Your Health, Tailored: How Machine Learning is Revolutionizing Personalized Care

Why Machine Learning Precision Medicine Matters Now
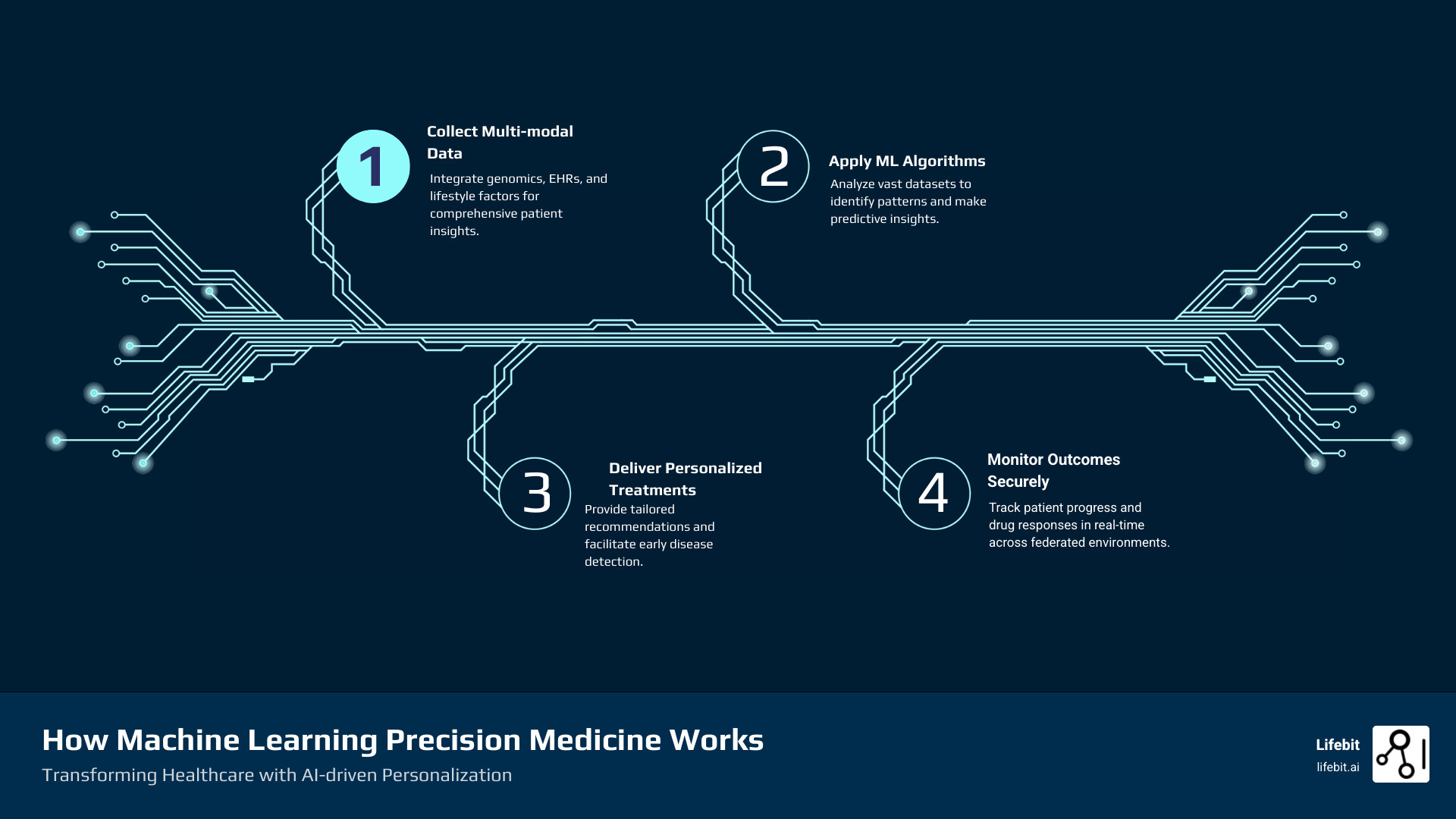
Machine learning precision medicine is changing healthcare by using algorithms to analyze patient data—including genomics, medical records, and lifestyle factors—to deliver personalized treatment plans that work better than one-size-fits-all approaches. Here’s how it’s being applied today:
- Predicting drug response based on genetic profiles to avoid adverse reactions
- Detecting diseases earlier by analyzing patterns across millions of patient records
- Reducing diagnostic errors through AI-powered clinical decision support
- Matching patients to clinical trials using genomic and phenotypic data
- Enabling real-time pharmacovigilance across federated health datasets
Traditional medicine treats everyone with the same condition identically. Machine learning precision medicine recognizes that your genes, environment, and biology make you unique—and your treatment should reflect that.
The challenge? Most healthcare organizations struggle with siloed data, slow onboarding, and regulatory barriers that prevent them from using AI at scale. Genomic data sits in one system. Electronic health records in another. Claims data somewhere else. Without the right infrastructure, even the most advanced algorithms can’t deliver insights.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over 15 years building federated platforms that bring machine learning precision medicine to life across secure, compliant environments—from genomic analysis to real-world evidence generation. Before founding Lifebit, I contributed to Nextflow and led research at the Centre for Genomic Regulation, creating tools that power precision medicine worldwide.
Basic machine learning precision medicine glossary:
The Power of Machine Learning Precision Medicine in Modern Healthcare
The shift from reactive “sick care” to proactive precision medicine is fueled by our ability to process information that is simply too vast for the human brain to handle alone. When we talk about machine learning precision medicine, we are talking about the ultimate clinical assistant—one that has read every medical paper, seen every patient record, and can spot a needle-thin pattern in a haystack of genomic sequences.
Research published in the BMJ on AI implementation in precision medicine highlights how these systems provide vital clinical decision-support. By processing real-time data, machine learning (ML) helps doctors make informed decisions faster, reducing the cognitive load that often leads to burnout and oversight. This is particularly critical in high-stakes environments like Intensive Care Units (ICUs) or oncology wards, where the volume of data generated per patient can exceed 50,000 data points per day.
In modern medicine, AI takes several forms to support this mission:
- Natural Language Processing (NLP): Beyond just reading notes, modern NLP models like clinical-BERT are used to extract “hidden” phenotypes from unstructured text. For example, an NLP algorithm can scan years of physician narratives to identify subtle mentions of fatigue or minor neurological shifts that, when aggregated, signal the onset of an autoimmune disorder long before a formal diagnosis is made.
- Computer Vision: This is the gold standard for analyzing medical imagery like MRIs, CT scans, or X-rays. Deep learning models, specifically Convolutional Neural Networks (CNNs), can now detect malignant lesions in mammograms with a sensitivity that rivals or exceeds senior radiologists, often identifying micro-calcifications that are nearly invisible to the naked eye.
- Predictive Analytics: This involves using historical data to forecast future events. In precision medicine, this is used for “Sepsis Prediction” models that monitor heart rate, blood pressure, and temperature in real-time to alert staff of a potential septic shock up to 12 hours before clinical symptoms manifest.
How Machine Learning Precision Medicine Reduces Diagnostic Errors
Diagnostic errors are a silent crisis in healthcare, contributing to an estimated 10% of patient deaths. These errors often stem from incomplete data or “information overload.” Machine learning precision medicine acts as a safety net. By automating Electronic Health Record (EHR) extraction, ML can flag discrepancies between a patient’s reported symptoms and their historical lab results.
For instance, if a patient is prescribed a standard dosage of a blood thinner, an ML system can cross-reference the patient’s genetic data (specifically the CYP2C9 and VKORC1 genes) to alert the physician if the patient is a “slow metabolizer,” which would otherwise lead to a dangerous internal bleed. Instead of replacing doctors, we use “augmented intelligence” to handle these complex cross-referencing tasks. This allows clinicians to focus on the patient sitting in front of them, while the AI ensures no critical genetic marker or lifestyle risk factor is missed.
Types of Algorithms Driving Machine Learning Precision Medicine
To understand how we get these insights, we have to look under the hood at the algorithms:
- Supervised Learning: We train these models on massive, labeled datasets (e.g., “this set of 100,000 images shows a stage-2 tumor”). This is the backbone of diagnostic tools and is currently the most common application in clinical settings.
- Unsupervised Learning: These algorithms look for hidden patterns in data without pre-existing labels. This is incredible for “disease subtyping.” For example, unsupervised learning has recently been used to discover that what we previously called “Type 2 Diabetes” may actually be five distinct physiological conditions, each requiring a different therapeutic approach.
- Deep Learning and Neural Networks: Inspired by the human brain’s architecture, these multi-layered models are used for the most complex tasks, such as predicting how a specific protein will fold or interpreting whole-genome sequences (WGS) to identify rare variants that cause pediatric developmental delays.
- Reinforcement Learning (RL): A newer frontier in precision medicine, RL is being used to optimize dynamic treatment regimes. For example, in chronic conditions like HIV or cancer, RL can help determine the optimal timing and dosage of drugs to minimize toxicity while preventing the virus or tumor from developing resistance.
Integrating Multi-modal Data for 360-Degree Patient Insights
The true magic of machine learning precision medicine happens when we stop looking at data in silos. Your health isn’t just your DNA; it’s a combination of your genetics, your environment, and your lifestyle. This is often referred to as the “biopsychosocial” model of health, and ML is the only tool capable of integrating these disparate data streams.
Building the foundation for genomics requires us to integrate multi-modal data. This means combining:
- Genomics: Your “blueprint” or DNA sequence.
- Transcriptomics & Proteomics: Measuring gene expression and protein levels to see what is actually happening in the body at a specific moment.
- Phenotype derivation: The observable characteristics (like height, blood pressure, or gait).
- Real-world data (RWD): Data from insurance claims, pharmacy records, and even wearable devices like Oura rings or Apple Watches that track sleep and activity levels.
The Challenge of Data Harmonization
One of the biggest hurdles in machine learning precision medicine is that data from different hospitals often “speaks different languages.” One hospital might record a lab result in mg/dL, while another uses mmol/L. To solve this, researchers use Common Data Models (CDMs) like OMOP (Observational Medical Outcomes Partnership). By mapping all data to a standardized format, ML models can be trained on global datasets, significantly increasing their accuracy and generalizability.
Structured vs. Unstructured Data: The Great Divide
One of the biggest problems in precision medicine is that 80% of medical data is “unstructured” (think handwritten notes, voice recordings, or PDF pathology reports). Here is how we compare them:
| Data Type | Examples | Ease of AI Use | Technical Requirement |
|---|---|---|---|
| Structured | Lab results, ICD-10 codes, age, weight | High (Ready for analysis) | SQL Databases, ETL pipelines |
| Unstructured | Clinical narratives, pathology reports, imaging | Low (Requires NLP/Vision) | Large Language Models (LLMs), OCR |
| Multi-omic | DNA, RNA, Metabolites | Medium (High complexity) | High-Performance Computing (HPC) |
By harmonizing these sources, we create a 360-degree view of the patient. This allows for more accurate “phenotyping”—the process of identifying specific disease traits that dictate how a patient will respond to a specific drug. For example, in asthma care, ML can distinguish between “eosinophilic” asthma and other types, ensuring the patient receives targeted biologics rather than just general steroids.
Real-World Applications: From Oncology to Rare Disease Diagnosis
We are no longer in the “experimental” phase of machine learning precision medicine. It is happening in clinics across the UK, USA, Europe, and beyond, fundamentally altering the trajectory of patient care.
Oncology and Radiogenomics
In cancer care, we use radiogenomics to link the features seen in medical images (like the shape or texture of a tumor) with the underlying gene expression. This helps oncologists predict how aggressive a cancer might be without needing as many invasive biopsies. For instance, deep learning for breast cancer prediction has shown remarkable success in identifying high-risk patients years before traditional symptoms appear. Furthermore, ML models can now predict “Immunotherapy Response,” helping doctors decide which patients will benefit from expensive checkpoint inhibitors and which should seek alternative therapies.
Rare Disease and the “Diagnostic Odyssey”
For the millions living with rare diseases, the “diagnostic odyssey” can last a decade and involve dozens of misdiagnoses. AI-driven interpretation of genomic variants can cut this time to days. By comparing a patient’s genome against a global database of known pathogenic variants, ML can highlight the single mutation responsible for a child’s condition. Programs like “Project Baby Bear” in California have demonstrated that rapid Whole Genome Sequencing (rWGS) in neonatal ICUs can change clinical management in over 30% of cases, saving lives and reducing hospital costs.
Cardiovascular Health and Polygenic Risk Scores (PRS)
Cardiovascular disease remains the leading cause of death globally. Machine learning precision medicine is introducing Polygenic Risk Scores, which aggregate the effects of millions of small genetic variations to predict a person’s lifetime risk of heart attack. Unlike traditional cholesterol tests, which only show a snapshot in time, a PRS can identify high-risk individuals in their 20s, allowing for decades of preventive lifestyle interventions.
Pharmacogenomics in Psychiatry
Psychiatry has traditionally been a field of “trial and error,” particularly with antidepressants. However, ML models are now being used to analyze a patient’s CYP450 genotype to predict which medications will be effective and which will cause severe side effects like weight gain or insomnia. This precision approach reduces the time it takes for a patient to find a therapeutic dose, significantly lowering the risk of treatment-resistant depression and suicide.
Metabolic Health and GLP-1s
With the rise of metabolic health awareness, ML is being used to track how patients respond to GLP-1 medications (like Ozempic or Wegovy). By analyzing real-time glucose data from Continuous Glucose Monitors (CGMs) and metabolic markers, we can tailor dosages to maximize weight loss and blood sugar control while minimizing side effects. This is the epitome of personalized metabolic care.
Overcoming the Critical Barriers to AI Adoption in Medicine
If machine learning precision medicine is so powerful, why isn’t it everywhere? The answer lies in a complex web of trust, security, and technical debt within the healthcare system.
Data Security and Privacy: The Federated Solution
Patient data is the most sensitive information on earth. In the past, researchers had to move data to their own servers to analyze it, which created massive security risks and violated data sovereignty laws. At Lifebit, we solve this through a federated AI platform. Instead of moving the data, we move the analysis to where the data lives.
This “Federated Learning” approach allows a model to learn from data at a hospital in London, a clinic in New York, and a biobank in Tokyo simultaneously, without the raw data ever leaving its original firewall. This ensures strict compliance with GDPR, HIPAA, and other regional regulations while still allowing for the massive sample sizes required for high-accuracy ML.
Fairness, Bias, and the Diversity Gap
Algorithms are only as good as the data they are trained on. Historically, genomic datasets have been overwhelmingly skewed toward populations of European descent. If a model is trained only on this data, its predictions for individuals of African, Asian, or Hispanic descent may be inaccurate or even harmful. Research on fairness in precision medicine highlights the urgent need for diverse datasets. To combat this, we must actively recruit diverse cohorts and use “Algorithmic Auditing” to check for bias before a model is deployed in a clinical setting. Precision medicine must be equitable, or it isn’t truly precise.
The “Black Box” Problem and Explainable AI (XAI)
For a doctor to trust an AI’s recommendation, they need to know why the AI made that choice. A “black box” model that says “this patient has a 90% risk of heart failure” without explanation is difficult to act upon. We are now seeing a shift toward Explainable AI (XAI), using techniques like SHAP (SHapley Additive exPlanations) or LIME. These tools highlight exactly which features—such as a specific lab value or a genetic marker—contributed most to the AI’s prediction, allowing the clinician to verify the logic against their own medical expertise.
Model Performance and Fraud Mitigation
Beyond clinical care, ML is being deployed for healthcare infrastructure. This includes fraud mitigation, where algorithms detect patterns of “upcoding” or phantom billing. By ensuring that healthcare resources are spent on actual patient care rather than administrative errors or bad actors, ML helps sustain the financial viability of precision medicine programs.
The Future of Augmented Intelligence and Preventive Care
The future of machine learning precision medicine is a move from “reactive” to “preventive.” We are entering an era of “P4 Medicine”: Predictive, Preventive, Personalized, and Participatory.
Digital Twins and Virtual Clinical Trials
One of the most exciting frontiers is the concept of the “Digital Twin.” This is a virtual representation of an individual patient, built from their multi-omic data and real-time sensor inputs. Researchers can use these digital twins to simulate how a patient might respond to a new drug before it is ever administered. This could revolutionize clinical trials, allowing for “In Silico” testing that identifies potential toxicities or failures much earlier in the drug development pipeline, potentially saving billions of dollars and years of research.
The Role of Generative AI in Drug Discovery
Generative AI is not just for chatbots; it is being used to design entirely new molecules. By training on the chemical structures of millions of known compounds, generative models can “dream up” new drug candidates that are optimized to bind to specific disease targets while minimizing off-target effects. This is accelerating the timeline for bringing new precision therapies to market for diseases that were previously considered “undruggable.”
Synergy Between Wearables and Clinical Care
Imagine a world where your wearable device detects a slight change in your heart rate variability and sleep patterns, syncs this with your genomic predisposition for cardiovascular issues, and alerts your doctor to adjust your medication before you ever feel a symptom. This synergy between noninvasive monitoring and advanced analytics will drastically reduce the global health burden.
By catching chronic diseases in their infancy—or preventing them altogether—we can reduce hospitalizations and lower the astronomical costs of late-stage treatments. Augmented intelligence will become the standard. Doctors won’t be replaced by machines; rather, doctors who use AI will replace doctors who don’t. This partnership is what will finally make healthcare truly individualized, scalable, and affordable for everyone.
Frequently Asked Questions about Machine Learning in Healthcare
What is the difference between AI and augmented intelligence in medicine?
While Artificial Intelligence often implies a machine acting independently, augmented intelligence focuses on the collaborative role of AI. It is designed to improve human intelligence and clinical expertise rather than replace it, providing tools that help doctors make better-informed decisions.
How does machine learning improve drug response predictions?
By analyzing a patient’s genetic makeup (pharmacogenomics) alongside their medical history, ML models can predict how a patient’s body will metabolize a specific drug. This helps avoid “trial and error” prescribing, reducing the risk of dangerous side effects and ensuring the patient gets the right dose the first time.
Can machine learning help reduce healthcare costs?
Yes, significantly. By improving diagnostic accuracy, predicting which patients are at risk of expensive emergency room visits, and streamlining drug findy, machine learning precision medicine can save billions in wasted treatments and administrative overhead.
Conclusion
The era of “one-size-fits-all” medicine is over. Through machine learning precision medicine, we are finally gaining the tools to treat patients as individuals. But to reach this future, we need a foundation built on security, diversity, and collaboration.
At Lifebit, we are proud to provide that foundation. Our federated AI platform enables secure, real-time access to global biomedical and multi-omic data without compromising patient privacy. Whether it’s through our Trusted Research Environment (TRE), our Trusted Data Lakehouse (TDL), or our R.E.A.L. (Real-time Evidence & Analytics Layer), we are helping biopharma and governments turn data into life-saving insights.
Lifebit platform capabilities include:
- Federated Governance: Securely analyze data across different locations and jurisdictions.
- Data Harmonization: Bringing together disparate datasets for unified analysis.
- Advanced AI/ML Analytics: Deploying state-of-the-art models directly where the data resides.
- AI-driven Safety Surveillance: Real-time pharmacovigilance to protect patients globally.
The future of health is custom, and it starts with the data.
Stop Siloing Data. Start Saving Lives.
Secure your research with Lifebit’s federated platform today.

