Why Safety Signal Detection is the Secret Sauce of Drug Safety

Why Safety Signal Detection is the Secret Weapon for Drug Safety
Safety signal detection is the process of identifying patterns in adverse event data that suggest a new or changing risk associated with a medicinal product. Historically, the field of pharmacovigilance was born out of necessity following the Thalidomide tragedy of the 1960s, which highlighted the catastrophic consequences of inadequate post-market monitoring. Today, it has evolved into a sophisticated, data-driven discipline that serves as the primary defense against unforeseen drug toxicities.
What is a safety signal?
- Information suggesting a new potentially causal association between a medicine and an adverse event, or a new aspect of a known association.
- Can come from multiple streams: spontaneous reports, clinical studies, scientific literature, or real-world data (RWD).
- Requires a structured validation process to confirm whether the risk is truly drug-related or merely a coincidental occurrence.
Why it matters:
- Detects rare events: Clinical trials are often powered to detect common side effects but are statistically blind to events occurring in 1 in 10,000 patients. Safety signal detection bridges this gap.
- Real-world populations: Trials exclude the elderly, pregnant women, and those with multiple comorbidities. Signal detection monitors the drug as it is used in the “wild.”
- Early Warning Systems: By identifying risks months or years before they become widespread, regulators can implement label changes or restricted distribution programs to save lives.
Key methods:
- Statistical screening: Utilizing disproportionality measures like PRR, ROR, IC, and BCPNN to find “statistical outliers” in massive databases.
- Clinical review: Expert analysis of case narratives to determine biological plausibility and temporal relationships.
- Aggregate analysis: Using MedDRA queries and frequency tables to look for trends across entire drug classes.
Safety signals aren’t just about catching problems—they’re about catching them early. Traditional manual review can’t keep pace with the explosion of post-market data from spontaneous reporting systems, electronic health records, and scientific literature. Modern pharmacovigilance demands a blend of statistical rigor, clinical judgment, and increasingly, AI-powered automation to scan massive datasets in real time.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over 15 years building platforms that transform how organizations access and analyze biomedical data—including safety signal detection across federated, real-world datasets. My background in computational biology, AI, and genomics has shown me how the right data infrastructure can accelerate life-saving insights while maintaining compliance and security.
Learn more about Safety signal detection:
Uncover Hidden Dangers: What Safety Signals Really Mean
In pharmacovigilance, a “signal” is like a smoke detector going off in a massive skyscraper. It doesn’t always mean there’s a fire, but it definitely means someone needs to go check the room. According to the Council for International Organizations of Medical Sciences (CIOMS) and the European Medicines Agency (EMA), a safety signal is information on a new or known adverse event that is potentially caused by a medicine and warrants further investigation.
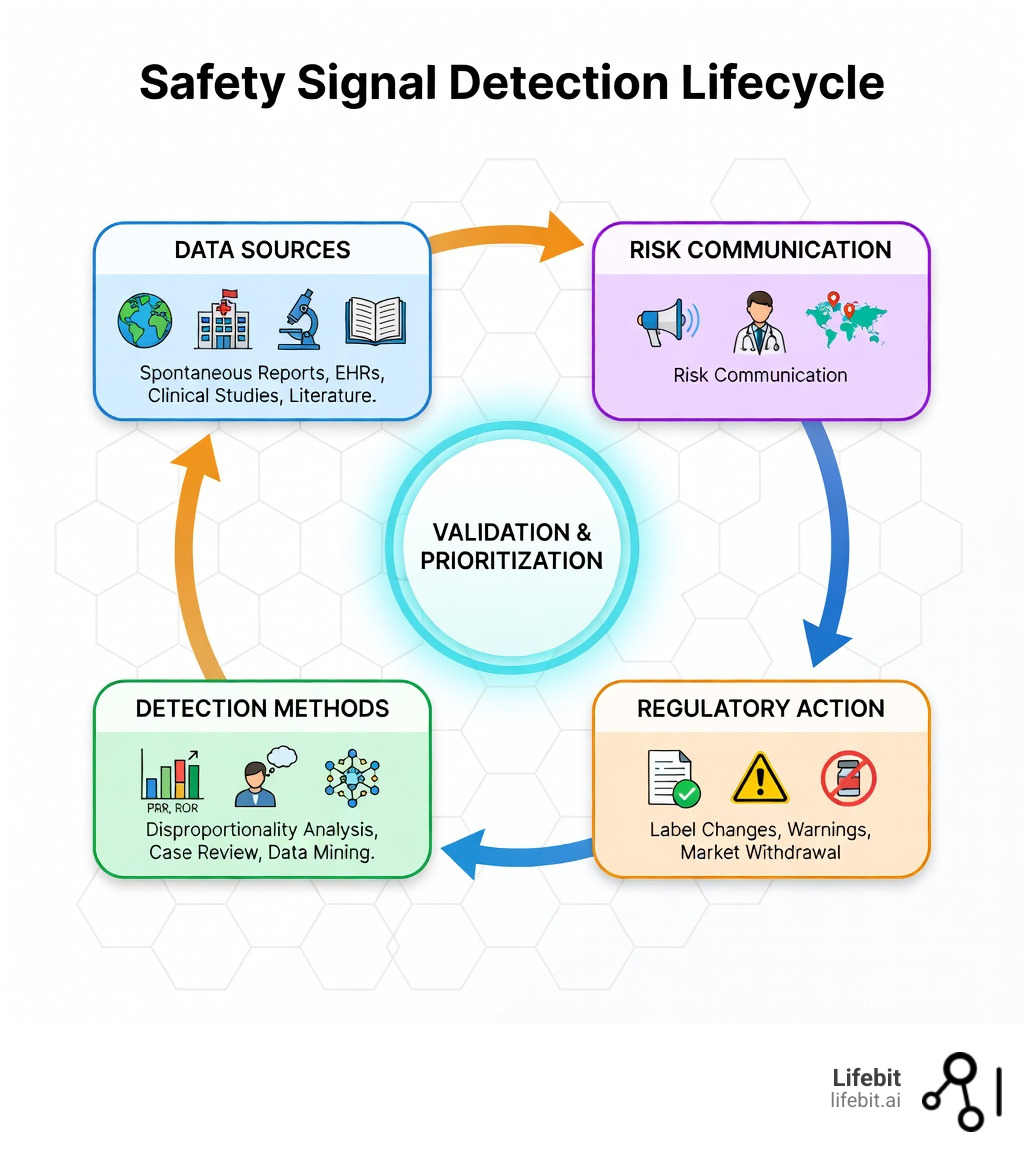
This definition is critical because it distinguishes between a simple “adverse event” (any medical occurrence) and a “signal” (an event with a suspected causal link). The signal management process is the backbone of the “Signal Management Cycle,” which includes detection, validation, prioritization, assessment, and recommendation for action. This cycle ensures that every potential risk is vetted through a standardized, transparent framework.
The Limitations of Clinical Trials
Think of it as the “missing link” between a drug and an unexpected side effect. While clinical trials are the gold standard for testing, they have inherent limitations known as the “Five Too’s”:
- Too few: Trials rarely exceed 3,000–5,000 patients.
- Too simple: Patients with multiple diseases or other medications are often excluded.
- Too median-aged: Children and the elderly are frequently underrepresented.
- Too narrow: Trials focus on specific indications, missing off-label use risks.
- Too brief: Long-term effects (like secondary cancers or organ failure) may take years to manifest.
For example, clinical trials are often impractical for detecting events rarer than 1 in 3,000 patients. This is why Safety signal detection is essential for post-market surveillance.
Primary Data Sources for Detection
We look for signals across several primary sources, each with its own strengths and weaknesses:
- Spontaneous Reports: These are the backbone of pharmacovigilance, coming from systems like the FDA’s FAERS, the UK’s Yellow Card scheme, or the Canada Vigilance Adverse Reaction Online Database. While they are prone to “under-reporting” and “reporting bias,” they are the fastest way to detect rare, idiosyncratic reactions.
- Clinical Studies: Even after a drug is approved, Phase IV trials and observational studies continue to provide high-quality, controlled data.
- Scientific Literature: Researchers worldwide publish case reports that might be the first hint of a problem. Literature screening is a regulatory requirement in most jurisdictions, as it often captures unique clinical insights not found in databases.
- Real-World Data (RWD): Electronic health records (EHRs) and insurance claims help us see how drugs perform in the “wild.” Unlike spontaneous reports, RWD provides a “denominator” (the total number of patients taking the drug), allowing us to calculate actual incidence rates.
The goal is to determine if there is a causal association. Is the drug actually causing the problem, or is it just a coincidence? To find out, we use a structured signal management process: detection, validation, prioritization, assessment, and action.
Get the Numbers Right: Proven Methods for Safety Signal Detection
When we have millions of reports, we can’t read them all one by one. That’s where math—specifically disproportionality analysis—comes in. We use statistical tools to see if a specific drug-event combination is popping up more often than we’d expect compared to all other drugs in the database. This is often visualized using a 2×2 contingency table, comparing the presence or absence of the drug against the presence or absence of the adverse event.
The Statistical Toolkit: Disproportionality Analysis
Here are the heavy hitters in the statistical toolkit used by global regulators:
- PRR (Proportional Reporting Ratio): A simple ratio used primarily by the EMA. If 10% of reports for Drug A are “kidney failure” but only 2% of reports for all other drugs are “kidney failure,” the PRR is 5. Generally, a PRR > 2 with at least 3 cases and a Chi-square value > 4 is considered a signal.
- ROR (Reporting Odds Ratio): Similar to PRR but uses odds. It’s often preferred by researchers because it allows for more complex statistical adjustments, such as controlling for age or gender, which can help reduce confounding factors.
- IC (Information Component): Used by the WHO Uppsala Monitoring Centre. It uses Bayesian logic to reduce “noise,” which is especially helpful when dealing with very small numbers of reports. It provides a measure of the “unexpectedness” of a drug-ADR combination.
- BCPNN (Bayesian Confidence Propagation Neural Network): A fancy way of saying we use advanced probability to find patterns even in sparse data. This method is robust against the “masking effect,” where a very common side effect of one drug hides a rare side effect of another.

Beyond Ratios: Frequency and Imbalance
Beyond these ratios, we also look at frequency imbalances. The FDA Medical Reviewer’s Guidance suggests that in clinical trials, we should pay close attention to adverse events with an incidence of ≥5% that is at least twice the rate seen in the placebo group. For example, if a drug causes tachycardia in 5.3% of patients vs. 2.7% in placebo, that’s a risk ratio of 2.0—a clear signal to investigate further.
The Challenges of Statistical Detection
Statistical methods are powerful but not perfect. They suffer from several biases:
- The Weber Effect: Reporting tends to peak in the first two years after a drug’s launch and then declines, regardless of the actual safety profile.
- The Masking Effect: If a drug is known to cause a specific side effect (e.g., nausea), other side effects for that drug may be statistically “masked” because the denominator of reports is so high.
- Under-reporting: It is estimated that only 1-10% of serious adverse events are ever reported to spontaneous systems.
For a deeper dive into how these methods are applied in the real world, check out this Chapter 11: Signal detection methodology and application or our guide on post-marketing drug surveillance.
Next-Gen Tools: How Lifebit’s FMQs and PFRR Outperform Old Methods
Traditional methods are great, but they have a “granularity problem.” In the Medical Dictionary for Regulatory Activities (MedDRA), a single medical concept like “anxiety” might be split into dozens of different Preferred Terms (PTs) like “nervousness,” “panic attack,” or “generalized anxiety disorder.” If you only look at one term at a time, you might miss the bigger picture. This is known as the “splitting vs. lumping” dilemma in pharmacovigilance.
This is where Lifebit leverages the latest regulatory innovations like FDA Medical Queries (FMQs) and the Product Fractional Reporting Ratio (PFRR). These aren’t just buzzwords; they are precision instruments for Safety signal definition and detection. Using these advanced tools is part of our ultimate guide to AI for pharmacovigilance.
Boost Accuracy with Focused Safety Signal Detection Queries
Standardized MedDRA Queries (SMQs) have been the industry standard for years, but they can be a bit “blunt.” They often trade specificity for sensitivity, meaning they catch a lot of noise. FMQs (FDA Medical Queries) are the sharper alternative, developed by the FDA to improve the efficiency of safety reviews.
FMQs work by grouping related MedDRA terms into clinically meaningful clusters. A “Narrow FMQ” provides about a 90% probability that the medical concept you’re looking for has actually occurred. FMQs can be algorithmic, meaning they don’t just look at side effect terms; they can also pull in lab data (like a CPK level >5x the normal limit) and medication history to confirm a diagnosis like Rhabdomyolysis. By aggregating related terms, FMQs ensure that we don’t undercount risks just because different doctors used different words to describe the same problem.
When to Use PFRR for Reliable Signal Detection
What happens if you’re a smaller biotech company with only one or two products on the market? Traditional disproportionality methods (like PRR) fail because they need a massive database of other drugs to compare against. If your database only contains your own drug, you have no “background” to measure against.
The Product Fractional Reporting Ratio (PFRR) solves this. It’s an intra-product metric. Instead of comparing your drug to everyone else’s, it compares the frequency of an event now to the frequency of that same event in the past for your specific drug.
- When to use it: It’s most useful for small or specialized databases, such as those for orphan drugs or rare diseases, where traditional methods create too many false positives.
- The Golden Rule: PFRR is generally not recommended for very low case volumes, typically fewer than 10 cases during the surveillance period. If you have fewer than 10 cases, qualitative clinical review (reading the actual stories) is still your best bet.
By utilizing these next-gen tools, Lifebit helps organizations move beyond simple “term-matching” to a more holistic, clinically relevant understanding of drug safety.
The 5-Step Workflow to Validate and Reduce Drug Risks
Once a potential signal is detected, the real work begins. We don’t just pull drugs off the shelf because of a statistical “blip.” We follow a rigorous 5-step workflow designed to filter out noise and focus on genuine patient risks:
- Detection: The “smoke” is identified via statistical screening, literature review, or a cluster of unusual case reports. This is the “signal generation” phase.
- Validation: We look at the “quality” of the reports. Is the data complete? Is there a clear timeline? We use clinical judgment to decide if the signal is “real” or just a data artifact. Validation involves checking if the event is already listed in the Investigator’s Brochure (IB) or the Summary of Product Characteristics (SmPC).
- Prioritization: Not all signals are equal. A signal for “mild headache” is handled differently than a signal for “liver failure.” We prioritize based on the severity of the event, the public health impact, the vulnerability of the patient population, and the novelty of the drug. High-priority signals require immediate escalation to regulatory authorities.
- Assessment: This is the deep dive. We look at dechallenge (did the side effect go away when the drug stopped?) and rechallenge (did it come back?). We apply the Bradford Hill criteria to assess causality—looking for consistency across different studies, biological plausibility, and the strength of the statistical association. This phase often involves looking at “case series” to find common patient characteristics.
- Action & Risk Mitigation: If the signal is confirmed, we act. This could mean:
- Updating the drug label with a “Black Box Warning.”
- Issuing a “Dear Healthcare Professional” (DHCP) letter.
- Implementing a Risk Evaluation and Mitigation Strategy (REMS).
- In extreme cases, a market recall or withdrawal.
This process ensures that we are moving from “reactive” to real-time pharmacovigilance, catching risks before they become widespread crises. The ultimate goal is to maintain a favorable benefit-risk balance for the product throughout its entire lifecycle.
Real-Time Safety: How Lifebit AI and Federated Data Change Drug Monitoring Forever
The future of Safety signal detection isn’t just about better math; it’s about better data access. Traditionally, safety data is siloed. A regulator in Canada might see one thing, while a pharma company in the UK sees another. Moving large datasets across borders is often impossible due to strict privacy laws like GDPR or the HIPAA.
Lifebit’s federated AI platform changes this by allowing secure, real-time access to global biomedical data without the data ever leaving its original location. This “data-to-code” approach is the next frontier in pharmacovigilance.
The Power of Federated Learning
- Federated Learning: We can run signal detection algorithms across multiple databases (like the Canada Vigilance database and UK EHRs) simultaneously. This is crucial for detecting rare signals that only appear when you look at a massive, diverse population. Instead of moving the data, we move the model to the data, aggregate the results, and return the insights.
- Natural Language Processing (NLP): AI can read thousands of unstructured case narratives in seconds. It can extract key details—like the time to onset, concomitant medications, and medical history—that manual reviewers might miss in a mountain of paperwork.
- Trusted Research Environments (TREs): We provide a secure space where researchers can collaborate on safety data while strictly adhering to privacy laws. This ensures that patient identities are protected while still allowing for the deep-dive analysis required for signal assessment.
Proactive vs. Reactive Monitoring
By combining AI-driven pharmacovigilance solutions with a federated approach, we enable “proactive safety.” We aren’t just waiting for reports to trickle in; we are actively scanning the horizon for the first sign of trouble. This includes monitoring social media for patient-reported outcomes and using predictive modeling to identify which patients are at the highest risk for specific adverse events.
In the era of personalized medicine and complex biologics, the ability to analyze data in real-time across global borders is no longer a luxury—it is a requirement for ensuring patient safety.
FAQs and What’s Next in Safety Signal Detection
The landscape of drug safety is shifting. With the rise of complex biologics, gene therapies, and personalized medicines, the old ways of “wait and see” are no longer enough. At Lifebit, we believe that every patient deserves the protection of a real-time, data-driven safety net. If you want to see how our platform can supercharge your safety monitoring, visit https://lifebit.ai/.
What’s the difference between a safety signal and an adverse event?
An adverse event (AE) is simply any untoward medical occurrence in a patient taking a drug—it doesn’t have to be caused by the drug. It could be a coincidence. A safety signal is a pattern or a specific piece of information that suggests a causal link exists. Every signal starts as an adverse event, but only a small fraction of adverse events are ever validated as signals.
How do regulators like the FDA and EMA communicate signals?
Regulators have several megaphones to ensure healthcare providers and patients stay informed:
- FDA: Issues “Potential Signals of Serious Risks” reports quarterly and updates drug labels. They also use the MedWatch system for public alerts.
- EMA: Publishes signal recommendations from the Pharmacovigilance Risk Assessment Committee (PRAC) and updates the EudraVigilance database.
- Health Canada: Uses the Health Product InfoWatch for monthly updates and the Drug and Health Product Register (DHPR) for public access to safety reviews and recalls.
What are the limits of traditional signal detection in small datasets?
Traditional methods rely on large numbers to be statistically significant. In small datasets (like for rare diseases or pediatric populations), you run into:
- Statistical Noise: One or two coincidental cases can look like a massive signal (a false positive) because the denominator is so small.
- Under-reporting: Because the patient population is small, you might not get enough reports to trigger a traditional PRR alert, leading to false negatives.
- Masking: If one drug has a very famous side effect, it can “hide” other signals in the database.
This is why we use tools like PFRR and prioritize qualitative, expert-led case reviews for smaller datasets. The future lies in combining these statistical methods with AI that can understand the clinical context of every single report.
Ready to transform your pharmacovigilance?
Explore Lifebit’s Federated AI Platform

