Clinical Research Singapore: A Hub for Global Health

Why Singapore Is the APAC Hub You Can’t Ignore for Clinical Trials
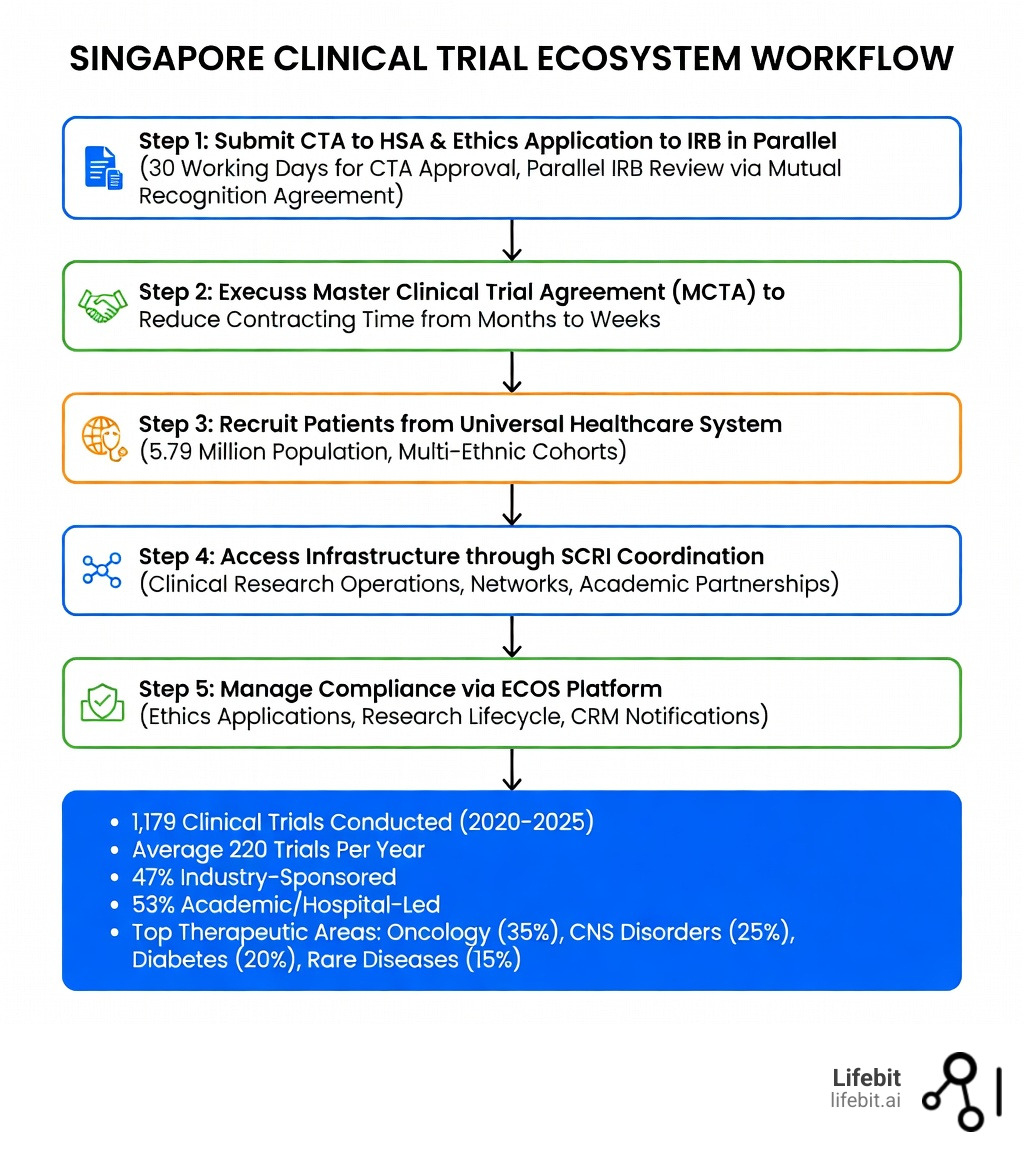
Clinical research Singapore has become the fastest-growing gateway for global pharma, biotech, and public health organizations looking to access diverse patient populations, streamlined regulatory pathways, and world-class healthcare infrastructure. Between 2020 and 2025, Singapore hosted 1,179 clinical trials with an average of 220 trials annually. The Health Sciences Authority (HSA) offers one of the fastest approval timelines in the region: 30 working days for Clinical Trial Authorisation (CTA) and just 15 working days for Phase I Bioequivalence studies. With a universal healthcare system covering 5.79 million people across multiple ethnicities, parallel IRB reviews through a Mutual Recognition Agreement, and government investment of US$18.5 billion in research and innovation under the RIE 2025 plan, Singapore delivers speed, quality, and scale for clinical development in Asia-Pacific.
Singapore’s strategic position as a “Biopolis”—a purpose-built biomedical research hub—has attracted over 60 global biopharmaceutical companies to establish regional headquarters or manufacturing plants here. This concentration of industry expertise, combined with the government’s commitment to the Research, Innovation and Enterprise (RIE) 2025 plan, ensures that the ecosystem is not just a site for trials, but a center for end-to-end drug development. The RIE 2025 plan specifically allocates significant funding toward Human Health and Potential (HHP), focusing on transforming the health of the population through precision medicine and advanced clinical research methodologies.
Key advantages of Clinical research Singapore:
- Regulatory speed: 30-day CTA approval, 15-day Phase I bioequivalence review, and the Clinical Trial Notification (CTN) scheme for low-risk trials.
- Multi-ethnic cohorts: Chinese, Malay, Indian, and other Asian populations in one location, providing a representative sample for the broader APAC region.
- Universal healthcare access: MediShield Life, MediSave, and MediFund support patient recruitment and long-term follow-up within a unified system.
- Infrastructure expansion: 13,600 new hospital beds planned between 2025–2030, alongside the integration of the National Electronic Health Record (NEHR).
- Top therapeutic areas: Oncology, CNS disorders, diabetes, rare diseases, and infectious diseases.
- Coordinated ecosystem: Singapore Clinical Research Institute (SCRI) serves as the national coordinating body, streamlining multi-site trial management.
- Industry and academic balance: 47% industry-sponsored, 53% academic/hospital-led trials, fostering a collaborative environment for translational research.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, a federated genomics and biomedical data platform powering secure, compliant analytics across siloed health datasets. With over 15 years in computational biology, AI, and health-tech entrepreneurship, I’ve worked alongside institutions advancing Clinical research Singapore through federated data environments that enable real-time insights without moving sensitive patient data. This guide will walk you through Singapore’s clinical trial ecosystem—from regulatory frameworks and patient recruitment advantages to the role of SCRI, precision health initiatives, and future infrastructure investments—so you can make informed decisions about launching or expanding trials in the region.
Glossary for Clinical research Singapore:
Why Clinical research Singapore is the Fastest Gateway to APAC Markets
When we look at the Asia-Pacific (APAC) landscape, speed and stability are the two most cited requirements for trial sponsors. Singapore doesn’t just meet these requirements; it sets the gold standard. With a stable, pro-business operating environment and the strongest intellectual property protection in the region, it has become a preferred “head office” hub for international biotechs. The World Bank consistently ranks Singapore among the top globally for ease of doing business, a factor that translates directly into the efficiency of clinical trial administration.
The numbers tell a compelling story. Between 2020 and 2025, Singapore hosted 1,179 clinical trials. While markets like China or Japan might offer larger raw population numbers, Singapore wins on efficiency and data quality. The HSA Clinical Trials Overview highlights a regulatory pathway that is remarkably streamlined. A Clinical Trial Authorisation (CTA) is typically processed within 30 working days, and Phase I Bioequivalence studies are even faster, taking approximately 15 working days. For trials involving therapeutic products already registered in Singapore or those with low risk, the Clinical Trial Notification (CTN) scheme allows for even more rapid commencement, often within 7 to 10 working days.
This efficiency is backed by a sophisticated stable and pro-business operating environment that includes skilled talent and advanced manufacturing capabilities. Singapore’s workforce is highly trained in Good Clinical Practice (GCP) and international regulatory standards, ensuring that data generated here is readily accepted by the FDA, EMA, and PMDA. For those of us focused on data, this environment is a goldmine. You can learn more about how this infrastructure supports data-heavy research in our Singapore biomedical data overview.
Scaling Clinical research Singapore Through Multi-Ethnic Genomic Data
One of the most unique aspects of conducting Clinical research Singapore is the ability to access a “microcosm of Asia.” Singapore’s population of 5.79 million is a diverse mix of Chinese (74%), Malay (13.5%), and Indian (9%) ethnicities. This is critical because Asian real-world data is historically under-represented in global life science research, despite Asians making up 60% of the world’s population.
By conducting trials here, sponsors can gather data that is representative of billions of people across the continent. This multi-ethnic cohort is the foundation for large-scale studies like the SG100K, which aims to sequence the genomes of 100,000 Singaporeans to better understand Asian-specific disease markers and drug responses. This project, led by the Precision Health Research, Singapore (PRECISE) office, is one of the most ambitious genomic initiatives in the region. For a deeper dive into how this data is structured, see our Singapore Clinical Data Complete Guide.
Streamlining Clinical research Singapore with Parallel IRB Reviews
In many countries, the “bottleneck” of clinical research is the Institutional Review Board (IRB) process, which can often take six months or more. Singapore has solved this through a Mutual Recognition Agreement among five major IRBs, including the National Healthcare Group (NHG) Domain Specific Review Board (DSRB) and the SingHealth Centralised Institutional Review Board (CIRB). This allows for parallel ethics reviews, meaning you don’t have to wait for HSA approval to start the IRB process, or vice versa.
Furthermore, the National Healthcare Group IRB Mutual Recognition ensures that collaborative studies across different hospital clusters can be reviewed by a single lead IRB. This “single point of entry” for ethics significantly reduces administrative burden. To manage this digitally, Singapore uses the ECOS (Ethics and Compliance Online System) platform, which provides a one-stop digital solution for the entire research lifecycle, from initial application to safety reporting and study closure. This system ensures that compliance doesn’t come at the cost of speed, providing real-time tracking for sponsors and investigators alike.
Cut Trial Start-Up Times with the Singapore Clinical Research Institute (SCRI)
If the HSA is the regulator, the Singapore Clinical Research Institute (SCRI) is the engine. Established in 2008 and appointed as the national coordinating body in 2021 under the Consortium for Clinical Research and Innovation, Singapore (CRIS), SCRI’s mission is to improve the standards of Clinical research Singapore by developing core capabilities and infrastructure that benefit both academic and industry partners.
SCRI operates through five key pillars that address the most common pain points in clinical development:
- Clinical Research Operations: Providing end-to-end support for trial management, including project management, monitoring, and data management services that adhere to international standards.
- Clinical Trials Infrastructure Coordination: Ensuring all hospitals and sites are “trial-ready” by standardizing processes and equipment across the public healthcare clusters.
- Clinical Research Networks (CRNs): Building specialized groups in areas like oncology, cardiovascular health, infectious diseases, and ophthalmology. These networks allow sponsors to quickly identify the best sites and investigators for specific therapeutic areas.
- Academic Research Partnerships: Bridging the gap between laboratory findings and clinical application by facilitating collaborations between universities and healthcare institutions.
- SCRI Academy: Training the next generation of Clinical Research Coordinators (CRCs) and investigators through certified programs, ensuring a steady pipeline of skilled professionals to support the growing number of trials.
One of SCRI’s most impactful tools is the Master Clinical Trial Agreement (MCTA). Historically, legal negotiations between sponsors and multiple trial sites could take months as each institution’s legal team reviewed the contract. The MCTA provides a pre-negotiated, standardized template that has been agreed upon by all major public healthcare institutions in Singapore. This reduces contracting time from several months to just a few weeks, allowing trials to move from “approved” to “active” in record time. For global sponsors, this predictability in timelines is a major competitive advantage when choosing a trial location.
Precision Health and Oncology: Solving the Region’s Biggest Health Burdens
In Singapore, the fight against disease is highly targeted and data-driven. Cancer is the leading cause of death, accounting for over 24% of all mortalities in the country. This reality has driven the government to pour resources into precision medicine, which aims to provide the right treatment for the right patient at the right time based on their genetic makeup.
A prime example is the National Cancer Centre Singapore Precision Oncology initiative, supported by a S$50 million national grant. This funding accelerates the use of genomic data to tailor treatments for Asian cancer patients, who often have different genetic mutations and responses to therapy compared to Western populations. For instance, EGFR mutations in lung cancer are significantly more prevalent in Asian non-smokers, a discovery that has led to more effective targeted therapies in the region.
Beyond oncology, the HELIOS (Health for Life in Singapore) study is a state-of-the-art population cohort study led by Lee Kong Chian School of Medicine in collaboration with Imperial College London. It tracks 50,000 individuals over 20 years to identify environmental, lifestyle, and genetic factors behind chronic diseases like diabetes and heart disease. Singapore has one of the highest rates of diabetes in the developed world, making this research a national priority.
These initiatives are generating massive, high-dimensional datasets—including genomic, proteomic, and imaging data—that require advanced AI and machine learning to process. The integration of these datasets allows for the identification of novel biomarkers and the development of predictive models for disease progression. If you are looking for partners in this space, you can find AI healthcare companies in Singapore that work with genomic data through our curated lists, which highlight the intersection of biotechnology and computational science in the city-state.
Stop Compliance Delays: Navigating CRM Notifications and PI Oversight
Managing Clinical Research Materials (CRM)—which include therapeutic products, medical devices, and placebos—is a critical part of trial logistics that can often cause delays if not handled correctly. In Singapore, the HSA Regulation of Clinical Research Materials requires a CRM notification before importing or supplying materials for local use. This ensures that all materials used in human subjects meet safety and quality standards.
However, there are smart shortcuts designed to facilitate research. Certain locally registered products are exempt from notification if they are used within their approved indications. Furthermore, the HSA allows for the import of unregistered materials specifically for research purposes without a full dealer’s license, provided the importer follows the CRM notification pathway. This flexibility is essential for Phase I trials involving experimental compounds that have not yet been approved anywhere in the world.
Equally important is the role of the Principal Investigator (PI) and the regulatory framework governing human research. The Human Biomedical Research Act (HBRA), enacted in 2015, provides a clear legal framework for the conduct of research involving human subjects, tissues, and data. Under the HBRA, PIs must ensure that “Appropriate Consent” is obtained and that the research is conducted with the highest ethical standards. In Singapore, a PI must be a qualified practitioner (doctor, dentist, or pharmacist) registered with the relevant local councils, such as the Singapore Medical Council (SMC).
PIs are responsible for:
- Maintaining the Investigator Site File (ISF), which must be audit-ready at all times.
- Ensuring all staff involved in the trial are on the delegation log and have received proper training.
- Reporting adverse events (AEs) and serious adverse events (SAEs) within strict HSA timelines (typically within 15 calendar days for serious unexpected adverse reactions).
- Ensuring the protection of participant privacy in accordance with the Personal Data Protection Act (PDPA).
If a PI needs to be away for more than four months, a formal change of PI application is required to ensure continuous oversight. This level of rigor and the clear legal definitions provided by the HBRA are exactly why Singapore’s trial data is trusted by the FDA, EMA, and other global regulators, facilitating the international acceptance of trial results.
13,600 New Beds: Building a Future-Ready Healthcare Ecosystem
Singapore is not resting on its laurels. Under the RIE 2025 Plan, the government has allocated US$18.5 billion for research, innovation, and enterprise. A significant portion of this is dedicated to expanding healthcare capacity and transforming the way care is delivered through the “Healthier SG” initiative, which focuses on preventive medicine and population health management.
The Ministry of Health Healthcare Expansion plan will add 13,600 new beds between 2025 and 2030 across new community hospitals and integrated acute care centers. This expansion isn’t just about physical space; it’s about integrating technology into the very fabric of the healthcare system. We are seeing a massive push into:
- AI diagnostics: Implementing AI-powered tools for early detection of conditions like diabetic retinopathy and various cancers through the National AI Office’s health initiatives.
- Robotic surgery and automation: Utilizing robotics to improve surgical precision and recovery times, as well as automating pharmacy and laboratory workflows to increase efficiency.
- Decentralized Clinical Trials (DCTs): Moving trials closer to the patient using home-based nursing, wearable sensors for remote monitoring, and tele-health consultations. This approach reduces the burden on participants and can improve recruitment and retention rates.
- National Electronic Health Record (NEHR): The continued expansion of the NEHR allows for a seamless flow of patient data across the entire healthcare ecosystem, which is invaluable for identifying eligible trial participants and tracking long-term outcomes.
Singapore’s universal healthcare system—comprising MediShield Life (health insurance), MediSave (medical savings accounts), and MediFund (a safety net for the needy)—ensures that patients from all walks of life can participate in trials without financial ruin. This creates a highly retained and motivated patient pool, which is the “secret sauce” for successful clinical outcomes. The government’s focus on “Value-Based Healthcare” also means that clinical research is increasingly focused on treatments that provide the best outcomes for the cost, aligning the interests of patients, providers, and payers. For more on the tech side, check out our guide on AI Healthcare Singapore.
Frequently Asked Questions about Clinical Research in Singapore
How long does it take to get clinical trial approval in Singapore?
The regulatory process in Singapore is one of the fastest in the world. The Health Sciences Authority (HSA) typically grants Clinical Trial Authorisation (CTA) within 30 working days. For Phase I Bioequivalence studies, the timeline is even shorter, at approximately 15 working days. Ethics reviews by IRBs are often conducted in parallel to save time, and the use of the ECOS platform streamlines the entire submission process.
What are the top therapeutic areas for trials in Singapore?
The primary therapeutic areas for Clinical research Singapore include oncology, CNS disorders (such as Alzheimer’s and Parkinson’s), diabetes, cardiovascular diseases, and rare diseases. This focus aligns with Singapore’s aging population and the high local burden of chronic diseases. Additionally, Singapore is a regional leader in infectious disease research, a capability that was further strengthened during the COVID-19 pandemic.
What is the role of the Singapore Clinical Research Institute (SCRI)?
SCRI is the national academic clinical research organization. It acts as a central coordinator that develops infrastructure, sets high standards for research, and facilitates collaborations between industry sponsors and academic institutions. Through its “SCRI Academy,” it also provides essential training for clinical research professionals, and its Master Clinical Trial Agreement (MCTA) significantly reduces trial start-up times.
Is clinical trial data from Singapore accepted by the US FDA or EMA?
Yes. Singapore follows the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Good Clinical Practice (GCP) guidelines. Data generated in Singapore is of high quality and is routinely accepted by major global regulatory bodies, including the FDA, EMA, and Japan’s PMDA, for drug registration purposes.
How does the Personal Data Protection Act (PDPA) affect clinical research?
The PDPA governs the collection, use, and disclosure of personal data. In clinical research, this means that sponsors and investigators must ensure that participants are fully informed about how their data will be used and that robust security measures are in place to prevent data breaches. The Human Biomedical Research Act (HBRA) provides additional specific protections for health-related data used in research.
Conclusion: How Lifebit Empowers Research in Singapore
As we have seen, Clinical research Singapore is a powerhouse of efficiency and innovation. The combination of rapid regulatory timelines, a multi-ethnic population, and a highly coordinated ecosystem makes it the ideal location for global biopharma companies. However, as trials become more data-intensive—incorporating multi-omic, genomic, and real-world data—the challenge moves from the clinic to the server. How do you analyze massive, sensitive datasets while keeping patient privacy 100% secure and complying with strict local laws like the PDPA and HBRA?
This is where Lifebit comes in. Our next-generation Lifebit Federated Biomedical Data Platform allows researchers to bring their analysis to the data. Instead of moving sensitive Singaporean patient records across borders or even between different hospital clusters—which is often prohibited or carries significant security risks—our Trusted Research Environment (TRE) enables secure, real-time collaboration.
By using federated AI and data orchestration, we help biopharma companies, research institutes, and government agencies in Singapore:
- Harmonize diverse datasets: Connect and standardize data from different hospital clusters (SingHealth, NHG, NUHS) without the need for a centralized, vulnerable database.
- Run advanced AI/ML analytics: Perform complex queries and train machine learning models on multi-ethnic genomic data to identify new drug targets and biomarkers.
- Maintain strict compliance: Ensure that all data processing occurs within the jurisdiction and under the control of the data owner, meeting all requirements of the PDPA and the Human Biomedical Research Act.
- Accelerate Discovery: Reduce the time spent on data cleaning and access requests, allowing scientists to focus on generating insights that lead to better patient outcomes.
Singapore provides the world-class clinical environment; Lifebit provides the world-class data environment. Together, we are accelerating the journey from a laboratory finding to a life-saving treatment for patients in Asia and across the globe. By bridging the gap between clinical excellence and data science, we are helping to realize the full potential of precision medicine in the heart of Asia.

