Beyond the Hype: What’s Driving the Real-World Data Market to Trillions?

Real World Data Market: Get Real-Time Patient Insights Without Compliance Risks
The real world data market is where healthcare information from everyday sources gets collected, analyzed, and used to shape medical progress. It’s not just a trend; it’s a fundamental shift in how we understand health and disease. For leaders in pharma, public health, and regulation, this market offers a powerful new way to get real-time insights from patient data — but it also comes with tough challenges around quality, access, and compliance.
Here’s a quick look at what the real world data market means for you:
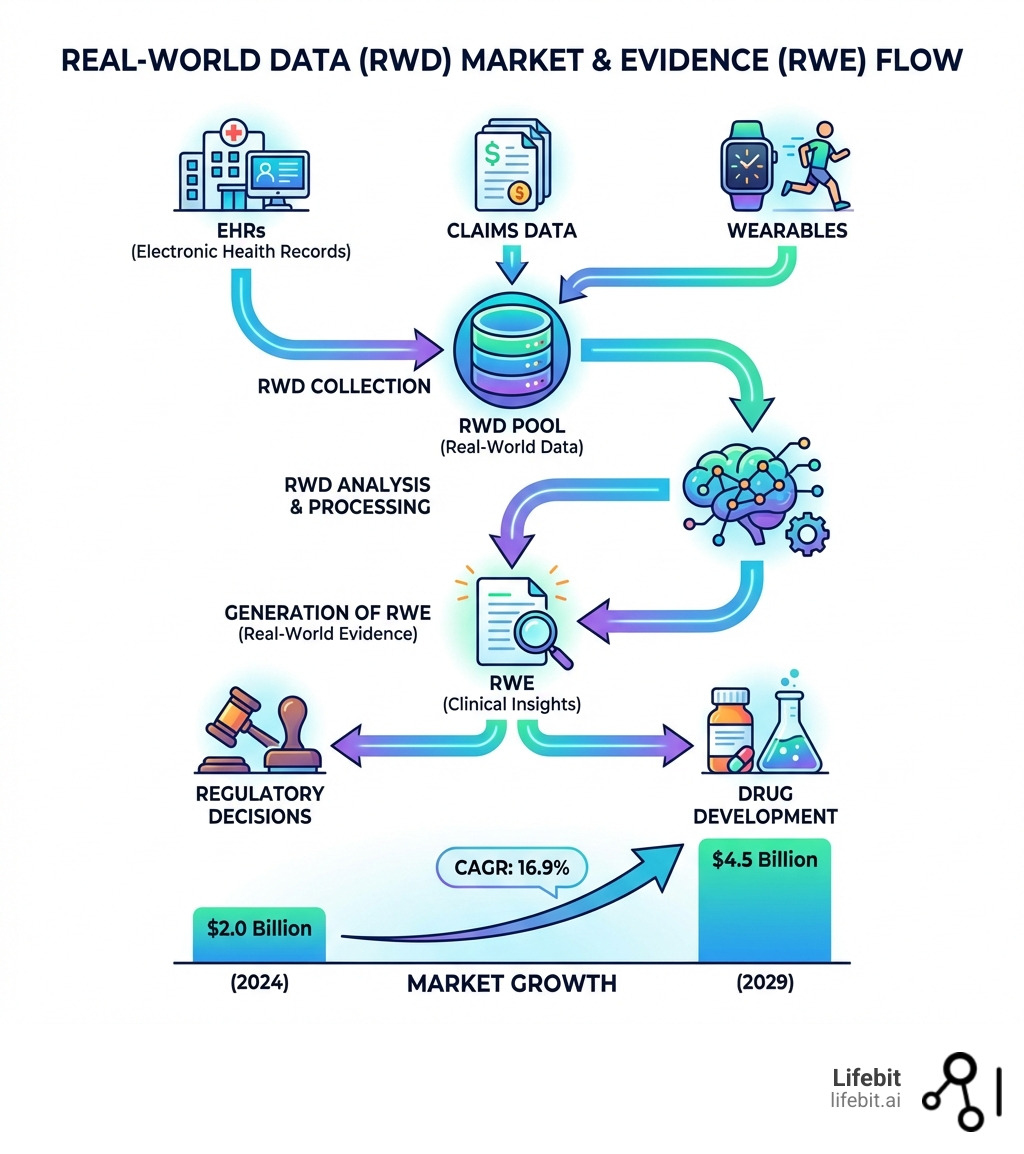
- What is Real-World Data (RWD)? This is health data routinely gathered outside of controlled clinical trials. Think electronic health records (EHRs), insurance claims, patient registries, and even data from wearable devices.
- What is Real-World Evidence (RWE)? This is the clinical insight you get after you analyze RWD. It shows how medical products perform in everyday patient care.
- Market Size & Growth: The global real world evidence solutions market is a powerhouse. It’s estimated at $2.0 Billion in 2024 and is projected to skyrocket to $4.5 Billion by 2029, growing at a robust 16.9% CAGR.
- Key Drivers: This explosive growth isn’t accidental. It’s fueled by a shift towards value-based care, the urgent need to cut costs and speed up drug development, and increasing acceptance from regulatory bodies like the FDA and EMA.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit. With over 15 years in computational biology and AI, I’ve seen how federated data analysis is changing the real world data market, empowering breakthroughs in precision medicine and drug findy. The insights we’re about to explore are critical for anyone looking to harness this data efficiently and ethically.
Learn more about real world data market:
Real World Data Market: How to Avoid the $2.5 Billion Drug Development Headache

If you feel like you’re hearing about the real world data market everywhere lately, it’s because the numbers are finally catching up to the hype. We are currently witnessing a massive surge in demand. In 2024, the market for real-world evidence solutions was valued at approximately $2.0 billion. By 2029, it is ready to hit $4.5 billion. That’s a compound annual growth rate (CAGR) of 16.9%—a pace that leaves traditional clinical research methods in the rearview mirror.
But why now? Why is the industry suddenly obsessed with data from the “real world”?
Why Value-Based Care Demands Real World Data
For decades, healthcare was a volume game: the more procedures or prescriptions, the more revenue. That is changing. We are moving toward value-based care, where providers and pharmaceutical companies are rewarded for actual patient outcomes. To prove that a drug works in a diverse, messy, real-world population—not just the “perfect” patients in a Phase III trial—you need RWD. Hospitals and payers are realizing that traditional approaches to patient care aren’t cost-effective anymore, and they are turning to RWD to find the gaps.
Stop the $2.5 Billion Drug Development Headache
Bringing a new drug to market now costs upwards of $2.5 billion. Even worse, only about 7.9% of drug development programs successfully reach the market. We simply cannot afford to keep failing at this rate. The real world data market offers a way to reduce these costs by optimizing trial designs, identifying the right patients faster, and even using “synthetic” control arms to replace some placebo groups.
Why Regulators Are Fast-Tracking Real World Evidence
Regulatory bodies are no longer just “interested” in RWD; they are actively building the infrastructure to use it. In the U.S., the 21st Century Cures Act was a watershed moment, requiring the FDA to establish a framework for using RWE in regulatory decision-making. Across the pond, the Real-world evidence | European Medicines Agency (EMA) is making similar strides, aiming to deliver 150 RWE studies annually by 2025 through the DARWIN EU network.
Understanding the nuance between the raw data and the evidence derived from it is key. You can dive deeper into this distinction here: Real-world data vs. real-world evidence.
Real World Data Market: Use Oncology and Post-Market Data to Speed Up Approvals
When we look at where the money is actually flowing within the real world data market, a few segments stand out as the clear winners. In 2023, the drug development and approvals segment accounted for the largest share of the industry. It’s not hard to see why: pharma companies are desperate for insights that can help them steer the treacherous path from lab to pharmacy shelf.
Why Oncology Leads the Real World Data Market
Within drug development, oncology is the undisputed heavyweight. Cancer treatments are increasingly personalized, targeting specific genetic mutations. Because these patient populations are often small, traditional clinical trials struggle to find enough participants. This makes Real-world data for clinical evidence generation in oncology absolutely vital. By looking at EHRs and genomic data from cancer centers across the globe, we can understand how these targeted therapies perform in the real world.
How Post-Market Surveillance Secures 30% of the Market
Once a drug or device is approved, the work isn’t over. Post-market surveillance is projected to hold about 30% of the RWD market share by 2025. Regulators and companies use this data to monitor long-term safety and effectiveness. If a rare side effect appears three years after launch, RWD is the tool that finds it.
Why Medical Device Makers Are Pivoting to RWE
It’s not just about drugs. The FDA has been very vocal about Leveraging Real World Evidence in Regulatory Submissions of Medical Devices | FDA. Whether it’s a heart valve or a new surgical robot, RWE helps manufacturers prove their devices are safe and effective in the hands of various surgeons across different hospital settings.
The Fastest-Growing Datasets in the Real World Data Market
To get these insights, we need high-quality fuel. The “services” segment currently holds the largest share of the market, but the “datasets” segment is growing at the fastest rate.
We typically categorize these datasets into several buckets:
- Claims and Billing Data: This is the “paper trail” of healthcare. It tells us what happened, when, and how much it cost.
- Electronic Health Records (EHR): This is where the clinical “meat” is. It includes doctor’s notes, lab results, and imaging.
- Pharmacy Data: Vital for tracking adherence. If a patient doesn’t fill their prescription, the drug won’t work.
- Patient Registries: Often focused on specific diseases, these provide deep, longitudinal data.
The real magic happens when you Link Claims EHR Data Real World Evidence. By combining the clinical detail of an EHR with the broad longitudinal view of a claims database, we get a 360-degree view of the patient journey.
Real World Data Market: Kill Data Silos and Access Global Insights with Federated AI
Despite the explosive growth, the real world data market isn’t without its growing pains. If we’re being honest, the biggest hurdle isn’t the data itself—it’s the people and the “walls” around the data.
How to Solve the Real World Data Talent Gap
There is a significant shortage of skilled professionals who can handle advanced digital solutions. It’s one thing to have a billion rows of data; it’s another thing entirely to have the data scientists, epidemiologists, and clinicians who know how to clean that data and ask it the right questions. This shortage is currently hindering market growth, as companies struggle to implement the complex software required for RWE generation.
Why Data Silos Are Killing Your Research Speed
Data is often stuck in silos. A hospital in London might have incredible genomic data, while a payer in New York has the longitudinal claims data. Because of strict privacy laws like GDPR in Europe and HIPAA in the U.S., moving this data across borders is a regulatory nightmare.
We’ve written extensively about these Challenges of Using Real-World Data in Research. The old way of doing things—copying data and moving it to a central server—is dead. It’s too risky and too slow.
Access Global Data Without Moving It via Federated AI
This is where we at Lifebit believe the future lies. Our federated AI platform allows researchers to “bring the analysis to the data” rather than moving the data to the analysis. By using a Trusted Research Environment (TRE), organizations can keep their data secure and compliant while still allowing authorized researchers to run models and gain insights. This overcomes the interoperability and privacy problems that have traditionally slowed down the real world data market.
How AI and Wearables Cut Insight Generation Time by 40%
If data is the fuel, AI is the engine. In 2024, roughly 67% of RWE providers incorporated AI-based analytics into their platforms. The result? A staggering 40% reduction in the time required to generate actionable insights.
Use NLP to Unlock Unstructured Patient Data
Much of the most valuable RWD is “unstructured”—think of a doctor’s handwritten notes or a pathology report. Natural Language Processing (NLP) allows us to “read” these documents at scale, turning messy text into structured data that we can actually analyze.
How Wearables Provide Real-Time Patient Monitoring
Devices like Apple Watches and Fitbits are changing the game for remote patient monitoring. We no longer have to wait for a patient to visit a clinic to see how they’re doing. We can track heart rate, sleep patterns, and activity levels in real-time. This is particularly useful in chronic disease management and decentralized clinical trials.
For a deeper dive into how these global assets are being used, check out the Exploring Real World Data around the globe SERIES INTRODUCTION.
Real World Data Market: How Regional Trends Impact Pricing and Reimbursement
The real world data market looks very different depending on where you are on the map.
- North America: The king of the hill. In 2024, North America held a massive 43% share of the market. This is driven by a well-established pharma sector, the widespread adoption of EHRs, and a regulatory environment (thanks to the FDA) that is pushing hard for RWE.
- Europe: A strong second, holding about 28.5% of the market. Europe’s strength lies in its centralized healthcare systems and initiatives like DARWIN EU, which are standardizing how RWD is used across the continent.
- Asia-Pacific (APAC): The one to watch. While it has a smaller share today, it is expected to grow at the highest CAGR (around 11%). Countries like Singapore are rapidly expanding their digital health infrastructure, and the sheer volume of patient data in the region is a goldmine for research.
The regional dynamics also influence how drugs are priced and paid for. You can see the Real-world data impact on price reimbursement to understand how these regional datasets are used to negotiate market access.
Real World Data Market: 3 Questions Every Pharma Leader Must Answer Now
RWD vs RWE: The Critical Difference for Your Strategy
Think of RWD as the raw ingredients (the flour, eggs, and sugar) and RWE as the finished cake. RWD is the raw data collected from EHRs, claims, and wearables. RWE is the clinical evidence—the actual “insight”—that you get after you analyze that data to see how a treatment is performing.
How the FDA Uses Real World Data to Approve Drugs
The FDA uses RWE to monitor post-market safety, support the approval of new indications for existing drugs, and satisfy post-approval study requirements. It’s a way for them to see how a drug performs in the “wild,” beyond the controlled environment of a trial.
Which Therapeutic Areas Are Dominating the RWE Market?
Oncology is the clear leader due to the need for precision medicine. However, we are seeing rapid growth in neurology, cardiovascular diseases, and immunology. Any area where patient outcomes are complex and long-term monitoring is required is a prime candidate for RWE.
Real World Data Market: Access Global Biomedical Data Securely with Lifebit
The real world data market is moving from a “nice-to-have” to a “must-have” for the entire healthcare ecosystem. But as we’ve seen, the path is littered with data silos, privacy concerns, and a shortage of experts.
At Lifebit, we’ve built a next-generation federated AI platform specifically to solve these problems. We provide secure, real-time access to global biomedical and multi-omic data without ever moving the data itself. Our platform, featuring the Trusted Research Environment (TRE) and our Real-time Evidence & Analytics Layer (R.E.A.L.), ensures that your research is compliant, secure, and fast.
If you’re ready to stop struggling with data silos and start generating world-class evidence, we should talk.
- Read our whitepaper on Innovating Healthcare RWD Strategies for Pharma Companies
- Find how our technology works: More info about Lifebit federation

