Finding Your Way Around a Clinical Trial Centre

Centre for Clinical Trials: How to Turn Lab Discoveries Into Real Patient Care
A centre for clinical trials is a dedicated facility or organization where new treatments, drugs, and medical interventions are rigorously tested in human volunteers under strict ethical and regulatory oversight. These centres sit at the heart of modern medicine — they are where scientific breakthroughs move from the lab into real patient care. Without these specialized hubs, the journey from a promising molecule in a petri dish to a life-saving medication in a pharmacy would be impossible.
Historically, clinical research was often fragmented, conducted in small pockets of academic hospitals without standardized protocols. Today, a modern centre for clinical trials is a highly sophisticated engine of innovation. These institutions must navigate a complex landscape of international regulations, such as Good Clinical Practice (GCP) guidelines, which ensure that the rights, safety, and well-being of trial participants are protected and that the clinical trial data are credible.
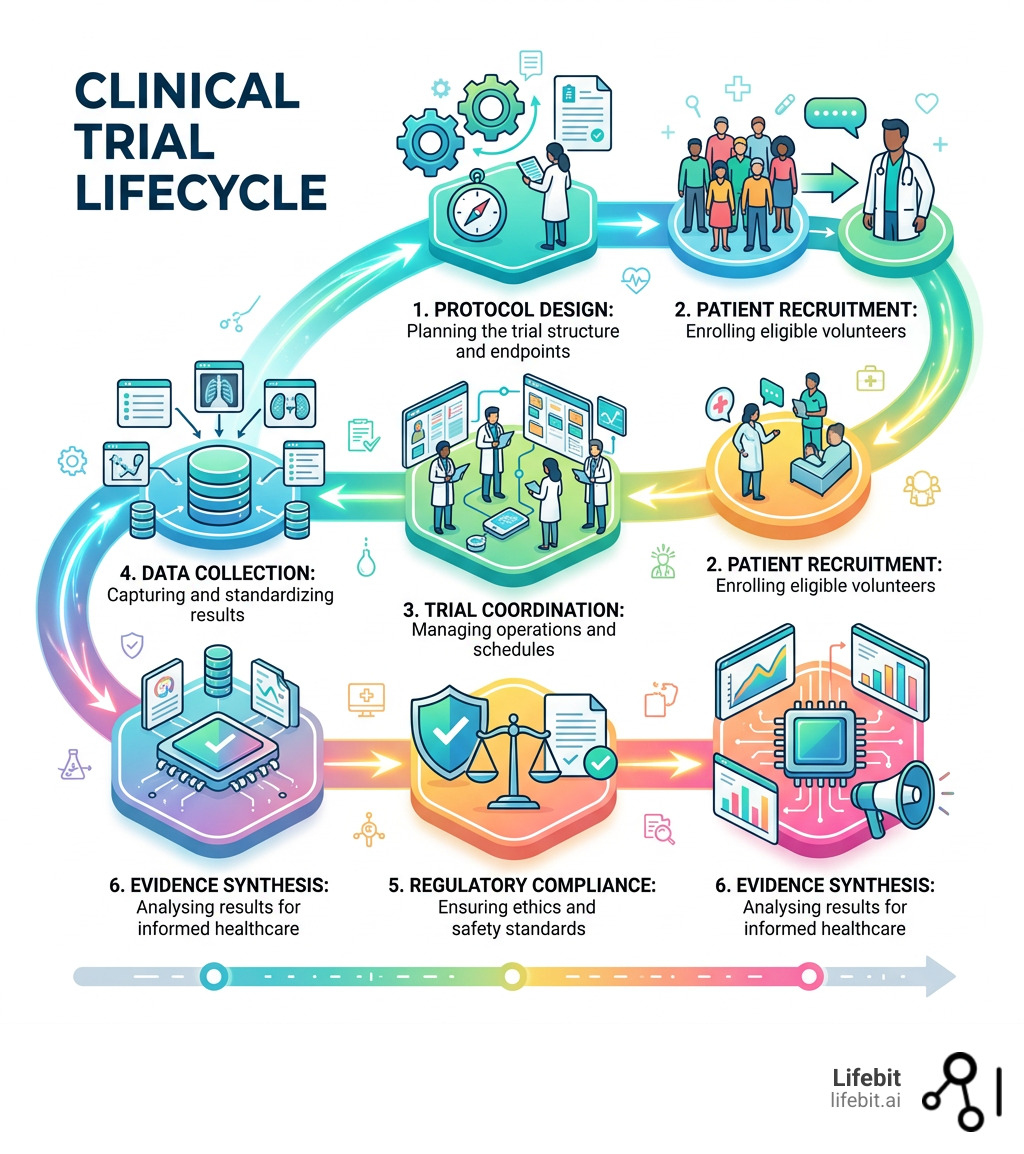
Here is a quick overview of what these centres do:
| Function | What It Means |
|---|---|
| Protocol design | Plan how a trial is run, who qualifies, and what is measured |
| Patient recruitment | Find and enroll eligible participants |
| Trial coordination | Manage day-to-day operations across one or many sites |
| Data collection | Capture and standardize trial results |
| Regulatory compliance | Ensure the trial meets ethics and safety standards |
| Evidence synthesis | Analyze results to inform real-world health decisions |
Some centres focus on a single disease area like cancer. Others run trials across dozens of therapeutic areas, from cardiovascular health to rare genetic disorders. Some are government-run research hospitals, such as the NIH Clinical Center. Others are academic institutions, independent trusts, or private contract research organizations (CROs) that partner with pharmaceutical companies to accelerate drug development timelines.
The scale varies too. The NIH Clinical Center in Bethesda, Maryland — the nation’s largest hospital devoted entirely to clinical research — operates as a 200-bed facility running federally supported studies. Meanwhile, community-based centres like those under the NCI’s NCORP network bring trial access directly to patients in their own neighborhoods, ensuring that research is not just high-quality, but also accessible to diverse populations.
What they all share is a common goal: generate reliable evidence to improve human health.
I’m Maria Chatzou Dunford, CEO and Co-founder of Lifebit, and my work at the intersection of genomics, federated data platforms, and AI has given me a front-row seat to how data infrastructure shapes the speed and quality of research conducted at every centre for clinical trials. In this guide, I’ll walk you through how these centres are structured, funded, and accessed — whether you’re a researcher, a patient, or a data and policy leader. We will explore the intricate balance between scientific rigor and patient-centric care that defines the world’s leading research institutions.
Quick centre for clinical trials terms:
Inside a Centre for Clinical Trials: How Protocols Protect Patient Safety
At its core, a centre for clinical trials functions as a bridge between laboratory discovery and the pharmacy shelf. These institutions are often housed within large research hospitals where the primary mission isn’t just treating patients with existing tools, but inventing the tools of tomorrow. This dual mission requires a unique organizational structure where clinical care and scientific inquiry coexist seamlessly.
When you Search the Studies at a major hub like the NIH, you aren’t just looking at a list of appointments; you are looking at highly structured scientific experiments. Every trial begins with a protocol design—a detailed “recipe” that dictates exactly how the study will be conducted. This protocol outlines the inclusion and exclusion criteria (who can join), the dosage of the intervention, the frequency of tests, and the primary endpoints (what constitutes success). This ensures that every participant is treated safely and that the data collected is scientifically valid and reproducible.
The Multidisciplinary Team
A successful centre for clinical trials relies on a diverse team of experts. This includes:
- Principal Investigators (PIs): Usually senior physicians who lead the study and take responsibility for its scientific and ethical conduct.
- Clinical Research Coordinators (CRCs): The “engine room” of the trial, managing the day-to-day logistics, patient schedules, and data entry.
- Biostatisticians: Experts who ensure the study is powered correctly to yield statistically significant results.
- Ethics Committees: Independent bodies that review the protocol to ensure participant rights are upheld.
Patient safety is the absolute priority. Before a single person is enrolled, an Institutional Review Board (IRB) must approve the study. Once the trial is live, researchers monitor participants closely, often using advanced diagnostic tools that are not yet available in standard clinical practice. For a deeper look at the foundational steps of these studies, check out our Clinical Trial Complete Guide.

The Role of the Coordinating Center for Clinical Trials (CCCT)
While individual hospitals see the patients, the Coordinating Center for Clinical Trials (CCCT) acts as the “brain” for large-scale research networks, particularly within the National Cancer Institute (NCI). Its primary role is to support the NCI’s clinical and translational research programs.
Instead of treating patients directly, the CCCT focuses on the infrastructure that makes research possible. It coordinates between different institutions, manages funding programs, and ensures that scientific steering committees are aligned. This is particularly important for multi-site trials, where a drug might be tested in 50 different hospitals simultaneously. Without this high-level coordination, data would remain siloed, making it nearly impossible to draw broad conclusions about new cancer therapies. Proper management of this information is vital, as outlined in our Clinical Trial Data Management Complete Guide.
Inside the NIH Clinical Center: America’s Research Hospital
Located in Bethesda, Maryland, the NIH Clinical Center is a unique beast in the medical world. It is a 200-bed research hospital where every single patient is a participant in a clinical study. Because it is federally funded, the costs for medical care received within a trial are typically covered by the NIH, removing financial barriers for participants who might be seeking experimental treatments for rare or terminal conditions.
What makes it special?
- High Reliability: It operates on principles of extreme patient-centric care within a high-stakes research environment, maintaining a low patient-to-nurse ratio.
- Unique Facilities: It includes specialized units for early-phase studies (Phase I) that require intensive monitoring, overnight stays, and rapid response teams.
- Education: It offers clinical electives for medical students to learn about rare disorders firsthand, training the next generation of physician-scientists.
If you can’t visit in person, you can actually Take the NIH Virtual Tour to see how this massive operation functions from the inside, from the state-of-the-art imaging suites to the specialized metabolic units.
Stop Data Fragmentation: How a Centre for Clinical Trials Secures Funding
Running a centre for clinical trials is expensive. From the specialized staff to the advanced imaging equipment like PET-CT scanners and high-throughput sequencers, the costs are astronomical. A single Phase III trial can cost upwards of $100 million. Funding often comes from a mix of government grants (like those from the NIH or MRC), philanthropic donations, and industry partnerships with biotech and pharmaceutical giants.
The CCCT provides several critical funding opportunities that keep the research ecosystem healthy:
- BIQSFP (Biomarker, Imaging, and Quality of Life Studies Funding Program): Supports the integration of biomarkers and imaging into large-scale trials, allowing researchers to see why a drug works, not just if it works.
- Clinician Scientist Awards: Helps doctors balance their time between treating patients and conducting research, ensuring that clinical insights inform laboratory work.
- NAVIGATE: A key partnership with the Veterans Health Administration to help veterans get easier access to NCI-funded trials, particularly in rural areas where access to specialized care is limited.
Managing these multi-million dollar initiatives requires robust Medical Trial Software to track spending, enrollment, and outcomes in real-time. Financial transparency is essential to maintain public trust and ensure that resources are allocated to the most promising scientific avenues.
Streamlining Data with the Clinical Trials Reporting Program (CTRP)
One of the biggest headaches in research is “data fragmentation.” When different centres use different formats to record patient outcomes, the data cannot be easily combined. To solve this, the NCI established the Clinical Trials Reporting Program (CTRP). This is a comprehensive database that tracks every interventional clinical trial funded by the NCI.
By maintaining this central repository, the CTRP helps:
- Identify Gaps: See which diseases aren’t getting enough research attention, allowing funders to pivot resources toward unmet medical needs.
- Avoid Duplication: Ensure two different centres aren’t accidentally running the exact same study, which wastes time, money, and participant effort.
- Standardize Data: Make sure that “success” is measured the same way in New York as it is in London, facilitating international meta-analyses.
For researchers, this level of organization is the foundation for a Clinical Trial Data Analysis Complete Guide. It allows for the secondary use of data, where researchers can look back at old trials to find new patterns or insights.
Scientific Steering Committees and Trial Oversight
Who decides which trial is worth running? That falls to the Scientific Steering Committees. These groups are made up of experts from the National Clinical Trials Network (NCTN), the Investigational Drug Steering Committee (IDSC), and the NCI Community Oncology Research Program (NCORP). They evaluate the scientific merit, feasibility, and potential impact of proposed studies.
Importantly, these committees also include Patient Advocates. These are individuals who have lived through the diseases being studied and provide a “real-world” reality check on trial design. They might point out that a trial requires too many hospital visits for a sick patient to manage, or that the side effects being measured aren’t the ones that matter most to patients. They ensure that Phase 2 and Phase 3 trials aren’t just scientifically sound, but also practical and ethical for the people who have to participate in them.
Use AI to Cut Research Time at Your Centre for Clinical Trials
Not every centre for clinical trials is a hospital. Some, like the Center for Clinical Trials and Evidence Synthesis (CCTES) at the Johns Hopkins Bloomberg School of Public Health, focus on the math and methodology behind the medicine. These institutions are the architects of research, designing the frameworks that other hospitals use to test new drugs.
Housed within the Department of Epidemiology, the CCTES is world-renowned for teaching researchers how to design better studies. They even offer a Coursera: Clinical Trials Operations specialization for those who want to build these skills from home. Their Trial Advisor Program acts as a consultancy, helping investigators polish their analysis plans before they even start, which significantly reduces the risk of trial failure due to poor design.
Advancing Methodology at a Clinical Trials Centre
The legacy of pioneers like Dr. Curtis Meinert continues to shape how we view “evidence synthesis”—the art of combining data from many different studies to find the truth. Modern centres are now integrating AI and Large Language Models to sift through thousands of research papers in seconds, identifying trends that a human researcher might miss.
AI is also being used for:
- Predictive Enrollment: Using historical data to predict which sites will recruit patients the fastest.
- Synthetic Control Arms: Using data from previous trials to create a “virtual” control group, reducing the number of patients who need to be given a placebo.
- Risk-Based Monitoring: Using algorithms to identify which trial sites are most likely to have data errors, allowing monitors to focus their efforts where they are needed most.
New designs, such as the 2-in-1 design in oncology, allow researchers to transition seamlessly from a Phase 2 to a Phase 3 trial based on interim data, saving years of development time. To understand how these technologies are changing the game, see our AI Clinical Trials Ultimate Guide.
Training the Next Generation of Researchers
To keep the wheels of innovation turning, these centres must train new talent. This happens through a variety of pathways:
- Graduate Coursework: Deep dives into biostatistics, epidemiology, and the ethics of human experimentation.
- NIH Research Festival: An annual event featuring lectures and workshops to showcase the latest breakthroughs and foster collaboration between different scientific disciplines.
- Clinical Rotations: Giving medical students “boots on the ground” experience in a research hospital, where they learn that medicine is an evolving science, not just a set of established facts.
Improving the way we find and keep participants is a major part of this training. High dropout rates can ruin a study, so researchers are now being trained in patient engagement strategies, often highlighted in our Improve Patient Recruitment Clinical Guide. This includes learning how to communicate complex scientific concepts in plain language and how to build trust with underrepresented communities.
Find a Centre for Clinical Trials: Access Life-Saving Treatments Today
For many patients, a centre for clinical trials represents a source of hope when standard treatments have failed. Whether it is a new immunotherapy for cancer or a gene therapy for a rare neurological condition, trials offer access to the cutting edge of medicine. But finding the right trial can feel like looking for a needle in a haystack, especially when dealing with the stress of a serious diagnosis.
How to Find the Best Match
Navigating the world of clinical research requires the right tools. Here are the most reliable platforms for finding a trial:
| Platform | Best For |
|---|---|
| CenterWatch | Browsing over 40,000 listings by medical condition and geographic location |
| Cancer.gov | Finding NCI-sponsored oncology trials and getting expert support via chat |
| ClinicalTrials.gov | The global gold standard for searching all registered studies across 200+ countries |
| Health Buddy App | Localized trial tracking in regions like Singapore, focusing on patient convenience |
Using these tools is a major step in Patient Recruitment in Clinical Trials. It is important to discuss any trial you find with your primary care physician or specialist to ensure it is a safe and appropriate option for your specific health profile.
Participating in Trials at International Centres
Research is a global effort. We see incredible work being done at the National Cancer Centre Singapore (NCCS), which has over 20 years of experience and has completed more than 500 trials. They are pioneers in home-based care and teleconsult services, which allow patients to participate in trials without having to live at the hospital. This “decentralized” approach is the future of research, making it easier for people with jobs and families to contribute to science.
Similarly, Aotearoa Clinical Trials in New Zealand and the UCL Clinical Trials Unit in London are pushing the boundaries of what is possible. These international hubs often collaborate on multi-regional trials to ensure that new drugs work for diverse populations with different genetic backgrounds. Our Clinical Trial Patient Recruitment Complete Guide explores these strategies in depth, emphasizing the need for diversity in research to ensure that treatments are effective for everyone, not just a narrow demographic.
Finding the Right Study for Your Condition
Many centres now offer volunteer notifications. You can sign up to receive an email the moment a study matching your specific symptoms or condition is posted. This is especially life-changing for those with rare disorders, where trials may only open once every few years and fill up within days. Digital platforms are also making the informed consent process easier, using videos and interactive tools to help patients understand the risks and benefits before they sign up. For more on the digital shift in this space, read our Digital Patient Recruitment Complete Guide.
Centre for Clinical Trials FAQ: Find Matches and Training Opportunities
How do I find a clinical trial that matches my symptoms?
The best way is to use a registry like Clinicalstudies.info.nih.gov. You can search by keyword (like “diabetes” or “migraine”) and filter by location. Many sites also allow you to match your symptoms to specific protocols automatically. Additionally, patient advocacy groups for specific diseases often maintain their own curated lists of trials that are particularly promising for their community.
What unique facilities does the NIH Clinical Center offer?
As a dedicated research hospital, it offers specialized metabolic suites for nutrition studies, advanced imaging (like 7-Tesla MRI) that isn’t available in standard hospitals, and an inpatient early-phase unit designed for intensive monitoring and pharmacokinetic sampling. It also houses the Department of Bioethics, which provides consultation services for complex ethical dilemmas that arise during research.
What educational opportunities exist for clinical trial professionals?
Beyond traditional university degrees, you can find graduate-level coursework in clinical trial management, NIH clinical electives for medical and dental students, and online specializations via platforms like Coursera. Many centres also host monthly seminars, “research festivals,” and GCP (Good Clinical Practice) certification workshops to keep professionals updated on the latest methodologies and regulatory changes.
Are clinical trials free for participants?
In many cases, yes. At the NIH Clinical Center, for example, all medical care related to the trial is provided at no cost. Many industry-sponsored trials also cover the cost of the experimental drug and required tests. Some trials even provide reimbursement for travel, lodging, and meals. However, it is vital to clarify this with the trial coordinator during the informed consent process, as “standard of care” costs may still be billed to your insurance in some settings.
Future-Proof Your Centre for Clinical Trials With Federated AI Platforms
The traditional centre for clinical trials is evolving. We are moving away from a world where data is locked inside a single building and toward a future of federated AI. In the past, if a researcher in Tokyo wanted to compare their data with a researcher in Berlin, they would have to physically move massive datasets, which is a nightmare for both security and patient privacy.
At Lifebit, we believe that the next generation of breakthroughs will come from connecting these global centres through a Trusted Clinical Environment. By using federated technology, researchers can analyze multi-omic and biomedical data from across five continents without the data ever leaving its secure home. This “brings the analysis to the data” rather than the other way around.
This approach solves several critical problems:
- Data Sovereignty: National health data stays within its country of origin, complying with laws like GDPR.
- Security: It eliminates the risk of data breaches during transfer.
- Scale: It allows for the study of rare diseases by pooling data from thousands of small centres into one massive, virtual cohort.
This protects patient privacy while accelerating the speed of discovery to a pace we’ve never seen before. We are entering an era of “Precision Medicine,” where a centre for clinical trials won’t just test if a drug works for the average person, but if it works for you, based on your unique genetic makeup and lifestyle.
Whether you are a patient looking for a trial or a researcher looking for a better way to manage data, the centre for clinical trials remains the most important destination on the map of human health. It is where hope meets science, and where the future of medicine is written every single day.

