Your Safety First: A Guide to Clinical Trial Protections

Clinical Trial Safety: 7 Protections That Stop Risks Before You Enroll
Clinical trial safety is built on a comprehensive system of safeguards designed to protect participants at every stage of research. Before you even consider joining a study, multiple layers of protection are already in place to minimize risks and ensure ethical conduct. This framework has evolved over decades, moving from basic ethical guidelines to a rigorous, multi-agency regulatory environment that prioritizes human life above scientific discovery.
Historically, the foundation of modern safety protocols can be traced back to the Nuremberg Code (1947) and the Declaration of Helsinki (1964). These documents established that the voluntary consent of the human subject is absolutely essential and that the risks of a study should never exceed the importance of the problem being solved. Today, these principles are codified into federal law, ensuring that every clinical investigator in the United States is legally and ethically bound to monitor and ensure participant safety.
Key Clinical Trial Safety Protections:
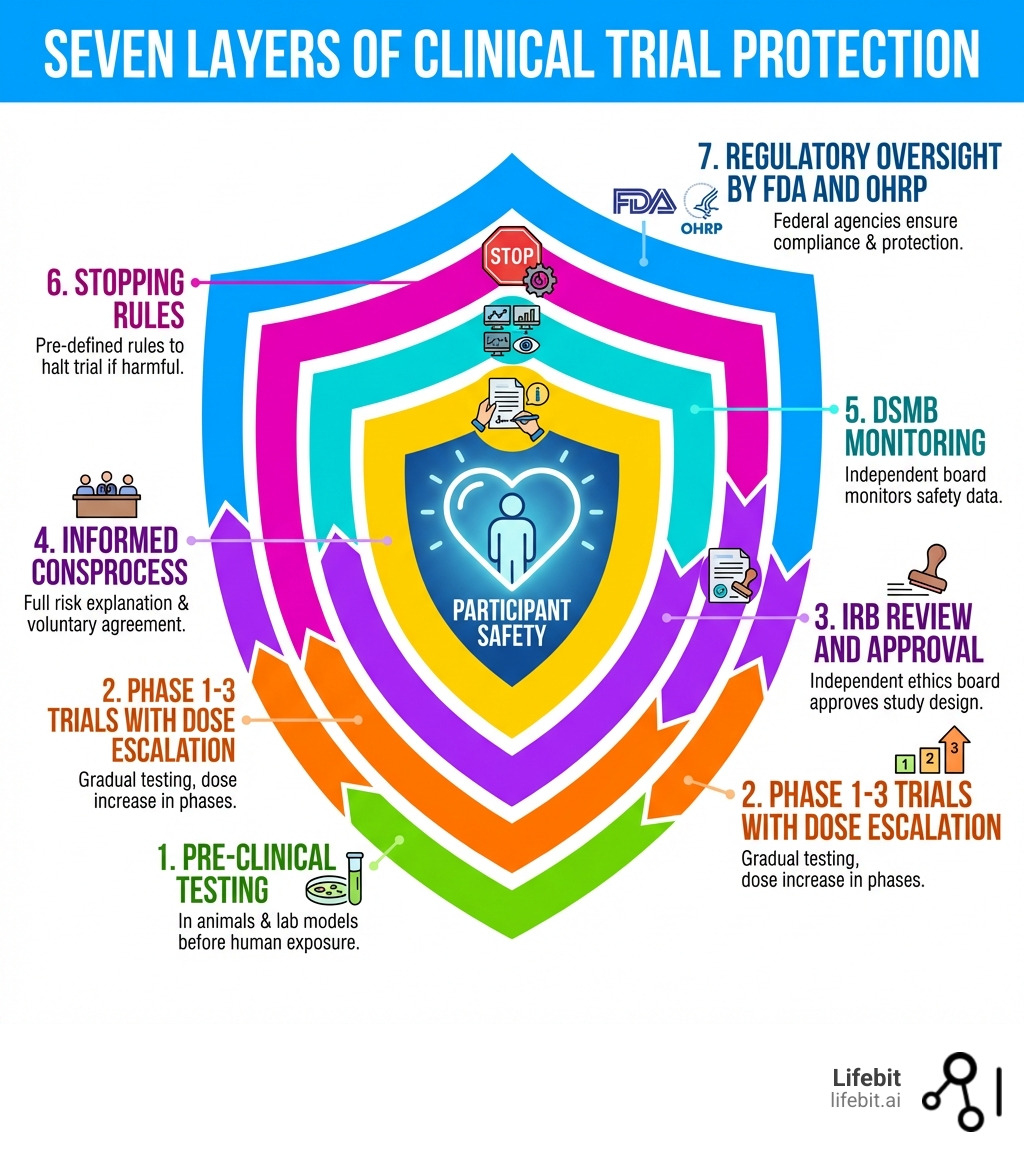
- Pre-clinical testing: Extensive laboratory and animal studies to identify toxicity before any human exposure.
- Institutional Review Boards (IRBs): Independent committees that approve, monitor, and review all research involving humans.
- Data Safety Monitoring Boards (DSMBs): Independent experts who monitor trial data in real-time and can stop trials if risks emerge.
- Informed consent: A continuous educational process that ensures participants understand all risks, benefits, and alternatives.
- FDA oversight: Strict adherence to Good Clinical Practice (GCP) and federal regulations (21 CFR Parts 50 and 56).
- Stopping rules: Pre-defined statistical triggers built into trial design to protect participants from harm or to stop a trial if a drug is proven effective early.
- Your right to withdraw: The absolute legal right to leave a study at any time, for any reason, without affecting your standard medical care.
The reality is simple: strict rules are in place to keep participants safe. Most clinical studies must be approved by an IRB before enrolling a single person. And if a DSMB determines that treatment is harmful, they have the authority to halt the trial immediately. This “safety-first” culture is the backbone of modern medicine.
Many people worry that clinical trials are risky or only for patients who have exhausted all other options. That’s a myth. While some trials do carry risks, particularly those testing new medications, the regulatory framework surrounding clinical research is designed specifically to minimize those risks and ensure that no participant is exposed to unnecessary harm.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, where we’ve spent over 15 years advancing Clinical trial safety through secure, federated data platforms that enable real-time safety monitoring and pharmacovigilance across diverse patient populations. Our work with public sector institutions and pharmaceutical organizations has shown how modern technology can strengthen traditional safety protocols by identifying subtle safety signals that might be missed in smaller, siloed datasets.
Clinical trial safety helpful reading:
How Pre-Clinical Testing and Dose Escalation Guarantee Clinical Trial Safety
When we talk about Clinical trial safety, we aren’t just talking about a single check-up or a one-page form. We are talking about a fortress of regulations designed to protect you. This framework begins long before a human ever receives a dose of an investigational drug, starting in the highly controlled environment of the laboratory.

Pre-Clinical Foundations: The Science of Toxicity
Before any clinical trial involving people is even considered, all investigational drugs are studied heavily in animals and cell cultures. This pre-clinical testing is essential because it helps researchers understand the ADME profile of a drug: how it is Absorbed, Distributed, Metabolized, and Excreted. We look for “target organ toxicity”—identifying if a drug might harm the liver, kidneys, or heart.
Researchers must submit an Investigational New Drug (IND) application to the FDA, which includes all animal data. The FDA then has 30 days to review the data to ensure the drug is safe enough to be tested in humans. If they find any concerns, they can issue a “clinical hold,” preventing the trial from starting until the safety issues are addressed.
The Phased Approach: A Safety Ladder
Once a drug moves into human trials, it follows a strict, step-by-step progression designed to minimize exposure while maximizing data collection:
- Phase 0: Sometimes used to conduct “microdosing” studies in a very small number of people (10-15) to see how the drug behaves in humans without causing any pharmacological effect.
- Phase 1: Focuses exclusively on safety and dosage. A small group (20-80 people) is monitored 24/7 to determine the “Maximum Tolerated Dose” (MTD).
- Phase 2: Expands to a larger group (100-300 people) to look at efficacy and further monitor safety. This is where we begin to see how the drug works in patients with the actual disease.
- Phase 3: Involves hundreds or thousands of patients to confirm effectiveness and monitor for side effects in a diverse population. This phase provides the “robust evidence” needed for FDA approval.
- Phase 4: Also known as post-marketing surveillance, this occurs after the drug is on the market to catch extremely rare side effects that might only appear in one in 100,000 people.
How Early Phase Testing Ensures Clinical Trial Safety
Phase 1 trials are the “gatekeepers” of human research. Their primary goal isn’t to see if a drug works, but to see if it is safe. To keep volunteers safe, we use several scientific benchmarks:
- NOAEL (No Observed Adverse Effect Level): This is the highest dose tested in animals that showed no significant harmful effects. It serves as the ceiling for our safety calculations.
- HED (Human Equivalent Dose): We use complex mathematical formulas to convert the animal dose to a human-appropriate scale, accounting for differences in metabolism and body surface area.
- MRSD (Maximum Recommended Starting Dose): We then apply a “safety factor” (usually a factor of 10 or more) to the HED. This ensures the first human dose is incredibly conservative—often a tiny fraction of what was safe in animals.
By using a dose escalation scheme (often called a “3+3 design”), researchers only increase the amount of drug given to a new group of participants after the previous, lower dose has been proven safe. If even one person has a serious reaction, the escalation stops. This meticulous “start low, go slow” approach is a cornerstone of Clinical trial safety.
Who’s Watching? How IRBs and DSMBs Stop Unsafe Clinical Trials Instantly
You might wonder, “Who is actually watching the researchers?” In clinical research, there are two main “watchdogs” that ensure ethical conduct and participant welfare. These groups are independent of the pharmaceutical companies and the doctors running the study, providing an unbiased layer of protection.
The Institutional Review Board (IRB)
An Institutional Review Boards (IRB) is a committee made up of at least five members, including doctors, scientists, and at least one member of the general public (like a community leader, ethicist, or patient advocate). Their job is to review the trial before it starts and at least once a year while it is running. They have the power to approve, require modifications to, or disapprove research. They ensure:
- The study is ethical: It follows the principles of the Belmont Report.
- Risks to participants are minimized: Using procedures that are consistent with sound research design.
- The benefits outweigh the risks: The potential knowledge gained must justify the risk to the individual.
- The informed consent form is clear: It must be written in plain language (usually at an 8th-grade reading level) so a non-scientist can understand it.
The Data and Safety Monitoring Board (DSMB)
While the IRB looks at the ethics and the site-level conduct, the Data and Safety Monitoring Board (DSMB) looks at the aggregate data across all study sites. These are independent experts in statistics and the specific disease being studied. Because they aren’t part of the research team, they can see “unblinded” data—meaning they know who is getting the real drug and who is getting the placebo—while the researchers remain “blinded” to prevent bias.
Real-Time Monitoring and Clinical Trial Safety Stopping Rules
One of the most powerful tools in Clinical trial safety is the “stopping rule.” These are pre-defined statistical conditions that, if met, require the trial to be paused or stopped immediately. These rules are written into the study protocol before the first patient is even enrolled.
A trial might be stopped early for three main reasons:
- Safety (Harm): If participants in the treatment group are experiencing unexpected or serious adverse events (SAEs) at a higher rate than the control group.
- Efficacy (Benefit): If the new drug is working so well that it would be unethical to keep giving the control group a less effective treatment. This happened in some early COVID-19 vaccine trials.
- Futility: If the data shows that the trial is unlikely to ever reach a statistically significant conclusion, it is stopped to avoid wasting participants’ time and exposing them to unnecessary risk.
Researchers are required to keep meticulous records of all adverse events—any negative change in health, from a mild headache to a serious complication. Serious Adverse Events (SAEs), such as hospitalizations or life-threatening events, must be reported to the FDA and the IRB within 7 to 15 days, ensuring no safety signal is missed.
Informed Consent: Your Right to Leave Any Clinical Trial Without Penalty
Informed consent is not just a form you sign; it is an ongoing educational process that continues throughout the entire duration of the study. It is the first and most important tool to help you understand how your rights, safety, and well-being will be addressed. It is designed to prevent the “therapeutic misconception”—the mistaken belief that the primary purpose of a clinical trial is to provide treatment rather than to conduct research.
What You Should Expect During the Process
During the informed consent process, the research team must have a face-to-face conversation with you (or a virtual one in decentralized trials) to explain:
- The purpose of the study: What are the researchers trying to learn?
- Inclusion/Exclusion criteria: Why you are a good fit and what conditions might make it unsafe for you to participate.
- Procedures and Duration: Every test, blood draw, and clinic visit you will undergo, and how long the study will last.
- Risks and Discomforts: All known side effects, from minor ones like nausea to rare but serious risks.
- Potential Benefits: While benefits aren’t guaranteed, the team must explain what they hope to achieve.
- Alternative treatments: What other FDA-approved drugs or therapies are available for your condition if you choose not to join the trial.
- Confidentiality: How your private health information will be protected.
- Your right to withdraw: You can leave the study at any time, for any reason, without any penalty to your regular medical care or loss of benefits.
This process is guided by The Belmont Report, which outlines three core values: Respect for Persons (treating individuals as autonomous agents), Beneficence (maximizing possible benefits and minimizing potential harms), and Justice (ensuring the fair distribution of the burdens and benefits of research).
Special Protections for Children and Vulnerable Populations
We take extra care when the participants are children, prisoners, or individuals with impaired decision-making capacity. Because children cannot legally give “consent,” we use a two-part process:
- Informed Permission: A legal guardian or parent must sign a detailed consent form after a full explanation.
- Assent: For children (often age 7 and up), the researchers explain the study in age-appropriate language. The child then gives their “assent” (agreement) to participate. If a child says “no,” even if the parent says “yes,” the child usually cannot be enrolled in the study unless the treatment is life-saving and unavailable elsewhere.
These strict rules were born out of a commitment to never repeat the mistakes of the past, such as the Tuskegee Syphilis Study, where participants were not informed of risks or given available treatments. Today, these historical lessons inform every safety regulation we follow in the US, UK, Canada, and beyond, ensuring that human rights are never sacrificed for scientific gain.
FDA Standards: How Real-Time Data Monitoring Makes Clinical Trials Safer
In the United States, the Clinical Trials and Human Subject Protection – FDA page outlines how the Food and Drug Administration oversees trials. They ensure that studies are designed, conducted, and reported according to federal law and Good Clinical Practice (GCP) regulations. GCP is an international ethical and scientific quality standard for designing, conducting, recording, and reporting trials that involve the participation of human subjects.
Other key agencies include:
- Office for Human Research Protections (OHRP): They oversee research supported by the Department of Health and Human Services (HHS) and ensure the “Common Rule” (the Federal Policy for the Protection of Human Subjects) is followed.
- National Cancer Institute (NCI): They fund and audit many oncology trials, ensuring they meet rigorous safety standards and that data integrity is maintained.
Modernizing Safety with Digital Health Data
At Lifebit, we believe that Clinical trial safety is entering a new era. Traditional monitoring relies on scheduled clinic visits, which only provide a “snapshot” of a patient’s health. However, digital health technologies allow for real-time surveillance. By integrating data from wearables, sensors, and Electronic Health Records (EHRs), we can detect safety signals much faster than ever before.
| Feature | Traditional Monitoring | Digital/Real-Time Monitoring (Lifebit) |
|---|---|---|
| Data Collection | Periodic (at clinic visits) | Continuous (via wearables/EHR) |
| Safety Signals | Reported by patient or doctor | AI-driven detection in real-time |
| Data Access | Fragmented across sites | Federated (securely linked) |
| Participant Burden | High (more travel/tests) | Lower (remote monitoring) |
| Data Security | Physical/Local storage | Trusted Research Environment (TRE) |
By using a Trusted Research Environment (TRE), we allow researchers to analyze sensitive data without it ever leaving its secure home (such as a hospital’s server). This protects participant confidentiality while providing the “robust evidence” the FDA requires for product safety. This federated approach means we can analyze data from thousands of patients across different countries simultaneously, identifying rare side effects in days rather than months. This is the future of pharmacovigilance—the science of collecting, monitoring, researching, and evaluating information from healthcare providers and patients on the adverse effects of medications.
Clinical Trial Safety: 4 Myths That Are Costing You Better Care
Misinformation about clinical trials can prevent patients from accessing potentially life-saving treatments. Let’s debunk the most common myths regarding Clinical trial safety.
Myth 1: Are clinical trials considered a last resort?
No! This is a common myth. While some trials focus on patients who have tried other treatments, many trials are for people at an early stage of a disease or even healthy volunteers. For example, prevention trials look for ways to stop a disease from ever occurring. Clinical trials offer access to state-of-the-art care and the next generation of medical breakthroughs long before they are available to the general public.
Myth 2: Will I always receive the experimental treatment?
Not necessarily. In a randomized controlled trial, you may be assigned to the experimental group or the control group. This is done to ensure the results are scientifically valid. The control group receives either the current “standard of care” (the best treatment currently available) or, in some cases, a placebo. You will be told during the informed consent process if there is a chance you will receive a placebo.
Myth 3: What is a placebo and is it safe?
A placebo is an inactive substance (like a sugar pill) that looks like the real drug. In clinical trials, placebos are only used when it is safe to do so—meaning that going without the active drug won’t cause you harm. If an effective treatment already exists for your condition, it is usually considered unethical to give a placebo alone. In those cases, you will usually receive the “standard of care” treatment plus either the new drug or a placebo.
Myth 4: Can I withdraw from a study at any time?
Yes. This is a fundamental right. You can stop participating at any time, for any reason, and you don’t even have to explain why. Your decision will not affect your regular medical care, your relationship with your doctor, or any benefits you are entitled to. We always recommend talking to the research team first so they can ensure you stop the treatment safely, as some medications require a “tapering” period to avoid withdrawal symptoms.
Myth 5: I’m just a “guinea pig” for big pharma.
This is a common fear, but the reality is that clinical trial participants are highly respected partners in research. Participants often receive more frequent medical check-ups, more detailed lab work, and more personal attention from a specialized medical team than they would in standard care. Furthermore, the strict oversight by IRBs and the FDA ensures that your safety is prioritized over the interests of any pharmaceutical company.
Secure Your Health: Why Real-Time Monitoring is the Future of Clinical Trial Safety
At Lifebit, our mission is to make the world’s biomedical data findable, actionable, and, above all, secure. We know that Clinical trial safety is the foundation of medical progress. Without the trust of participants, we cannot develop the treatments of tomorrow. By using our federated AI platform, researchers can perform real-time adverse drug reaction surveillance, catching potential issues long before they become serious problems.
The landscape of clinical research is shifting toward “Precision Medicine,” where treatments are tailored to an individual’s genetic makeup. This makes safety monitoring even more critical, as a drug that is safe for one person might be risky for another based on their DNA. Our technology allows for the secure analysis of genomic data alongside clinical data, ensuring that safety protocols are as personalized as the treatments themselves.
Whether it’s through the rigorous oversight of an IRB, the watchful eye of a DSMB, or the cutting-edge technology of a Trusted Research Environment, your safety is always the first priority. Clinical trials are the bridge to the future of medicine, and we are here to ensure that bridge is built on the strongest possible safety standards.
If you are considering a trial, talk to your healthcare provider and ask questions. You are an active partner in your own safety, and the entire medical community—from the FDA to independent researchers at Lifebit—is working together to protect you. By participating in a trial, you aren’t just helping yourself; you are helping to ensure that future generations have access to safer, more effective medical care.

