example, AI can identify which patients will respond to immunotherapy versus those who need targeted kinase inhibitors.
- Clinical Trial Matching: AI can scan a patient’s genetic profile and instantly match them with the most relevant clinical trials globally. This is critical because many precision drugs are only available through trials, and manual matching is often too slow for late-stage patients.
- Radiogenomics: This emerging field uses AI to link imaging features (like the shape or texture of a tumor) with gene expression. This allows doctors to predict how aggressive a cancer might be or how it might mutate without needing multiple invasive biopsies.
- Liquid Biopsies: AI is being used to detect “circulating tumor DNA” (ctDNA) in a simple blood draw. This allows for the monitoring of cancer recurrence months before a tumor would be visible on a traditional scan.
Precision Medicine AI in Pharmacogenomics and Drug Response
Have you ever wondered why a standard dose of a common drug like warfarin works perfectly for one person but causes dangerous bleeding in another? The answer is in your genes.
Pharmacogenomics is the study of how genes affect a person’s response to drugs. Research on AI in pharmacogenomics is helping us use AI in personalized medicine to predict drug efficacy and minimize adverse drug reactions (ADRs). By analyzing multi-omics data, AI can predict how a drug will be absorbed, distributed, metabolized, and excreted (the “ADME” process).
This isn’t just theoretical. Systems are now being developed to provide real-time, genotype-guided dosing recommendations directly within the hospital workflow. For example, AI can flag if a patient has a CYP2D6 gene variant that makes them a “poor metabolizer” of certain antidepressants, allowing the doctor to adjust the dose or switch medications immediately, avoiding weeks of ineffective treatment and side effects.
Overcoming Barriers: Data Privacy, Ethics, and Global Adoption
Despite the promise, several major hurdles stand in the way of universal adoption. At Lifebit, we focus heavily on these “tribulations” of precision medicine AI:
- Data Privacy and Sovereignty: How do we analyze 50 petabytes of data without moving it or risking a breach? The answer lies in federated learning meets precision medicine, where the AI goes to the data, not the other way around. This allows institutions to keep their data behind their own firewalls while still contributing to global research.
- Algorithmic Bias: If an AI is trained mostly on data from one ethnic group (historically, those of European descent), it may underdiagnose or provide inaccurate risk scores for others. This is a “garbage in, garbage out” problem. Diversifying datasets to include populations from the Global South and underrepresented minorities is a moral and clinical necessity to ensure health equity.
- Explainability (XAI): Doctors need to know why an AI made a recommendation. “Black box” models, where the logic is hidden, aren’t acceptable in a clinical setting where lives are at stake. We need “interpretable AI” that can point to the specific biomarkers or clinical notes that led to a high-risk score.
- Regulatory Hurdles: Frameworks like the EU AI Act and new FDA guidelines are beginning to define how AI should be validated. Clinical adoption requires rigorous, prospective trials to prove that AI-led care actually results in better patient outcomes compared to the current standard of care.
Major initiatives like the NIH PRIMED-AI program are working to address these gaps. PRIMED-AI funding opportunities are expected to launch in early 2026, focusing on integrating imaging with multimodal data to build trust and clinical adoption. Organizations like the AMA have also developed CME courses and advocacy principles to ensure AI is used safely, fairly, and transparently.
Future Directions: Multi-Omics, Digital Twins, and Real-Time Monitoring
The next frontier of precision medicine AI is moving toward a continuous, “360-degree” view of human health that evolves in real-time.
Digital Twins in Healthcare
One of the most transformative concepts is the “Medical Digital Twin.” This is a virtual representation of a patient, built from their genetic, clinical, and lifestyle data. Researchers can use AI to run simulations on this digital twin—testing how a new drug might affect the patient’s heart or how a specific diet might impact their glucose levels—before any real-world intervention occurs. This moves us from “precision medicine” to “predictive medicine.”
Organs-on-Chips and AI
Another exciting development is Organs-on-Chips. These are tiny microchips lined with living human cells that mimic the function of real organs. When combined with AI, they allow us to test how a specific patient’s cells will react to a new drug in a lab setting. Research on organs-on-chips suggests this could virtually eliminate the risk of toxic drug reactions and significantly speed up the drug discovery process by bypassing some animal testing phases.
Generative AI for Drug Discovery
Generative AI is also being used to design entirely new molecules that have never existed in nature. By training on the structures of millions of proteins, AI can “hallucinate” new drug candidates that are perfectly shaped to bind to a specific disease-causing protein. This could reduce the time it takes to find a drug lead from years to just weeks.
Real-Time Monitoring and Digital Biomarkers
We are also seeing a shift toward Real-Time Monitoring. Instead of waiting for a yearly check-up, AI-driven wearables and sensors can provide a constant stream of “digital biomarkers.” This allows for immediate adjustments to treatment—such as fine-tuning insulin doses for a diabetic patient based on real-time activity levels or detecting early signs of a heart arrhythmia during sleep. This continuous feedback loop ensures that the treatment is always optimized for the patient’s current state.
Frequently Asked Questions about Precision Medicine AI
How does AI improve patient outcomes in precision medicine?
AI improves outcomes by reducing diagnostic errors, identifying the most effective treatments faster, and predicting adverse reactions before they happen. In oncology, for instance, AI-driven machine learning precision medicine guide strategies have helped identify therapeutic opportunities that traditional testing missed, such as identifying rare mutations that make a patient eligible for a specific targeted therapy. By personalizing the dose and the drug, AI minimizes the “trial and error” period, which is often when the most significant side effects occur.
What are the main challenges to AI adoption in clinical settings?
The primary barriers are data silos, concerns over patient privacy, and the need for clinical validation. Many doctors are also concerned about the “explainability” of AI—they need to understand the logic behind a recommendation to trust it with a patient’s life. Furthermore, integrating AI into the existing hospital workflow (EHR systems) is a significant technical challenge. There is also the issue of liability: if an AI makes a recommendation that leads to a poor outcome, who is responsible? These legal and ethical questions are still being debated by regulatory bodies.
What is the role of electronic health records (EHRs) in personalized care?
EHRs provide the longitudinal “story” of a patient’s health. While genomics tells us what might happen, EHRs tell us what is happening. When AI integrates EHR data (like lab results, history of infections, and medication history) with genomic information, it can identify trends—like a slow change in kidney function over five years—that might indicate a developing chronic condition long before it becomes acute. This integration is the key to moving from reactive sick-care to proactive health management.
Is AI in precision medicine only for cancer patients?
No. While oncology has been the early adopter, precision medicine AI is expanding rapidly into cardiology (predicting heart failure), endocrinology (personalized diabetes management), and psychiatry (matching patients to the right antidepressant based on genetic metabolism). It is also becoming a cornerstone of rare disease diagnosis, helping to identify genetic causes for unexplained symptoms in children and adults alike.
Stop Guessing. Start Targeting: How Lifebit Powers the Future of Care.
The transition from “average” medicine to precision care is no longer a matter of if, but how fast. At Lifebit, we believe the answer lies in making the world’s most sensitive biomedical data safely accessible.
Our platform provides a federated trusted research environment that allows biopharma and public health agencies to run advanced AI/ML analytics on global, multi-omic data without ever moving the records. By focusing on data harmonization and secure collaboration, we are helping our partners deliver precision medicine at scale.
Whether you are looking to accelerate drug discovery or improve patient monitoring, the future of healthcare is personal.
Ready to see how federated AI can transform your research? Explore Lifebit’s Solutions for Precision Medicine

