The Ultimate Guide to Post-Marketing Drug Safety Surveillance

Why Drug Safety Data Is the Backbone of Every Safe Medicine on the Market
Drug safety data is the information collected before, during, and after a drug reaches patients — and it’s what stands between a life-saving treatment and a preventable harm.
Here are the key sources and systems you need to know:
| Source | What It Covers | Who It’s For |
|---|---|---|
| FDA FAERS | Adverse events, medication errors, product quality complaints | Researchers, regulators, industry |
| FDA Drug Safety Communications | New warnings, labeling changes, recalls | Healthcare professionals, consumers |
| NIOSH Hazardous Drugs List (2024) | Drugs posing occupational risks in healthcare settings | Healthcare workers, employers |
| Safety Data Sheets (SDS) | Safe handling, health effects, disposal of drugs and chemicals | Pharmacists, lab workers, distributors |
| MedWatch | Voluntary adverse event reporting portal | Consumers, healthcare professionals |
| AEMS Public Dashboard | Unified adverse event data across all FDA-regulated products | Public, researchers, industry |
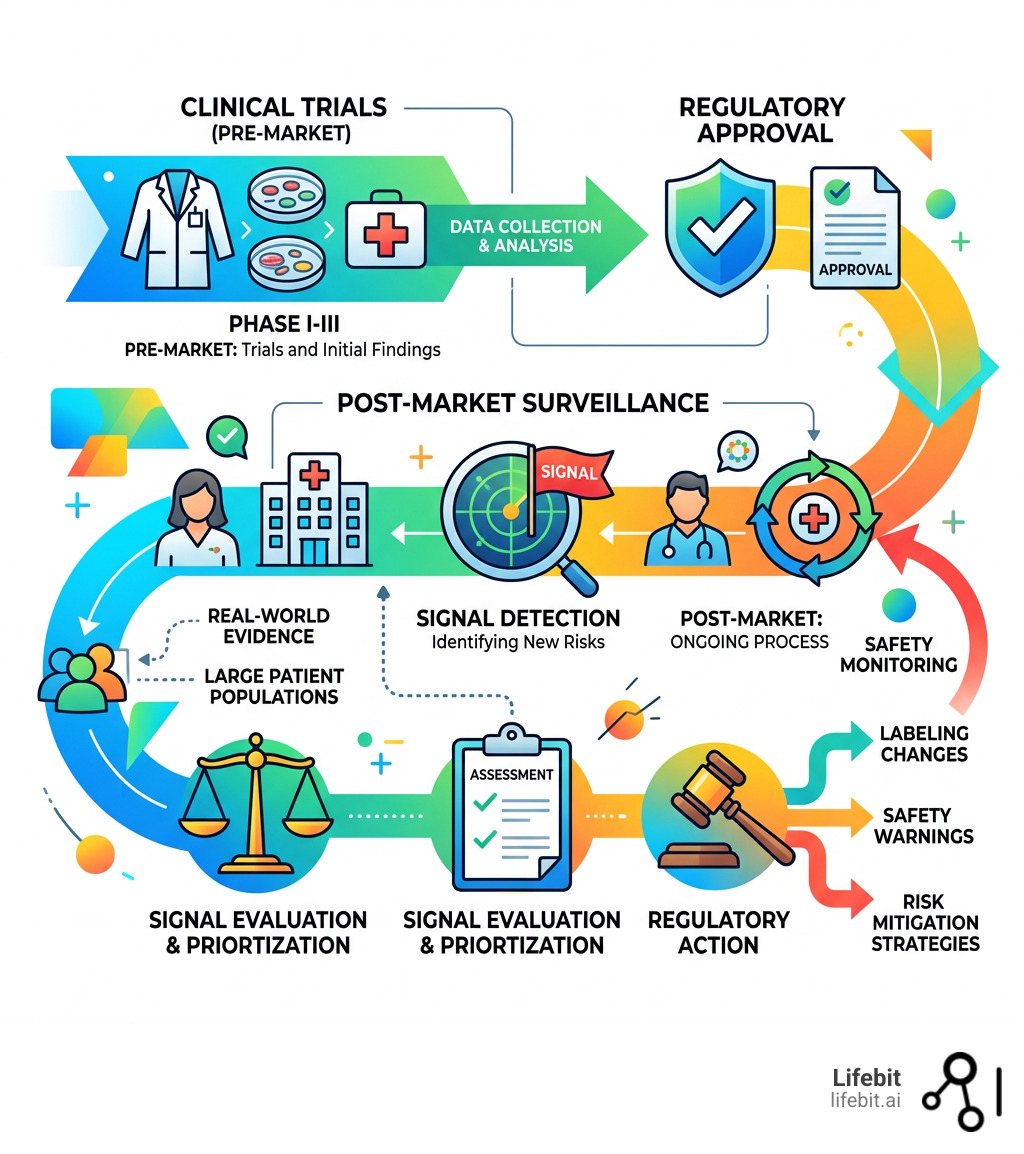
After a drug is approved, the real-world safety story is just beginning. Patients are more diverse than clinical trial populations. Rare side effects only surface at scale. Drug interactions emerge over years, not months. That’s why post-marketing surveillance — the ongoing monitoring of drugs once they’re in the hands of real patients — is not optional. It’s essential.
Yet the systems built to capture this data are fragmented, often manual, and riddled with limitations. Reports go unverified. Duplicates pile up. Critical signals hide inside millions of unstructured text entries — emails, call center notes, social media posts — that no human team can read fast enough.
For pharma companies, regulators, and public health institutions, the challenge isn’t just accessing drug safety data. It’s making sense of it, at scale, in time to act.
I’m Dr. Maria Chatzou Dunford, CEO and Co-founder of Lifebit, and over 15 years building computational biology tools and federated AI platforms, I’ve seen how fragmented infrastructure slows the flow of critical drug safety data to the people who need it most. This guide draws on that experience to help you navigate the full landscape — from FDA databases to AI-powered surveillance.
Primary Sources for Accessing Critical Drug Safety Data
When we talk about drug safety data, we aren’t just looking at one spreadsheet. We are looking at a massive, interconnected web of regulatory filings, clinical observations, and occupational guidelines. For anyone working in healthcare or drug development in the US, UK, Canada, or Europe, knowing where to pull this data is the first step toward proactive post-marketing drug surveillance.
The FDA is the primary gatekeeper for this information in the United States. Their Drug Safety Communications provide the most current updates on new safety issues, helping us make informed treatment decisions. These communications often lead to labeling changes or, in more serious cases, drug recalls. However, global safety monitoring requires looking beyond US borders.
Global Regulatory Databases
- EudraVigilance (EMA): The European Medicines Agency maintains this centralized database for the European Economic Area. It is one of the largest repositories of drug safety data in the world, facilitating the electronic exchange of individual case safety reports (ICSRs).
- Yellow Card Scheme (MHRA): In the UK, the Medicines and Healthcare products Regulatory Agency (MHRA) uses the Yellow Card system. A unique feature here is the “Black Triangle” (▼) symbol, which is assigned to new medicines or vaccines that are under intensive monitoring, encouraging healthcare providers to report even minor suspected side effects.
- Canada Vigilance Program: Health Canada’s post-market surveillance program collects reports of suspected adverse reactions to health products, including prescription and non-prescription medications, natural health products, and radiopharmaceuticals.
Navigating the FDA Adverse Event Reporting System (FAERS) Public Dashboard
The FDA’s Adverse Event Reporting System (FAERS) is perhaps the most significant repository of drug safety data available to the public. It contains millions of reports concerning adverse events, medication errors, and product quality complaints.
To make this data more accessible, the FDA launched the FAERS Public Dashboard. This interactive tool allows us to query human adverse event data in a user-friendly way. For example, if you want to see the reported side effects of a specific biologic over the last five years, the dashboard can visualize those trends instantly. Technically, the FAERS database is built on international standards. It adheres to the ICH E2B standards for safety reporting and uses MedDRA terminology (Medical Dictionary for Regulatory Activities) to code adverse events and medication errors. This standardization is what allows researchers in London, New York, and Singapore to speak the same “safety language.”
Occupational Hazards and the 2024 NIOSH List
Drug safety isn’t just about the patient; it’s about the people handling the medicine. The NIOSH List of Hazardous Drugs in Healthcare Settings, 2024 (DHHS Publication Number 2025-103) is the gold standard for protecting healthcare workers. This list, which was updated in December 2024 to supersede the 2016 version, identifies drugs that pose risks such as carcinogenicity, reproductive toxicity, or organ toxicity at low doses.
For nurses, pharmacists, and lab technicians, this list dictates the protocols for handling antineoplastic agents (chemotherapy) and other high-risk medications. If you are managing a pharmacy in Canada or the US, staying compliant with the latest NIOSH updates is a matter of workplace safety law. This includes implementing closed-system drug transfer devices (CSTDs) and ensuring proper personal protective equipment (PPE) is used during compounding and administration.
Understanding SDS and GHS in Chemical Safety Communication

While FAERS tells us what happens inside the body, Safety Data Sheets (SDS)—formerly known as Material Safety Data Sheets (MSDS)—tell us how to handle the substances safely in a lab or warehouse. These documents are mandated by the OSHA Hazard Communication Standard (HCS).
An SDS is a comprehensive 16-section document that provides a deep dive into the chemical’s profile. The sections are strictly organized to ensure that critical information is found in the same place every time:
- Identification: Product identifier and manufacturer contact info.
- Hazard Identification: GHS classifications and label elements.
- Composition/Information on Ingredients: Chemical identity and concentrations.
- First-Aid Measures: Necessary measures by route of exposure.
- Fire-Fighting Measures: Suitable extinguishing media.
- Accidental Release Measures: Containment and cleanup.
- Handling and Storage: Safe practices and incompatibilities.
- Exposure Controls/Personal Protection: PELs, TLVs, and PPE.
- Physical and Chemical Properties: Appearance, odor, pH, etc.
- Stability and Reactivity: Chemical stability and hazardous reactions.
- Toxicological Information: Routes of exposure, symptoms, and delayed effects.
- Ecological Information: Ecotoxicity and persistence.
- Disposal Considerations: Waste treatment methods.
- Transport Information: UN number and shipping names.
- Regulatory Information: Safety, health, and environmental regulations.
- Other Information: Date of preparation or last revision.
Decoding GHS Pictograms and Hazard Classifications
To ensure that safety information is understood regardless of language, the United Nations developed the Globally Harmonized System (GHS) guidelines. The GHS uses a set of standardized pictograms—symbols inside red-bordered white diamonds—to communicate specific hazards. These are accompanied by Hazard Statements (H-statements), which describe the nature of the hazard, and Precautionary Statements (P-statements), which describe recommended measures to minimize or prevent adverse effects.
These classifications cover:
- Physical Hazards: Such as explosives (Class 1), flammable gases, and corrosive substances that can eat through steel.
- Health Hazards: Including acute toxicity (substances that are fatal if swallowed), skin sensitization, and germ cell mutagenicity.
- Environmental Hazards: Risks to aquatic life or the ozone layer.
By using these universal symbols, the GHS ensures that whether a drug is being manufactured in Singapore or transported through Canada, the risks are clearly communicated to everyone in the supply chain.
Limitations and Challenges of Post-Market Surveillance Systems
As much as we rely on drug safety data, it is important to acknowledge that the current systems are far from perfect. One of the biggest hurdles we face in real-time pharmacovigilance is the “signal-to-noise” ratio. The FAERS and AEMS (Adverse Event Monitoring System) databases rely heavily on spontaneous reporting, which introduces several systemic biases.
The Challenges of Spontaneous Reporting
- The Weber Effect: This is a well-documented phenomenon where reporting of adverse events for a new drug tends to peak at the end of the second year after its introduction and then declines, regardless of the actual rate of adverse events. This can create a false sense of security for older drugs.
- Under-reporting: It is estimated that only a small fraction of actual adverse events are ever reported to regulatory bodies. This is particularly true for non-serious or well-known side effects.
- Incomplete Data: Many reports lack critical details like dosage, patient history, or concurrent medications. Without a complete clinical picture, it is difficult to determine if the drug in question was the primary cause of the event.
Why FAERS and AEMS Data Cannot Establish Causality
A common mistake is using drug safety data from public dashboards to calculate “incidence rates.” You simply cannot do this. Because FAERS doesn’t track the total number of people taking a drug (the denominator), we can’t determine how common a side effect actually is. Causation requires clinical verification and statistical rigor.
To address this, pharmacovigilance experts use quantitative signal detection methods, such as:
- Proportional Reporting Ratio (PRR): A measure used to determine if a specific adverse event is reported more frequently for a particular drug compared to all other drugs in the database.
- Empirical Bayes Geometric Mean (EBGM): A more complex statistical tool used by the FDA to identify “signals of disproportionate reporting” (SDRs).
These metrics do not prove causality, but they act as a “smoke detector.” If the PRR for a specific drug-event combination exceeds a certain threshold, it triggers a formal safety review. This “lifecycle monitoring” approach ensures that we catch rare events that clinical trials—which usually involve a few thousand people—simply aren’t large enough to see.
Advanced Tools for Monitoring Drug Shortages and Labeling Changes
To stay ahead of these risks, we use tools like the FDA-TRACK Patient Safety Tools dashboard. This platform provides transparency into how the Center for Drug Evaluation and Research (CDER) monitors safety after a drug hits the market.
Key tools in this ecosystem include:
- REMS (Risk Evaluation and Mitigation Strategies): These are extra safety measures for high-risk drugs. For example, a drug might only be dispensed if the patient and doctor are enrolled in a specific monitoring program.
- FDALabel: A database that allows you to search the full text of drug labels, including side effects and clinical trial results.
- Drug Shortages Database: Essential for ensuring that patients don’t lose access to life-saving treatments due to manufacturing issues.
Specialized Drug Safety Data for Specific Conditions
For some patients, general drug safety data isn’t enough. Take, for example, the American Porphyria Foundation’s drug database. Acute Porphyria is a rare condition where certain drugs can trigger life-threatening attacks.
The Foundation uses a unique rating system:
- OK!: Very likely safe based on consistent evidence.
- BAD!: Very likely to trigger an attack.
- NO INFO: Not enough data to make a recommendation.
This is a perfect example of how generic vs. brand-name data and drug class groupings (medicines with similar chemical structures) are used to provide life-saving guidance for specific patient populations.
The Future of Pharmacovigilance: AI and NLP Integration
The future of drug safety isn’t in more spreadsheets; it’s in smarter algorithms. Most drug safety data today is “unstructured”—it’s buried in doctor’s notes, call center transcripts, and even social media posts. This is where AI for pharmacovigilance comes in.
Natural Language Processing (NLP) in Safety Monitoring
Emerging technologies like Natural Language Processing (NLP) allow us to scan millions of lines of text to find adverse events that would otherwise go unnoticed. Key techniques include:
- Named Entity Recognition (NER): Automatically identifying drug names, dosages, and symptoms within a block of text.
- Relationship Extraction: Determining if the text suggests a link between the drug and the symptom (e.g., “Patient developed a rash after taking Drug X”).
- Sentiment Analysis: Monitoring patient forums to gauge the severity and impact of side effects on quality of life.
For instance, Adverse Drug Event Detection with NLP tools can automatically correlate specific symptoms with drug entities across social media, providing a “real-world evidence” layer that traditional reporting misses. This allows for the detection of “soft signals” long before they appear in formal regulatory reports.
The Power of Federated AI
At Lifebit, we take this a step further with federated AI. One of the biggest barriers to global drug safety is data privacy. Regulations like GDPR in Europe and HIPAA in the US make it difficult to move patient data across borders for analysis. Federated learning solves this by moving the AI model to the data, rather than the data to the model.
This allows biopharma companies and governments to analyze multi-omic data integration and safety signals across global borders—from London to Singapore—without compromising security. By training models on diverse, decentralized datasets, we can identify how genetic variations (pharmacogenomics) influence drug safety, leading to truly personalized medicine where safety profiles are tailored to an individual’s genetic makeup.
Frequently Asked Questions about Drug Safety
How can I report an adverse event to the FDA?
You can use the MedWatch online reporting form. Consumers and healthcare professionals can voluntarily report serious adverse events, product quality issues, or medication errors. For the pharmaceutical industry, reporting certain adverse events is mandatory and must follow strict regulatory timelines (e.g., 15-day alert reports for serious and unexpected events).
What is the difference between SDS and MSDS?
The primary difference is the format. MSDS (Material Safety Data Sheet) was the old, non-standardized format that varied by manufacturer and country. SDS (Safety Data Sheet) is the new, standardized 16-section format required by the GHS and OSHA’s International Hazard Communication Standard. SDS makes it much easier to find critical information quickly during an emergency because the layout is always identical.
Who is responsible for drug safety within a pharma company?
In many regions, including the EU, companies must appoint a Qualified Person Responsible for Pharmacovigilance (QPPV). This individual is legally responsible for the establishment and maintenance of the company’s pharmacovigilance system and must be available 24/7 to respond to regulatory inquiries.
How does GDPR affect drug safety data?
GDPR (General Data Protection Regulation) requires that all personal health data used in safety reporting be handled with extreme care. While safety reporting is a legal obligation that often overrides the need for individual consent, data must still be pseudonymized or anonymized whenever possible to protect patient privacy during the analysis of safety signals.
How do I access raw FAERS data for research?
If the public dashboard isn’t detailed enough for your research, you can download FAERS Quarterly Data Files. These are raw extracts available in ASCII or SGML formats. Be warned: you’ll need experience with relational databases (like MySQL or Oracle) or SAS to analyze them. Alternatively, you can submit a FOIA (Freedom of Information Act) request for specific case reports or product summaries.
Conclusion: Stop Chasing Data. Start Finding Signals.
The sheer volume of drug safety data generated every day is overwhelming. But in that noise lies the key to better patient outcomes and safer medicines. The old way of doing things—manual reporting, siloed databases, and reactive monitoring—is no longer enough.
We believe that the next generation of safety will be built on AI-driven pharmacovigilance solutions. By using the Lifebit Federated Biomedical Data Platform, researchers can securely access global data, integrate multi-omic insights, and detect safety signals in real-time.
Whether you are a regulator in Europe, a researcher in New York, or a healthcare provider in Singapore, the goal remains the same: ensuring that the right medicine reaches the right patient safely.
Ready to transform how you handle safety data? Learn how Lifebit’s federated platform can scale your pharmacovigilance today.

