The Vigilant Eye: Mastering Drug Safety Signal Detection
Stop Missing Rare Risks: How Drug Safety Signal Detection Saves Lives
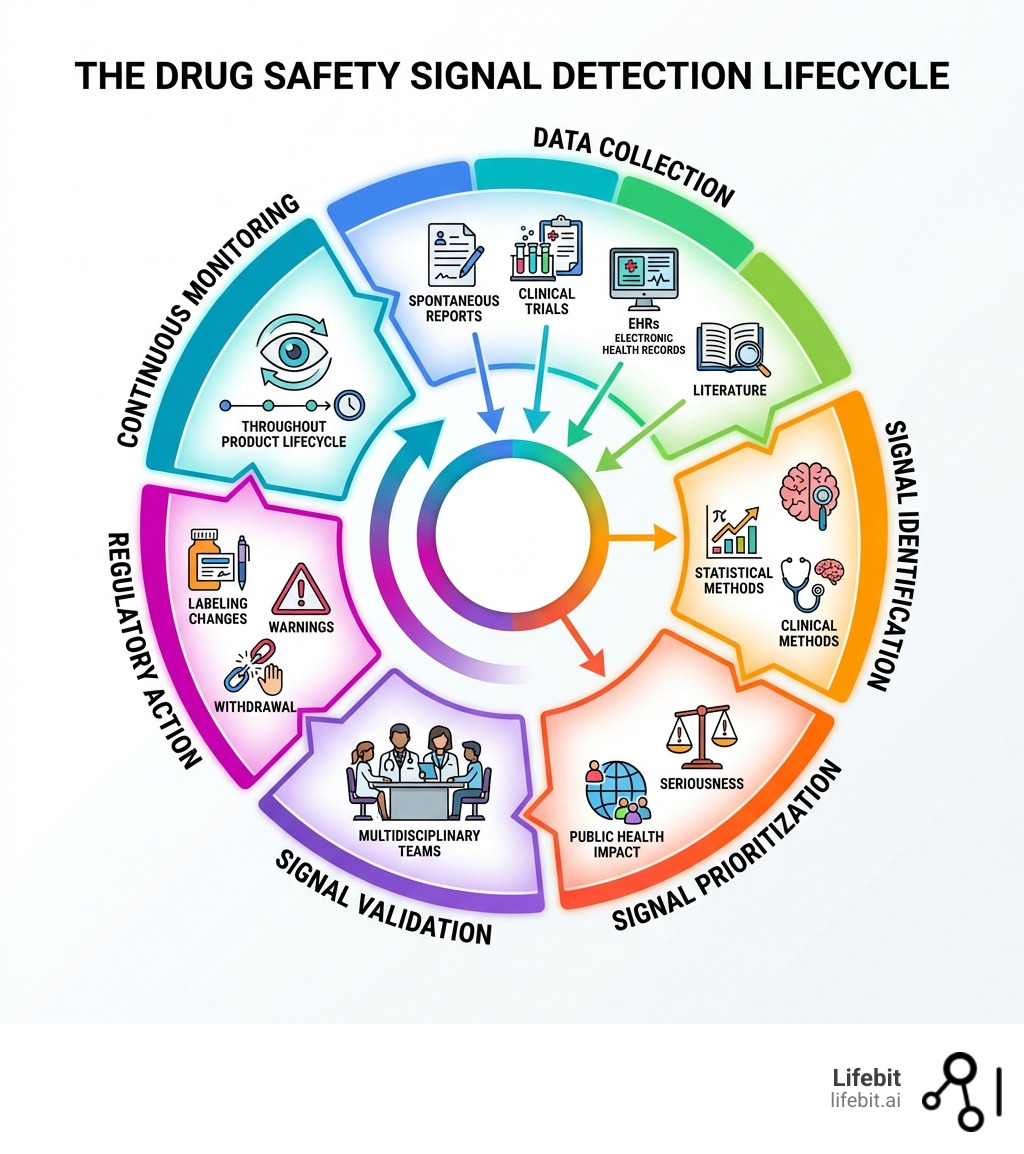
Drug safety signal detection is the systematic process of identifying potential new risks or changes in known risks associated with medicines after they reach the market. At its core, signal detection involves:
- Analyzing adverse event data from spontaneous reports, clinical trials, electronic health records, and scientific literature
- Identifying patterns that suggest a previously unknown or inadequately documented association between a drug and an adverse event
- Prioritizing signals based on seriousness, frequency, and public health impact
- Validating findings through multidisciplinary medical and statistical review
- Taking action through regulatory measures like labeling changes, safety warnings, or market withdrawal when necessary
Pre-approval clinical trials are limited in size and scope—they typically involve fewer than 10,000 patients and run for relatively short durations. This means rare adverse effects (occurring in less than 1 in 2,000 patients), long-term safety issues, and risks in specific populations like pregnant individuals or children often remain undetected until a drug is widely used in the real world. Historically, the need for robust signal detection was underscored by tragedies like the 1937 Elixir Sulfanilamide disaster, where the use of diethylene glycol as a solvent led to over 100 deaths and prompted the 1938 Federal Food, Drug, and Cosmetic Act. This was followed by the thalidomide crisis of the 1960s, which led to the establishment of modern pharmacovigilance and the requirement for proof of safety and efficacy.
More recently, the withdrawal of Vioxx (rofecoxib) in 2004 due to cardiovascular risks highlighted the necessity of continuous, real-time monitoring of drugs even after they have passed rigorous Phase III trials. Between 1998 and 2007, consumer reports to the FDA’s Adverse Event Reporting System (FAERS) increased from approximately 24,000 to 175,000 annually. Today, FAERS contains over 25 million reports, with more than 2 million added each year, demonstrating the staggering volume of post-market safety data that requires systematic monitoring. This growth has necessitated the adoption of the E2B(R3) standard for the electronic transmission of individual case safety reports (ICSRs), ensuring that data can be shared and analyzed across global regulatory jurisdictions with high precision.
The challenge today isn’t just detecting signals—it’s doing so in real-time across siloed, diverse datasets while maintaining regulatory compliance and data security. Traditional pharmacovigilance systems struggle with data silos, under-reporting, poor data quality, and the inability to rapidly integrate evidence from electronic health records, claims databases, and genomic data. Regulatory authorities like the EMA and FDA now expect sponsors to have robust, traceable signal detection systems that can identify emerging risks before they escalate into public health crises. The shift from reactive to proactive pharmacovigilance requires a fundamental change in how we handle data, moving away from manual case-by-case review toward automated, high-throughput screening of massive datasets.
I’m Dr. Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve built federated AI platforms that enable secure, in-situ drug safety signal detection across 275+ million patient records without moving sensitive data. My background in computational biology, bioinformatics, and AI-powered healthcare infrastructure has shown me that the future of pharmacovigilance lies in breaking down data barriers while maintaining the highest standards of compliance and governance.
Drug safety signal detection terminology:
What is a Safety Signal? The ‘Heads-Up’ That Prevents Public Health Crises

In pharmacovigilance (PV), a “signal” is essentially a hypothesis. The Council for International Organizations of Medical Sciences (CIOMS) defines a signal as information arising from one or multiple sources that suggests a new potentially causal association, or a new aspect of a known association, between an intervention and an event. This could be an adverse event or even a beneficial one, though we usually focus on the former to protect patients.
Think of a signal as a “heads-up” that something might be wrong. It isn’t a confirmed fact yet; rather, it is evidence judged to be of sufficient likelihood to justify further investigation. This investigation is what we call signal validation. During this phase, we move from a “potential risk” (an untoward occurrence with some basis for suspicion) to an “identified risk” (a confirmed association). The CIOMS VIII and IX working groups have further refined these definitions to include “important identified risks” and “important potential risks,” which are categorized based on their impact on the individual patient and the public health at large. An “important identified risk” is one that has a significant impact on the benefit-risk balance of the product and usually requires inclusion in the safety labeling.
The scientific research on signal detection methodology and application emphasizes that signals are not just about “new” side effects. They also include:
- Increases in frequency or severity of a known side effect.
- New interactions with other drugs, foods, or underlying medical conditions.
- Risk in specific populations (e.g., the elderly, children, or those with renal dysfunction) that were not adequately represented in clinical trials.
- Signals of lack of efficacy, which can be particularly critical for life-saving medications like antibiotics or oncology treatments.
- Designated Medical Events (DMEs): These are serious medical conditions that are often drug-induced, such as Torsade de Pointes, Acute Liver Failure, or Stevens-Johnson Syndrome. Because of their high specificity to drug toxicity, even a single well-documented case of a DME can constitute a valid signal.
Clinical vs. Statistical Signals
We generally categorize signals into two flavors: qualitative and quantitative.
- Clinical (Qualitative) Signals: These often come from individual case reports or small case series. A single, well-documented report—for example, a patient who experiences a rare reaction, stops the drug (dechallenge), and the reaction goes away, then restarts the drug (rechallenge) and the reaction returns—can be a powerful clinical signal. These require high-quality narratives and expert medical review. These are often referred to as “sentinel cases” because they provide the first warning of a potential problem. Clinical signals are particularly effective for identifying acute, idiosyncratic reactions where the temporal relationship is clear.
- Statistical (Quantitative) Signals: These are born from “data mining.” They emerge when we look at aggregate data and see numerical differences that shouldn’t be there. For instance, if a drug-event combination appears significantly more often in a database than would be expected by chance, we flag a disproportionality signal. These methods are essential for identifying signals that are too rare to be seen in individual cases but become apparent when looking at millions of patient-years of exposure. Statistical signals provide the “breadth” of monitoring, while clinical signals provide the “depth.”
4 Data Sources You Need for Real-Time Drug Safety Signal Detection
To catch every possible risk, we have to cast a wide net. Gone are the days when we relied solely on a doctor’s handwritten note sent by mail. Today, drug safety signal detection draws from a massive ecosystem of data, often referred to as Real-World Data (RWD).
- Spontaneous Reports: These are the backbone of PV. They are voluntary reports from healthcare professionals and patients. While they suffer from under-reporting, they are often the first source of information for rare and unexpected events. Systems like the FDA’s MedWatch and the MHRA’s Yellow Card scheme rely on these reports to build a global picture of drug safety.
- Clinical Trials: While limited in size, pre-approval and Phase IV trials provide controlled data that can reveal signals early. Post-marketing surveillance (Phase IV) is particularly useful as it observes the drug in a broader, less controlled population, often including patients with multiple comorbidities who were excluded from earlier trials.
- Scientific Literature: Researchers often publish case reports of “Suspected Unexpected Serious Adverse Reactions” (SUSARs) long before they hit regulatory databases. Systematic literature screening is a regulatory requirement for most marketing authorization holders, requiring the use of specialized databases like Embase and MEDLINE.
- Electronic Health Records (EHRs) and Claims Data: These offer “real-world evidence” (RWE). They allow us to see how millions of people actually use a drug in the wild, often revealing long-term effects that trials miss. Initiatives like the FDA’s Sentinel System use a distributed data model where queries are sent to data partners (like health insurers) who run the analysis locally, preserving patient privacy while monitoring safety across 100+ million lives. Similarly, the OHDSI (Observational Health Data Sciences and Informatics) collaborative uses the OMOP Common Data Model to standardize EHR data across different countries, enabling global-scale safety studies.
To handle these streams, many organizations are moving toward real-time pharmacovigilance to ensure no signal sits in a queue while a patient is at risk. Furthermore, emerging sources like social media monitoring and wearable device data are being explored. While social media data is often “noisy,” it can provide early warnings of patient-reported outcomes (PROs) that haven’t yet reached a clinical setting. Wearables offer continuous physiological monitoring, which could potentially detect subtle signals like heart rate variability or sleep disturbances associated with a new medication long before they manifest as clinical symptoms.
The Role of Spontaneous Reports and Consumer Data
Spontaneous reporting systems (SRS) are vital. Interestingly, the role of the “consumer” (the patient) has exploded. This shift is partly due to increased health literacy and the ease of digital reporting.
- The Netherlands (Lareb): Direct patient reports increased to approximately 20% of their total volume between 2004 and 2007, showing that patients are often more motivated than busy clinicians to report side effects.
- The US FDA: Consumer reports in the AERS database jumped from 22% (24,000 cases) in 1998 to a staggering 46% (175,000 cases) by 2007.
- Digital Change: By 2009, over 84% of expedited reports to the FDA were submitted electronically, allowing for faster processing and automated screening.
While patient reports are sometimes criticized for lacking technical medical detail, they often provide richer information on “quality of life” impacts and can be earlier indicators of a problem than formal clinical channels. They capture the patient’s lived experience, which is increasingly recognized as a critical component of benefit-risk assessment.
Beyond Manual Review: Advanced Methods for Drug Safety Signal Detection
When we have millions of records, we can’t just “look” at them. We use disproportionality analysis (DA) to find the “needle in the haystack.” These methods compare the observed number of cases for a drug-event pair against what we would expect if there were no association. This is typically done using a 2×2 contingency table to calculate various scores.
| Method | Full Name | Primary Use Case |
|---|---|---|
| PRR | Proportional Reporting Ratio | Frequentist approach; easy to calculate; used for initial screening. It compares the proportion of a specific AE for a drug to the proportion of that AE for all other drugs in the database. |
| ROR | Reporting Odds Ratio | Similar to PRR but uses odds ratios; handles smaller datasets better and allows for adjustment of confounding factors through logistic regression. |
| IC | Information Component | Bayesian approach used by the WHO; uses “shrinkage” to reduce false positives in rare events by pulling the estimate toward the null. |
| EBGM | Empirical Bayes Geometric Mean | Used by the FDA; very stable even with low case counts. It uses the Multi-item Gamma Poisson Shrinker (MGPS) algorithm to identify higher-than-expected reporting rates. |
Bayesian shrinkage is a lifesaver here. It essentially “pulls” the scores of rare events toward the null until there is enough data to prove a real association, preventing us from chasing every single statistical fluke. This is crucial because, in a database with thousands of drugs and tens of thousands of possible adverse events, random chance will inevitably produce some high disproportionality scores. The MGPS algorithm, for instance, calculates the Empirical Bayes Geometric Mean (EBGM). If the lower 5th percentile of the distribution (EB05) is greater than 2, it is typically considered a signal of interest that warrants further medical review. Organizations often leverage AI-driven pharmacovigilance solutions to automate these complex calculations across global datasets.
Machine Learning and AI in Drug Safety Signal Detection
Traditional statistical methods are great, but they struggle with “unstructured” data—like the free-text narratives in a doctor’s report or a patient’s social media post. This is where drug safety AI changes the game.
- Natural Language Processing (NLP): NLP can read through thousands of medical journals or case narratives to extract symptoms, dosages, and timelines that a human might miss. Advanced models like Transformers (e.g., BERT, BioBERT, or specialized LLMs) can understand the context of medical language, distinguishing between “the patient did not have a headache” and “the patient complained of a headache.” These models perform Named Entity Recognition (NER) to map unstructured text to standardized MedDRA codes with high precision.
- Pattern Recognition and Deep Learning: AI can find “latent correlations”—complex relationships between multiple drugs (polypharmacy), underlying conditions, and environmental factors that traditional 2×2 contingency tables can’t see. For example, an AI might detect that a specific combination of a blood pressure medication and a common herbal supplement increases the risk of kidney injury, a signal that would be invisible when looking at either product in isolation.
- Automated Screening and Triage: AI helps in “triage,” quickly filtering out “noise” (like duplicate reports, which are a major headache in PV, or poorly documented cases) so human experts can focus on high-priority threats. This significantly reduces the “signal-to-noise” ratio, allowing safety teams to be more efficient and responsive to emerging threats.
From Detection to Action: The 5-Step Signal Management Lifecycle
Detecting a signal is only the beginning. The Official EMA signal management guidelines outline a strict lifecycle that every Marketing Authorization Holder (MAH) must follow to ensure patient safety and regulatory compliance.
- Detection: Identifying the signal through data mining, literature review, or individual case review. This is the “hypothesis generation” phase where potential associations are first flagged.
- Validation: Checking the quality of the data. Is it a real medical event? Is the timing right (temporal relationship)? Are there enough cases to suggest a pattern? Validation aims to filter out signals that are clearly due to chance, confounding factors, or duplicate reporting. A validated signal is one where the data is sufficient to justify a full medical assessment.
- Prioritization: Is this a “Designated Medical Event” (DME) like liver failure, Stevens-Johnson syndrome, or anaphylaxis? If so, it goes to the top of the pile. Prioritization considers the severity of the event, the vulnerability of the population affected (e.g., children or the elderly), and the potential public health impact if the risk is not addressed immediately.
- Assessment/Medical Review: This is the “art” of PV. Physicians review the full clinical context, looking for “confounders” (like the patient’s existing illness or concomitant medications) that might actually be the cause. They use established causality assessment tools like the Naranjo Scale, which uses a 10-question system to determine the probability of a drug causing an adverse event. Questions include: Did the reaction appear after the drug was administered? Did the reaction improve when the drug was discontinued (dechallenge)? Did the reaction reappear when the drug was readministered (rechallenge)? A score of 9 or higher indicates a “definite” causal relationship. Alternatively, the WHO-UMC system categorizes causality into levels such as Certain, Probable, Possible, or Unlikely based on clinical and pharmacological evidence.
- Recommendation for Action: If the signal is confirmed as a “validated signal” and then an “identified risk,” what do we do? This involves a benefit-risk reassessment to determine if the drug’s benefits still outweigh its newly discovered risks.
Regulatory Actions and Risk Mitigation
Once a signal is validated as a “genuine risk,” regulatory bodies like the EMA, MHRA, and FDA have several tools in their shed to protect the public:
- Labeling Changes: Adding a new side effect to the “Adverse Reactions” section or a “Boxed Warning” (the most serious type of warning) to the prescribing information.
- Safety Communications: Issuing a “Dear Healthcare Professional” (DHPC) letter to warn doctors directly about the new risk and provide guidance on how to monitor patients.
- Restricted Use: Changing the drug’s status, such as moving it from over-the-counter to prescription-only, or restricting its use to certain second-line treatments.
- Risk Evaluation and Mitigation Strategies (REMS): Requiring doctors to have special training, pharmacies to be certified, or patients to undergo regular laboratory testing (e.g., liver function tests) to continue receiving the medication.
- Market Withdrawal: In rare cases, if the risks outweigh the benefits (like the historical cases of thalidomide or more recently, certain weight-loss medications), the drug is removed from the market entirely. This is always a last resort but remains a critical tool for public safety.
Break Down Data Silos: Solving the Biggest Problems in Signal Detection
The path to perfect safety isn’t easy. We face several systemic problems that require technological and collaborative solutions:
- Data Silos: Safety data is often trapped in different countries, hospitals, or departments due to strict privacy regulations. Lifebit’s federated AI platform solves this by allowing analysis to happen where the data lives. Instead of moving sensitive patient records to a central server (which creates security risks and regulatory hurdles), the algorithm travels to the data. This ensures compliance with local laws like GDPR in Europe and HIPAA in the US while still allowing for global-scale analysis.
- Under-reporting and the ‘Herzog Effect’: It’s estimated that only a small fraction (often less than 10%) of adverse events are ever reported. This creates a “passive” system that relies on people being motivated to speak up. Furthermore, the “Weber Effect” suggests that reporting for a new drug peaks in the first two years after launch and then declines, regardless of the actual occurrence of side effects. This can lead to a false sense of security as a drug matures on the market.
- The Masking Effect and Competition Bias: If one drug is reported very frequently for a specific side effect (e.g., a well-known blockbuster drug), it can “hide” or mask the signals of other, newer drugs for that same effect in the database. Statistical techniques like “stripping” or “stratification” are needed to uncover these hidden signals.
- Protopathic Bias and Indication Bias: Protopathic bias occurs when a drug is prescribed for an early symptom of a disease that has not yet been diagnosed, leading to a false causal link between the drug and the disease. Indication bias occurs when the underlying condition being treated is itself the cause of the adverse event, rather than the medication. Advanced signal detection strategies must use sophisticated temporal analysis to account for these biases.
- Data Quality and Standardization: Reports often come in with missing information, vague descriptions, or non-standard terminology. The adoption of the MedDRA (Medical Dictionary for Regulatory Activities) terminology has helped, but mapping diverse data sources (like EHRs and claims) to a common data model like OMOP is still a significant challenge.
- Global Compliance: Keeping up with the EMA’s GVP Module IX, FDA mandates, and MHRA post-Brexit rules is a full-time job. Using pharmacovigilance compliance solutions is no longer optional—it’s a necessity for any global pharmaceutical company to avoid heavy fines and, more importantly, to protect patients.
Drug Safety Signal Detection: Your Top Questions Answered
What are the primary differences between clinical and statistical signals?
Clinical signals are “bottom-up”—they start with a deep dive into an individual patient’s story. They rely on clinical judgment, dechallenge/rechallenge data, and biological plausibility (does it make sense that this drug would cause this reaction?). Statistical signals are “top-down”—they look at thousands of patients to find a mathematical “blip.” While statistical signals can find rare events in large populations that a single doctor might never notice, clinical signals are better at identifying highly specific, acute reactions where the causal link is obvious in a single patient.
How do regulatory bodies like the EMA and MHRA manage signals?
They follow the GVP (Good Pharmacovigilance Practices) Module IX. The EMA’s Pharmacovigilance Risk Assessment Committee (PRAC) reviews signals and makes recommendations that apply across Europe. The MHRA uses its Yellow Card scheme and specialized signal detection teams to monitor the UK population. Both prioritize transparency, often publishing lists of signals currently under investigation to keep the public and healthcare community informed. They also collaborate through the International Council for Harmonisation (ICH) to standardize safety reporting worldwide.
What are the key considerations for drug safety signal detection strategies?
A modern strategy must be multidisciplinary (combining doctors, data scientists, and statisticians) and proactive. It should use diverse data sources (not just spontaneous reports) and implement real-time monitoring. Finally, every decision must be traceable; if an auditor asks why you dismissed a signal three years ago, you need a documented audit trail to prove your reasoning. This includes documenting the data sources used, the statistical thresholds applied, and the medical rationale for the final decision.
How is AI changing the future of signal detection?
AI is moving us toward “predictive pharmacovigilance.” Beyond just detecting events that have already happened, researchers are using AI to predict which patients are at the highest risk of a side effect based on their genetic profile or medical history. This allows for personalized medicine where a drug might be avoided in a specific patient before a reaction ever occurs. Additionally, AI is being used to monitor “digital twins”—virtual models of patients—to simulate drug effects and identify potential safety issues before a drug even enters human trials.
Secure Your Pharmacovigilance Strategy with Federated AI
The “Vigilant Eye” of drug safety signal detection is what keeps modern medicine safe. As we move into an era of personalized medicine and complex biologics, the volume and complexity of data will only grow.
At Lifebit, we believe that the answer lies in technology that respects data privacy while enabling global collaboration. Our pharmacovigilance platform uses federated AI to connect the world’s most sensitive biomedical and multi-omic data. By bringing the analysis to the data, we help biopharma and regulators detect signals faster, more accurately, and more securely than ever before. Patient safety shouldn’t have to wait for data to travel across borders—with Lifebit, the insights are already there.

